Clear Sky Science · en
Detecting chromatin state alterations in PBMCs associated with Type 2 Diabetes Mellitus
A New Window into Diabetes Through Blood Cells
Type 2 diabetes is usually tracked with blood sugar numbers and long-term measures like HbA1c. But these tests say little about how a person’s immune system is coping with the disease, even though low-level inflammation and immune stress are central to many complications. This study explores a different kind of diabetes check-up: using advanced microscopy and computer analysis to look at how DNA is packed inside immune cells, with the goal of creating a simple, scalable test that could flag early changes as people move from health to prediabetes to diabetes.
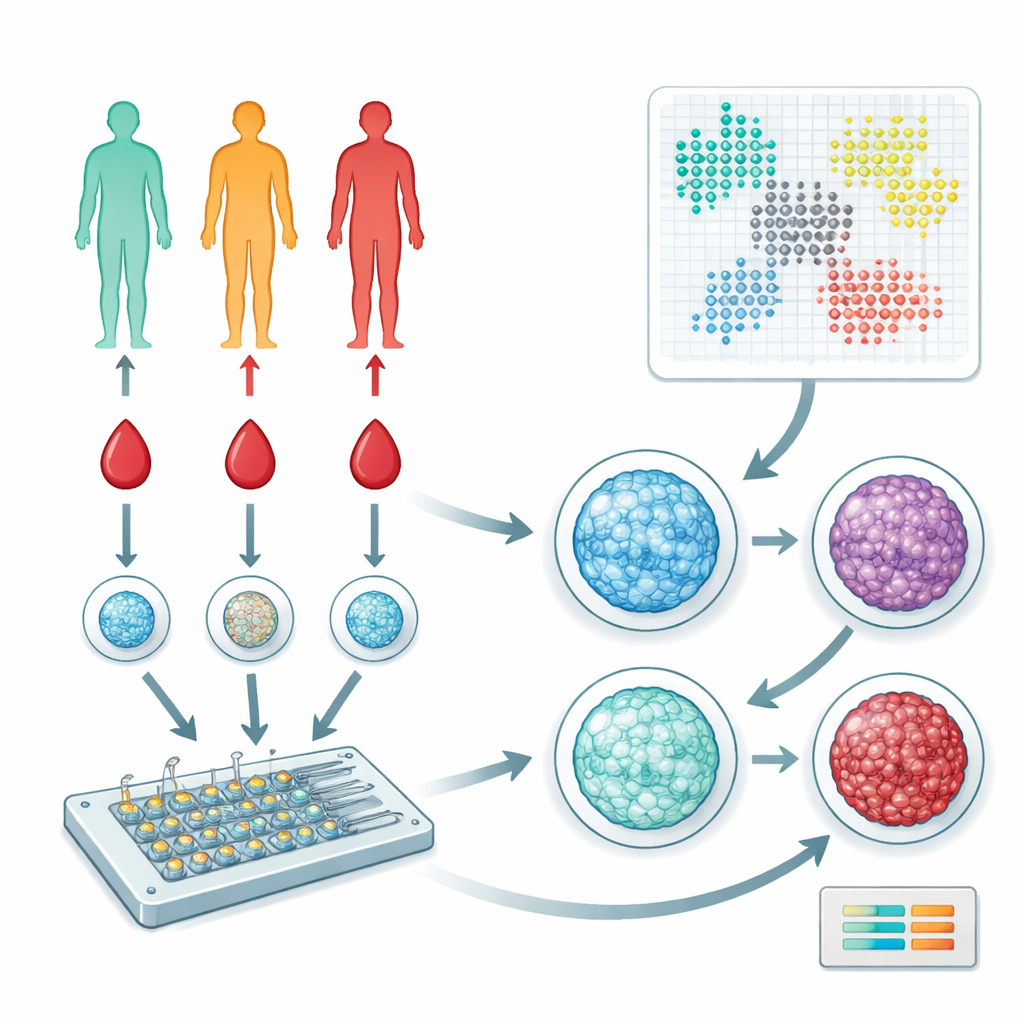
Looking at Blood Cells in a New Way
The researchers focused on peripheral blood mononuclear cells, a mixed group of immune cells that circulate in our blood and help coordinate responses throughout the body. They collected blood from 57 people spanning healthy, prediabetic, and type 2 diabetic stages. Instead of sequencing genes or measuring dozens of proteins, the team used a microfluidic device and fluorescent dyes to image how DNA is arranged inside the cell nucleus. This DNA–protein complex, called chromatin, can be more open or more condensed, and its organization subtly reflects how a cell is functioning and what signals it has received.
Turning Images into Patterns
To make sense of tens of thousands of nuclear images, the team used a type of artificial intelligence called a variational autoencoder. This software compresses each image into a set of numerical features that capture nuclear size, shape, and the fine-grained texture of chromatin. They then applied graph-based clustering methods to group nuclei into distinct “states” that correspond to different immune cell subpopulations or activation levels. Parallel analyses with carefully designed shape and texture measurements (handcrafted features) provided an independent check on the AI-based results, and both approaches converged on a set of chromatin patterns that recur across individuals.
Distinct Cell States Track Disease Stage
When the authors compared these chromatin-based clusters across health, prediabetes, and diabetes, clear shifts emerged. Certain nuclear states were enriched in healthy individuals, others in people with prediabetes, and still others in those with diabetes. Features that described where dense chromatin pockets sat within the nucleus—close to the edge or shifted inward, tightly grouped or more separated—were especially informative. Using only the fraction of a person’s cells falling into each cluster, a standard machine-learning model could distinguish diabetic from healthy donors and, with even higher accuracy, diabetic from prediabetic donors. These predictions held up in tests on held-out and independently split subsets of the cohort, suggesting that chromatin organization carries robust information about disease progression at the level of individual patients.
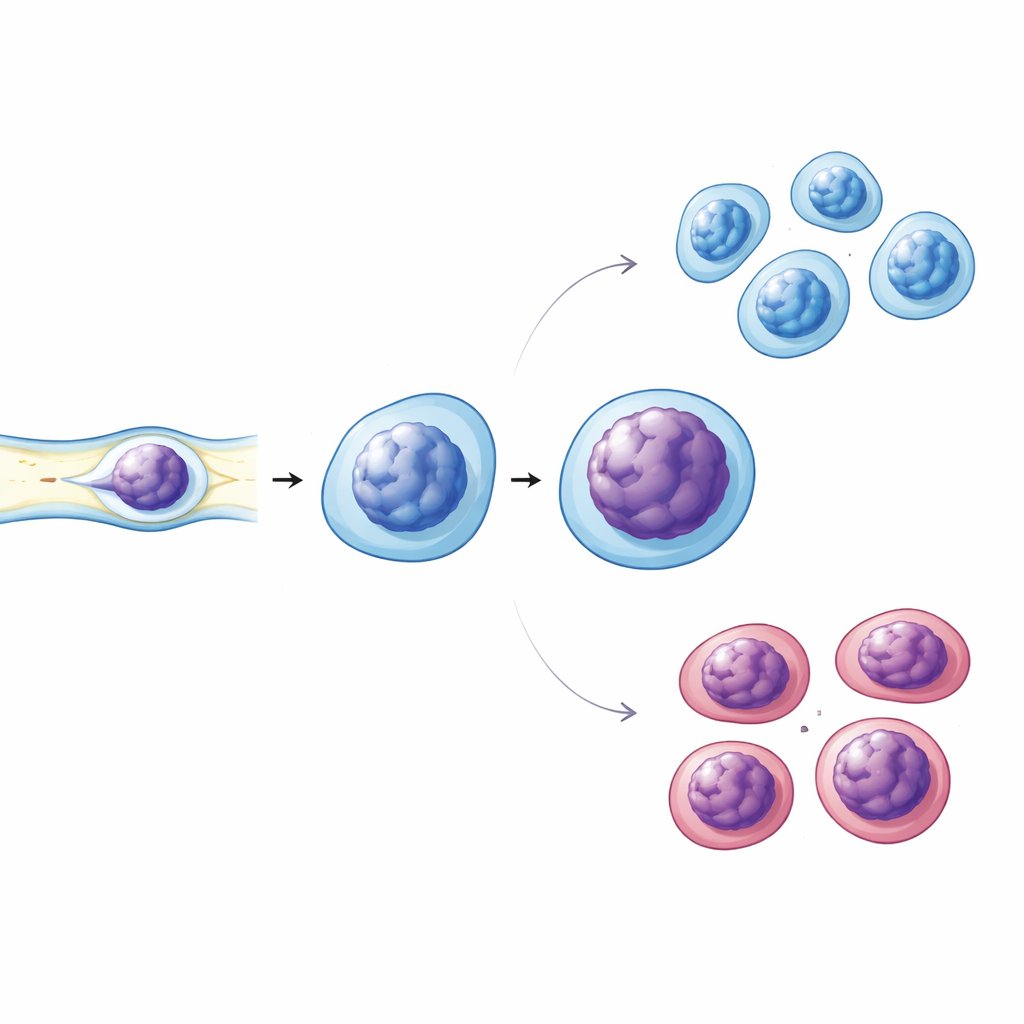
Mechanical Stress Reveals Subtle Weaknesses
Immune cells constantly squeeze through narrow spaces in blood vessels and tissues, so the team also asked whether their mechanical behavior changes in diabetes. They used narrow microchannels to gently compress live immune cells while imaging their nuclei. Diabetic cells showed more pronounced nuclear deformation and signs of altered chromatin condensation compared with cells from healthy and prediabetic donors. Follow-up staining experiments revealed that diabetic nuclei had less of a key structural protein, Lamin A/C, which helps maintain nuclear stiffness and shape, and they showed slightly higher levels of tightly packed chromatin marks. The fraction of cells bearing an activation marker on their surface was also higher in diabetes, linking these nuclear changes to heightened immune activation.
From Lab Insight to Future Clinic Tool
Taken together, the work shows that simple fluorescent imaging of chromatin in routine blood samples can capture meaningful, patient-specific changes in the immune system as type 2 diabetes progresses. While this approach does not yet replace standard glucose or HbA1c tests, it offers a complementary readout of immune stress and cell function—information that today’s clinical markers largely miss. Because the assay relies on relatively low-cost dyes and microfluidic devices, the authors argue it could eventually be scaled and automated for clinical use, enabling more personalized tracking of diabetes and its complications.
Citation: Afarani, M.M., Gupta, R., Uhler, C. et al. Detecting chromatin state alterations in PBMCs associated with Type 2 Diabetes Mellitus. Commun Med 6, 268 (2026). https://doi.org/10.1038/s43856-026-01513-w
Keywords: type 2 diabetes, immune cells, chromatin imaging, biomarkers, machine learning