Clear Sky Science · en
In vitro γ-aminobutyric acid A (GABAA) receptor activity and binding interactions at the α+/γ2– interface of 53 prescription and designer benzodiazepines
Why this matters for everyday life
Benzodiazepines are among the most widely used medicines for anxiety, insomnia, and seizures, but many are also taken recreationally or misused, sometimes with deadly consequences. In recent years, a wave of “designer” versions has appeared on the illicit market, often sold as look‑alike pills with little or no medical testing. This study asks a simple but urgent question: how strongly do these new compounds affect the brain’s main calming system, and how do small tweaks in their chemical structure change their effects?
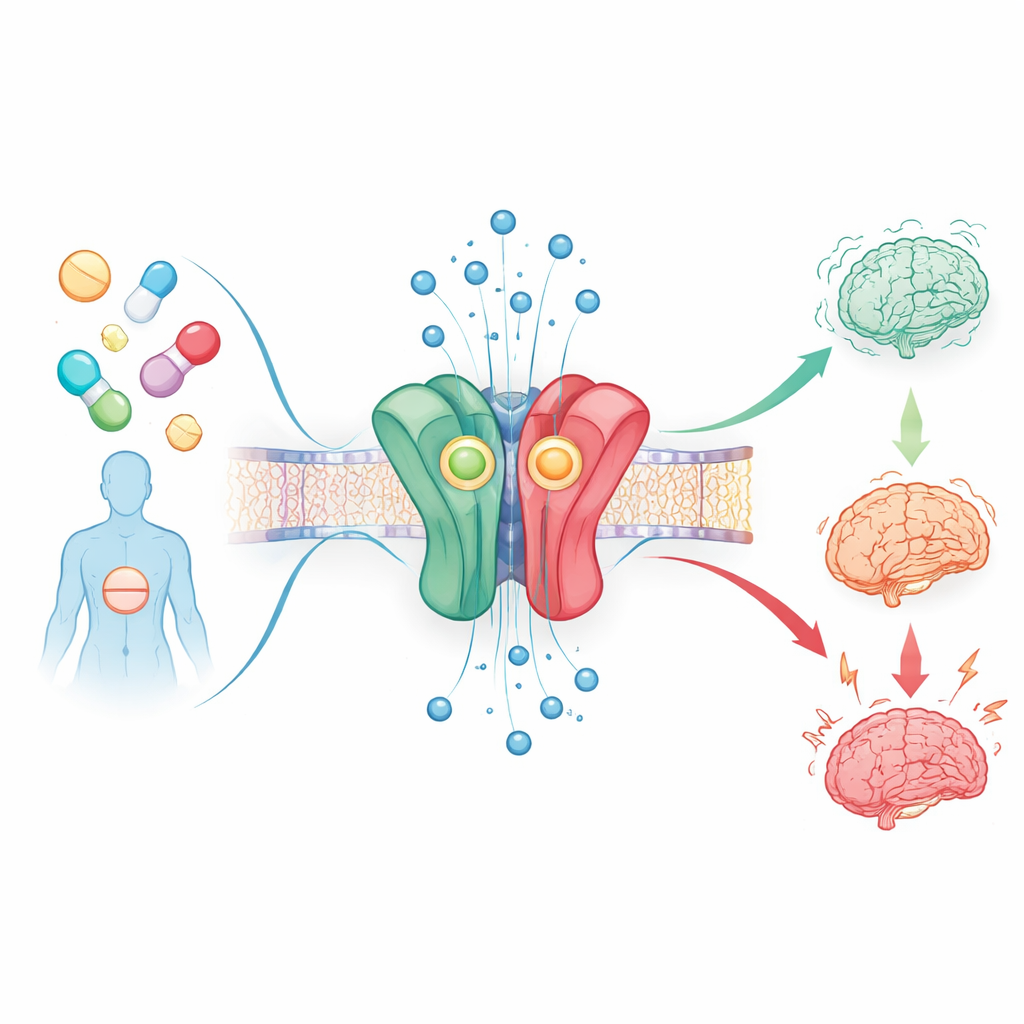
The brain switch these drugs act on
All of the drugs in this study act on the same key target in the brain: the GABAA receptor, a protein channel that lets chloride ions flow into nerve cells and dampen their activity. Ordinary benzodiazepines attach to a particular contact point on this receptor and make the channel open more readily when the natural messenger GABA is present, which is why they reduce anxiety and promote sleep. The researchers used a sophisticated cell‑based test to measure how 53 different prescription and designer benzodiazepines influence this receptor subtype, known as α1β2γ2, by tracking the tiny electrical currents that flow when the channel opens.
A wide range of strength and direction
The team found that most of the compounds boosted the receptor’s activity to some degree, but not all in the same way. Some designer drugs were up to about 60 times more potent than diazepam, meaning only a trace amount was needed to have a strong effect in the test. Others barely did anything at all. Two related compounds stood out for doing the opposite of what most benzodiazepines do: instead of enhancing the calming current, they weakened it. These “negative modulators” are important because they could produce very different experiences and risks than users expect when they think they are taking a standard sedative.
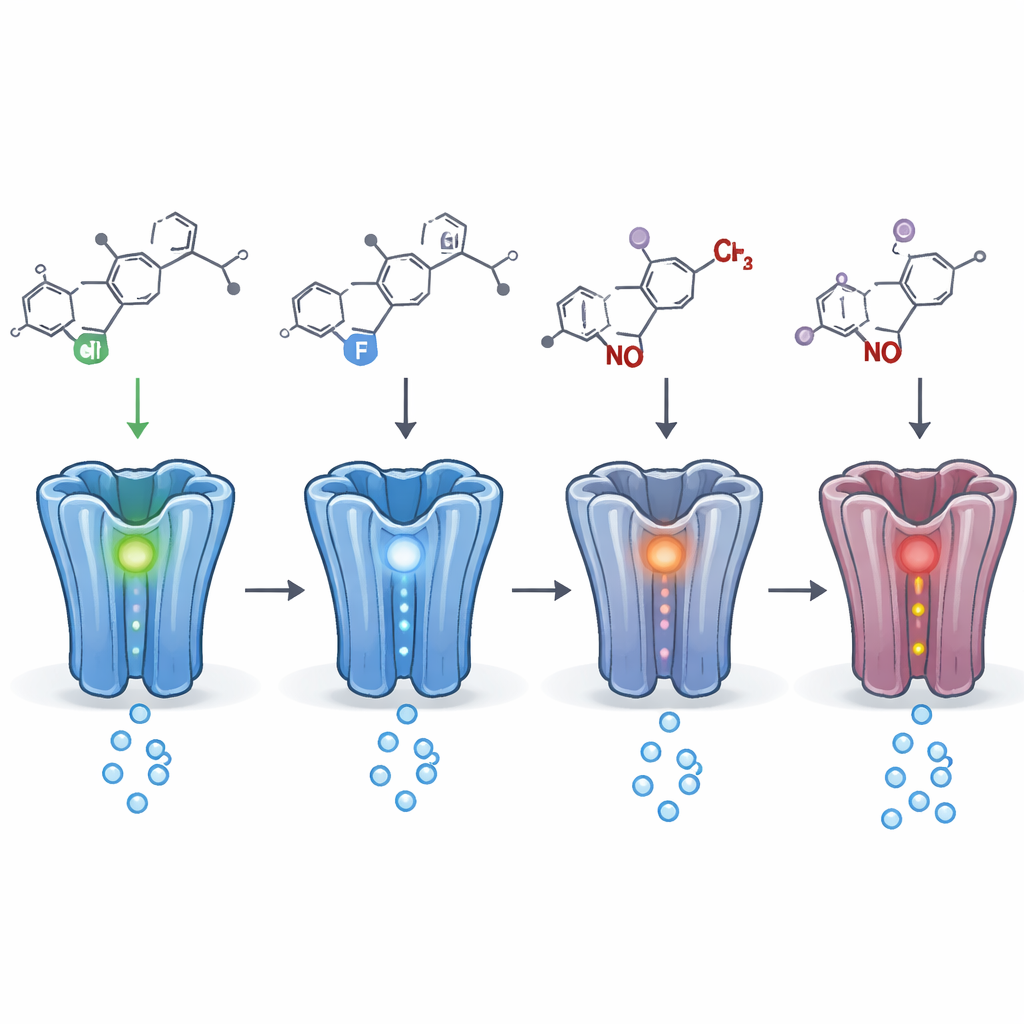
Tiny chemical changes, big functional shifts
By comparing many close chemical relatives, the authors could map which small structural changes mattered most. Swapping one halogen atom (such as chlorine, fluorine, or bromine) for another at specific positions often changed how powerful a drug was at the receptor. In some cases, moving a single halogen from one spot on an attached ring to another flipped a drug from a calming enhancer to an inhibitor. Changes at other positions, such as adding or removing small side chains or making “prodrug” versions that are converted in the body, tended to reduce or delay activity but rarely removed it entirely. Metabolites—the breakdown products formed in the body—almost always retained meaningful activity at the receptor, and sometimes were as strong or stronger than the parent drug.
Hidden binding sites and mixed signals
To find out where on the receptor these drugs were acting, the researchers added flumazenil, a medicine that blocks the classic benzodiazepine site and is used to reverse overdoses. For the majority of compounds, flumazenil wiped out their effects, confirming that their action comes mainly from this known site. But a handful of drugs still showed some boosting effect even in the presence of flumazenil, hinting that they may also interact with other contact points on the same receptor. The negative modulators, and a few compounds that seemed inactive at first glance, continued to reduce receptor activity even when the main site was blocked, showing that their inhibitory effects must be mediated elsewhere on the receptor surface.
What this means for users, clinicians, and policy
For a layperson, the core message is that not all benzodiazepine‑like pills are alike, even if they look similar or share a name. The designer versions now circulating on the street span a huge range of strength and can act through more than one contact point on the brain’s calming switch. Some are far more powerful than familiar medicines, some are weak, and a few even push in the opposite direction. Because many active metabolites also contribute, the overall effect can be stronger and longer‑lasting than expected. This work provides a detailed map linking small chemical tweaks to functional outcomes at a key brain receptor, helping toxicologists interpret poisonings, harm‑reduction groups advise on relative risk, and regulators anticipate how future designer drugs might behave before they reach widespread use.
Citation: Norman, C., Liin, S.I., Jauregi-Miguel, A. et al. In vitro γ-aminobutyric acid A (GABAA) receptor activity and binding interactions at the α+/γ2– interface of 53 prescription and designer benzodiazepines. Commun Chem 9, 155 (2026). https://doi.org/10.1038/s42004-026-02001-x
Keywords: designer benzodiazepines, GABAA receptor, drug safety, structure–activity relationships, recreational sedatives