Clear Sky Science · en
Macrophage efferocytosis promotes inflammation resolution and accelerates wound healing
Why our bodies struggle to close stubborn wounds
Chronic skin wounds, such as diabetic foot ulcers, can linger for months, causing pain, infections, and even amputations. This review article explains why some wounds refuse to heal and spotlights a little-known cleanup job done by immune cells called macrophages. When this cleanup falters, inflammation smolders instead of switching off, and the skin cannot rebuild itself properly. Understanding and boosting this natural clearance system could lead to new, less invasive treatments for hard-to-heal wounds.
The normal rhythm of wound repair
Healthy skin repair unfolds in four overlapping stages: stopping the bleeding, cleaning up damage, growing new tissue, and reshaping the scar. Right after injury, blood vessels clamp down and a clot forms, acting as both a plug and a scaffold. Soon after, a wave of immune cells floods in. First responders called neutrophils kill invading microbes and digest debris. They are followed by monocytes that mature into macrophages, which adapt their behavior over time: early on they are fiery and attack-oriented, later they become soothing and repair-focused. As the danger passes, skin cells, blood vessel cells, and connective tissue cells proliferate and migrate to close the gap, while collagen fibers are gradually reorganized to give the new tissue strength.
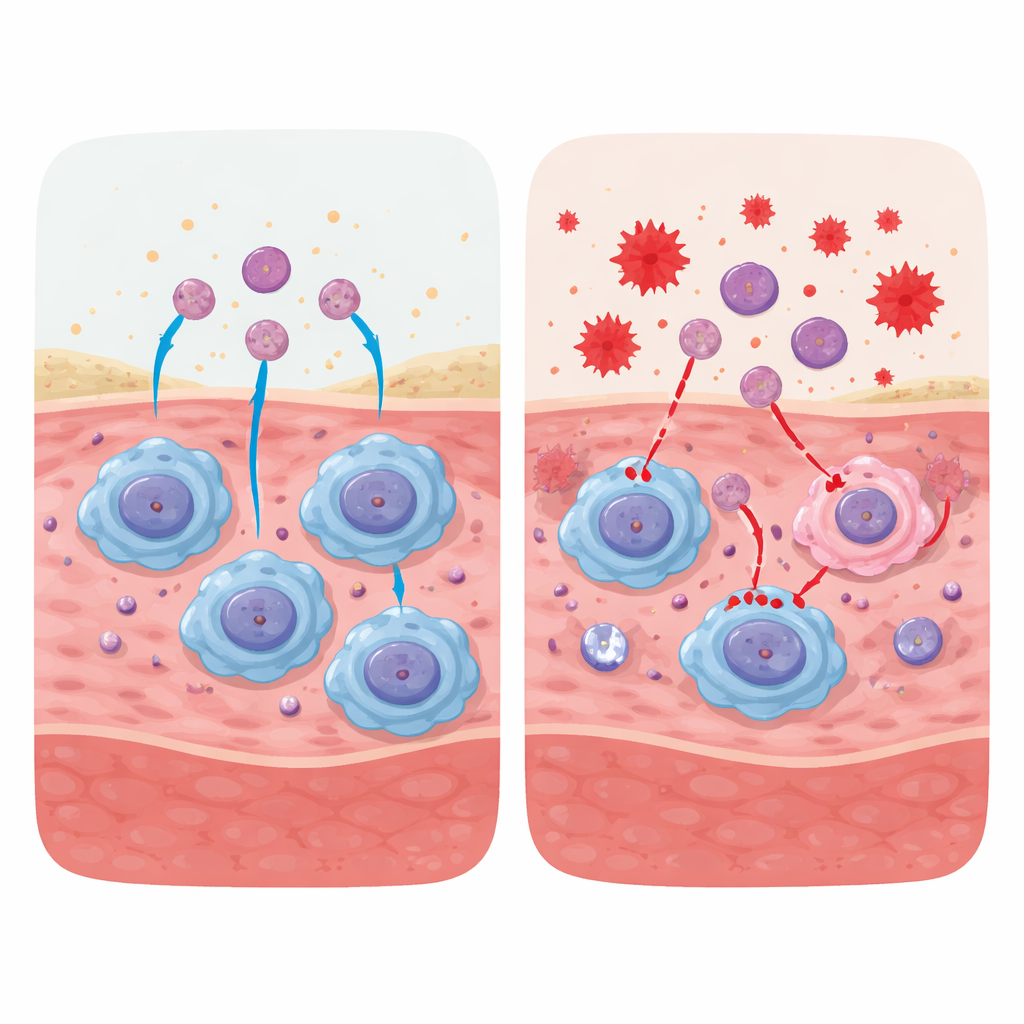
When cleanup fails and wounds get stuck
In diabetes, aging, radiation injury, and other chronic conditions, wounds often stall in the inflammatory phase. High blood sugar, persistent oxidative stress, and danger signals from damaged molecules disrupt how macrophages mature and behave. Instead of shifting into a repair mode, these cells remain locked in a pro-inflammatory state and build up at the wound site. At the same time, their ability to engulf and digest dying cells is weakened. As a result, dead and dying neutrophils accumulate, can burst open, and spill irritant contents. Extra web-like structures from neutrophils and a flood of inflammatory molecules further poison the local environment. The wound enters a vicious cycle: ongoing inflammation prevents proper tissue rebuilding, which in turn generates more cell damage and inflammation.
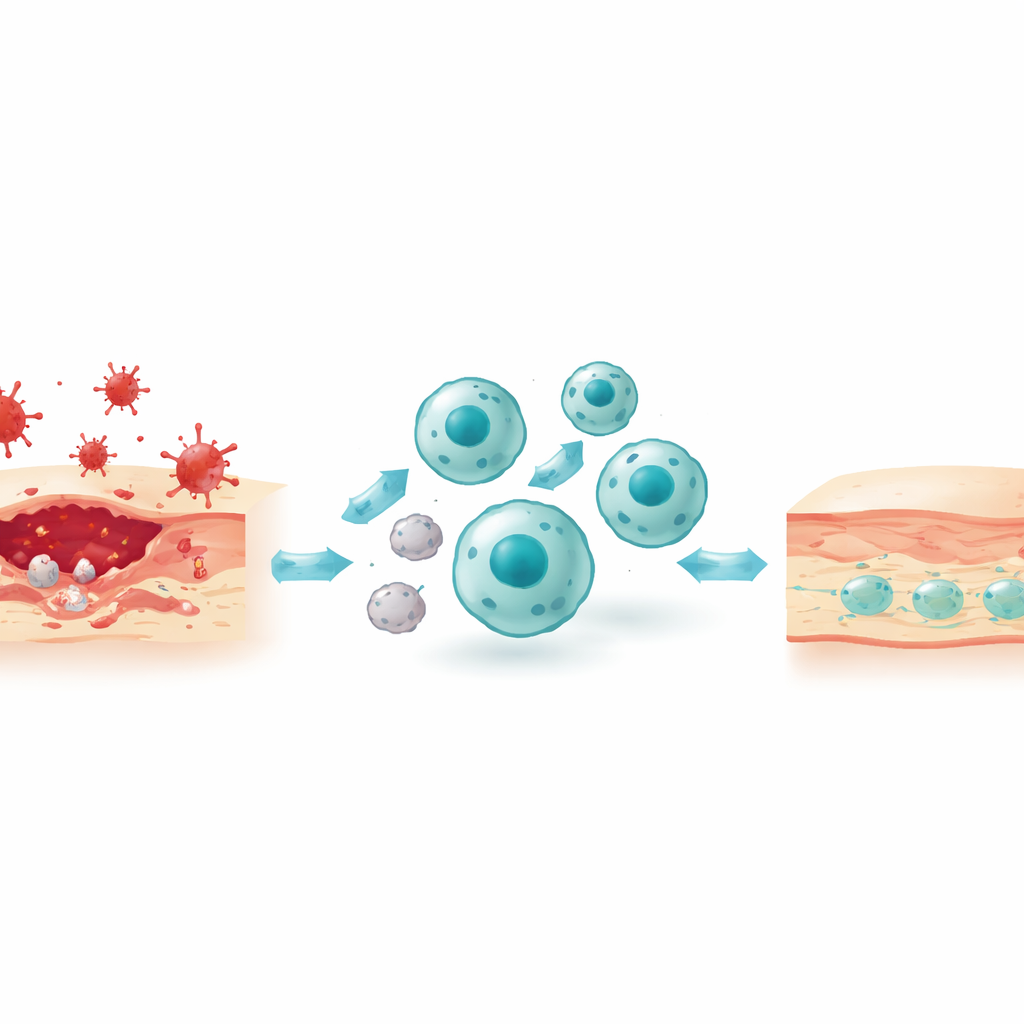
The hidden process of cellular housecleaning
A central theme of the paper is efferocytosis, the programmed removal of dying cells by macrophages. This is not a simple one-step gulp. First, apoptotic (programmed-to-die) cells emit “find-me” cues such as lipids and nucleotides that draw macrophages toward them. These dying cells also flip specific fats to their outer surface to act as “eat-me” flags, which are recognized either directly by receptors on macrophages or indirectly through “bridging” proteins in the surrounding fluid. Next, the macrophage reshapes its actin skeleton to wrap around and internalize the target, forming a sealed bubble that later fuses with enzyme-filled sacs for digestion. The breakdown products—fats, amino acids, nucleotides—are then recycled or exported, and in the process rewire macrophage metabolism toward a calmer, pro-resolving state that releases anti-inflammatory mediators and growth factors.
How better efferocytosis can rescue chronic wounds
The authors survey a wide array of molecular switches that can enhance efferocytosis and thereby calm chronic inflammation. Some strategies boost early recognition, for example by increasing signals that attract macrophages or by stabilizing the “eat-me” flags on dying cells. Others focus on preserving or restoring key macrophage receptors that are often cut or downregulated in chronic disease, or on supplying bridging molecules that connect dying cells to these receptors. Additional approaches act deeper inside the cell, nudging the energy system toward glycolysis, fine-tuning cholesterol and amino acid handling, or improving the fusion of ingestion bubbles with lysosomes. Specialized lipid mediators derived from omega-3 fatty acids also form a positive feedback loop: they are produced during efferocytosis, further enhance this cleanup, and help resolve inflammation. Together, these mechanisms show that improving cell clearance is not merely about waste disposal; it actively reprograms macrophages to support blood vessel growth, matrix remodeling, and orderly tissue repair.
New treatment ideas: helping the helpers
Beyond drugs and biomolecules, the review highlights emerging cell-based and material-based therapies. Infusing a patient’s own peripheral blood mononuclear cells into non-healing wounds can replenish monocytes that mature into reparative macrophages, restoring balance between inflammatory and healing phases. Smart biomaterials and dressings can be engineered to deliver efferocytosis-boosting signals, growth factors, or anti-oxidant compounds directly to the wound bed. The authors also discuss how targeting mitochondria, taming excessive neutrophil structures, and managing cellular senescence may all converge on improving macrophage function.
What this means for patients with hard-to-heal wounds
The article concludes that failed cell cleanup is a common thread linking many types of chronic wounds. Macrophage efferocytosis acts as a master switch: when it works, inflammation is turned off at the right time and the wound moves on to rebuilding; when it fails, the tissue remains trapped in a damaging limbo. By designing therapies that restore or amplify this natural housecleaning—whether through molecules, cells, or smart materials—clinicians may be able to shorten healing times, reduce complications, and improve quality of life for people living with stubborn skin wounds.
Citation: Gao, J., Zhu, D., Wang, J. et al. Macrophage efferocytosis promotes inflammation resolution and accelerates wound healing. Commun Biol 9, 613 (2026). https://doi.org/10.1038/s42003-026-10107-0
Keywords: wound healing, macrophages, inflammation, diabetic foot ulcers, efferocytosis