Clear Sky Science · en
MCT4 deficiency suppresses tumor incidence and metastasis by downregulating IGF1 expression and enhancing anti-tumor immunity
Why this cancer study matters
Cancer cells rewire how they use energy, and this metabolic makeover not only fuels tumors but can also help them hide from the immune system. This study focuses on a single transport protein, MCT4, that pumps out the waste product lactate from fast‑growing cells. By creating mice that completely lack MCT4, the researchers ask a simple but powerful question: if we turn off this metabolic valve, do tumors grow more slowly and become easier for the immune system to attack?
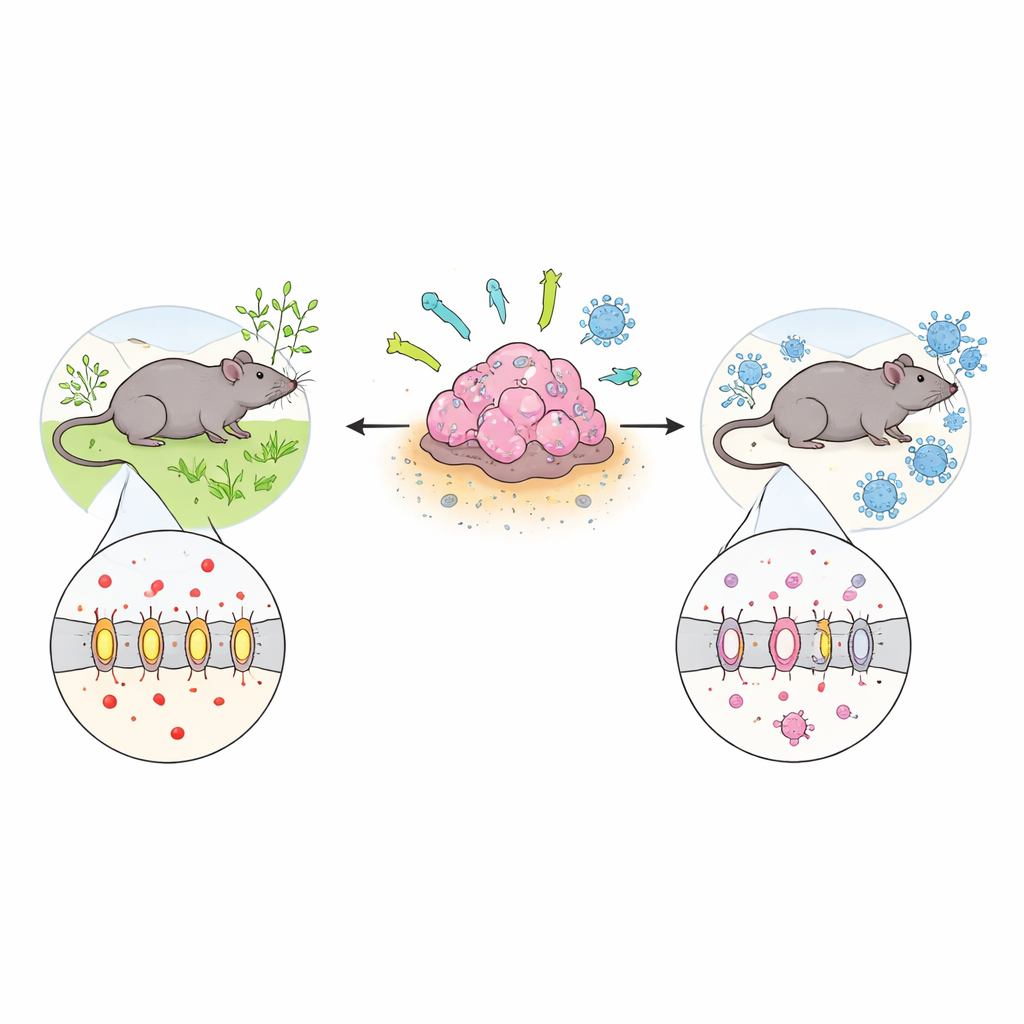
The energy shortcut that tumors exploit
Most healthy cells generate energy efficiently in the presence of oxygen. In contrast, highly active cells – including many cancer cells – prefer a quicker but "messier" route called glycolysis, which produces large amounts of lactate. To avoid internal acid buildup, tumor cells rely on transporters like MCT4 to pump lactate out into the surrounding tissue. In normal tissues MCT4 is found mainly in hard‑working cells such as skeletal muscle, but analysis of large cancer databases showed that many tumor types, including breast, lung, brain, and head‑and‑neck cancers, switch MCT4 on at high levels. Patients whose tumors have more MCT4 tend to have worse survival, suggesting this transporter is not just a passenger but an active driver of aggressive disease.
Building mice without the lactate valve
To probe MCT4’s role directly, the team used gene‑editing tools to generate BALB/c mice in which the MCT4 gene is completely deleted. These animals were healthy overall, but slightly smaller and had modestly higher blood sugar, hinting that MCT4 also influences whole‑body metabolism. When breast cancer (4T1) or lung cancer (M109) cells were transplanted into these mice, tumors grew about 40 percent more slowly than in normal mice and formed fewer lung metastases. Tumor samples from normal mice ramped up MCT4 protein during growth, whereas tumors in knockout mice kept MCT4 low. Across multiple chemical cancer‑induction models – for breast, lung, and oral cancers – MCT4‑deficient mice consistently developed fewer, smaller, and less invasive lesions.
Growth signals and the tumor neighborhood
Digging deeper, the investigators measured levels of insulin‑like growth factor 1 (IGF1), a hormone that promotes cell growth and survival. In healthy MCT4‑knockout mice, IGF1 in the blood was about 45 percent lower than in normal animals, and both IGF1 and lactate were markedly reduced in the fluid surrounding tumors. When the researchers supplied extra IGF1 or lactate to the knockout mice, tumor growth partially bounced back, indicating that MCT4 supports cancer not only by handling lactate but also by boosting IGF1‑driven growth signals. Yet the rescue was incomplete, suggesting other pathways are involved.
Reawakening the immune attack
The study then turned to the immune system, which constantly patrols tissues for abnormal cells. Under baseline conditions, immune cell numbers in the blood looked similar in normal and MCT4‑deficient mice. But once tumors were present, differences became striking. Mice lacking MCT4 had more helper T cells, killer T cells, natural killer cells, and macrophages both in blood and within tumors. Their macrophages shifted from a tumor‑friendly, wound‑healing state (often called M2) toward a more inflammatory, tumor‑fighting state (M1). The team linked MCT4 activity to higher levels of the signaling molecule CCL28, which attracts immune‑suppressive myeloid‑derived suppressor cells. Knockout mice showed lower CCL28 in blood and tumor fluid and fewer of these suppressor cells, helping to tilt the balance toward an effective immune response.
The big picture for future cancer therapies
Taken together, the findings position MCT4 as a central switch connecting three hallmarks of cancer: altered metabolism, runaway growth signaling, and local immune suppression. Removing this transporter in mice lowers growth‑promoting IGF1, reduces lactate buildup, cuts off recruitment of suppressive immune cells, and allows protective T cells and natural killer cells to infiltrate tumors more effectively. Because mice without MCT4 live normal lives with only mild metabolic changes, the work suggests that carefully designed MCT4‑blocking drugs could offer a way to slow tumor growth and boost the body’s own anti‑tumor defenses at the same time, potentially complementing existing immunotherapies.
Citation: Wang, S., Guo, H., Feng, L. et al. MCT4 deficiency suppresses tumor incidence and metastasis by downregulating IGF1 expression and enhancing anti-tumor immunity. Commun Biol 9, 539 (2026). https://doi.org/10.1038/s42003-026-10002-8
Keywords: cancer metabolism, tumor immunity, lactate transporter, IGF1 signaling, MCT4 inhibition