Clear Sky Science · en
Machine learning-driven discovery of therapeutic nucleoside hydrogels for periodontitis
Why smart gels for sore gums matter
Gum disease is one of the most common health problems worldwide, yet current treatments often struggle to deliver medicine exactly where it is needed and keep it there long enough to work. This study describes how researchers used computer learning tools to design tiny, drug-like building blocks that can assemble into soft gels. These gels are placed around teeth to fight harmful mouth bacteria and protect the bone that holds teeth in place.
From random trial to guided design
Soft water-rich gels made from small molecules have long been seen as promising materials for drug delivery and tissue repair. In particular, gels built from nucleosides, the same family of molecules that form DNA and RNA, are attractive because they blend well with living tissue and can heal minor damage to their structure. Until now, useful versions of these gels were often found by chance or by slowly tweaking chemical groups, a slow and uncertain process. The authors set out to replace much of this guesswork with a more systematic path that predicts which nucleoside molecules can form gels and how they might behave in the body.
Teaching computers to spot useful molecules
The team gathered large public datasets describing how thousands of small molecules behave in biological tests, including toxicity, antibacterial power, antiviral action, anti inflammatory effects, and activity against tumor cells. For each molecule they calculated thousands of numerical features that encode aspects of its structure. Using several types of machine learning models, they trained computers to link these structural features to nine different biological activities. To avoid overfitting and improve accuracy, they carefully filtered and selected the most informative features before building the models.
Balancing safety, strength, and antibacterial power
Designing a gel for gum disease requires more than killing germs. The material must be safe for human cells and able to form a stable, injectable gel that can stay in the gum pocket. To juggle these needs, the researchers introduced two new scoring tools. The Molecular Bioactivity Specificity Index highlights which single activity a molecule is particularly good at, such as targeting a key gum disease bacterium while remaining quiet on other fronts. The Composite Molecular Attribute Score weighs several properties at once, ranking molecules that best combine strong gel formation, low toxicity, and high antibacterial potential.
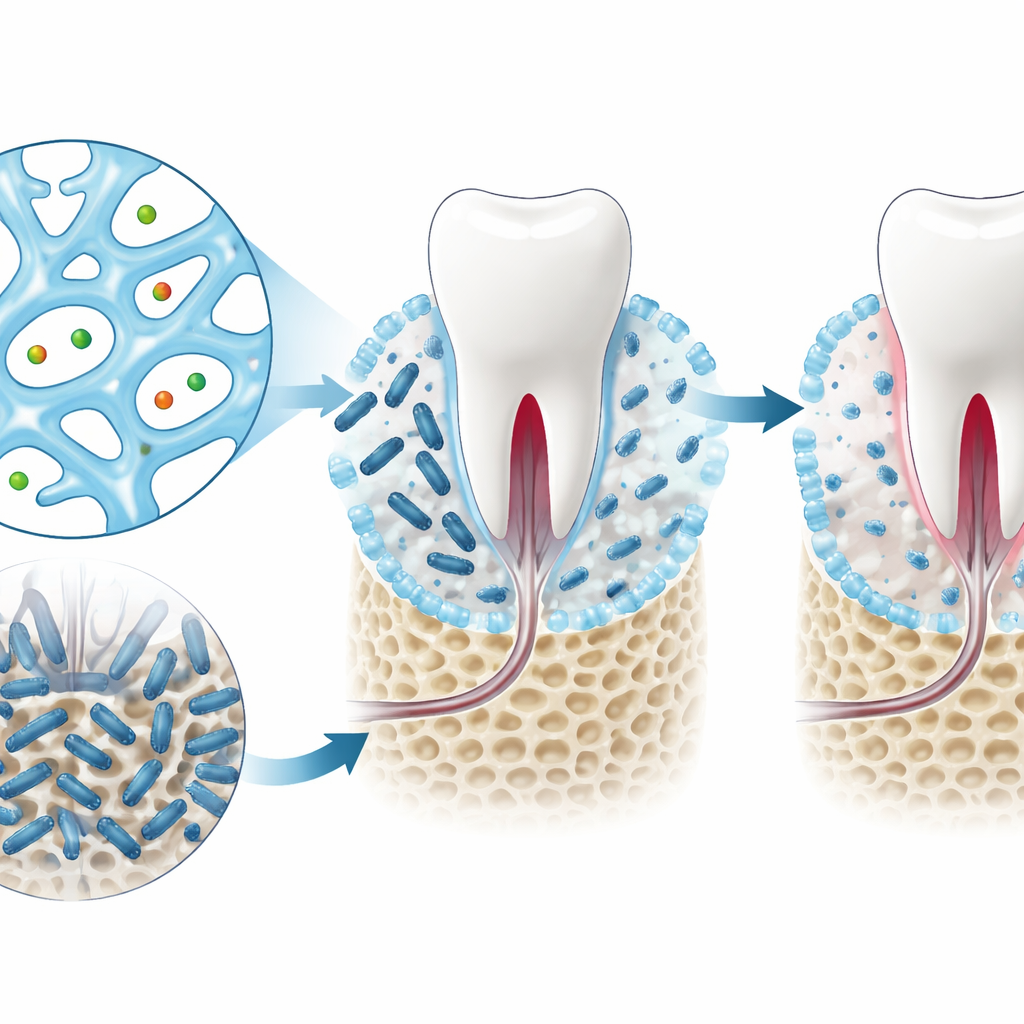
From computer hits to working hydrogels
Using their models and scoring system, the authors screened more than seven thousand nucleoside based candidates and narrowed the list to four that could realistically be made in the lab. Among these, two related compounds, known as GMP and dGMP, stood out once tested. Mixed with silver ions, they formed soft, porous gels with good mechanical strength and the ability to recover their structure after being disturbed. In lab dishes, both the free molecules and their gel versions sharply reduced the growth of Porphyromonas gingivalis, a major bacterium linked to periodontitis, while remaining gentle to mammalian cells.
Testing gum protection in living animals
The researchers next tested the gels in mouse models of gum disease. In one set of experiments, they treated established periodontitis by injecting the gels into gum pockets after infection with P. gingivalis. In another, they used the gels shortly after infection to see if they could prevent damage from developing. In both cases, scans of the jawbone showed that treated mice lost less supporting bone around their teeth and maintained denser, healthier bone structure. Tissue slices from the gums revealed reduced signs of inflammation and higher levels of markers tied to healing and tissue repair. Importantly, checks of major organs and injection sites showed no clear tissue damage or ongoing inflammation.
What this means for future oral care
To a lay reader, the key message is that the team built a smarter way to design soft, medicine carrying gels for the mouth by letting computers learn from large chemical and biological datasets. Their approach pinpointed two nucleoside based gels that can safely fight a major gum disease bacterium and help preserve the bone around teeth in mice. While more work is needed before these materials reach the clinic, the study suggests that machine learning guided design could speed the development of targeted treatments for gum disease and perhaps other oral conditions.
Citation: Li, W., Wen, Y., Huang, Z. et al. Machine learning-driven discovery of therapeutic nucleoside hydrogels for periodontitis. Int J Oral Sci 18, 41 (2026). https://doi.org/10.1038/s41368-026-00438-3
Keywords: machine learning, periodontitis, hydrogels, oral microbiome, biomaterials