Clear Sky Science · en
Metagenomic analysis of antimicrobial resistance, virulence, and mobile genetic elements in the gut microbiota of Caprinae species
Why goat and sheep guts matter for our health
The bacteria living in the intestines of farm animals do more than help them digest grass. They can also act as a hidden storehouse for genes that make bacteria harder to kill with antibiotics and potentially more harmful to people. This study dives deep into the gut microbes of goats and sheep and their relatives to see how many such genes are lurking there, how they are organized, and how easily they might move between species, including into humans.
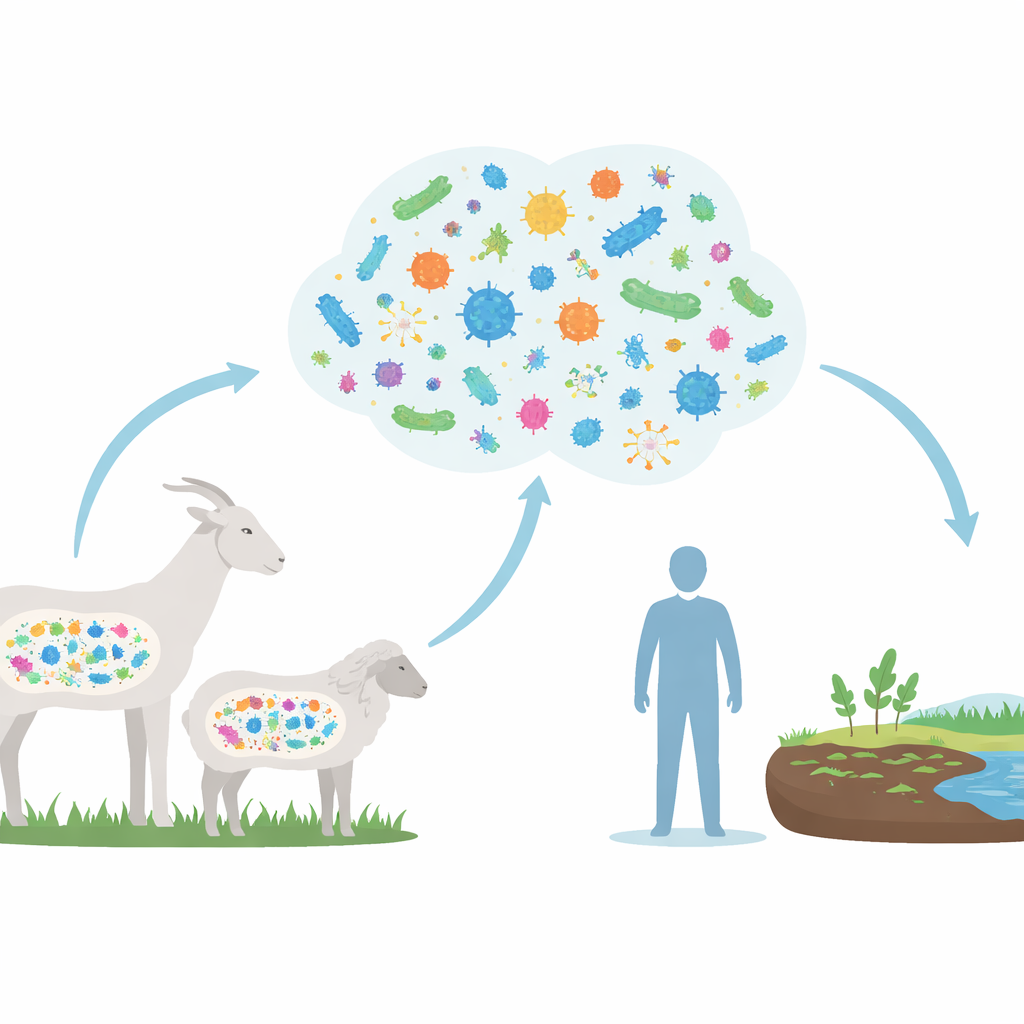
Looking inside the gut community
The researchers combined DNA data from 779 gut samples taken from goats, sheep, and a few wild Caprinae species. Instead of growing microbes in the lab, they used powerful computers to piece together more than 17,000 draft genomes directly from the mixed DNA in the samples. These genomes revealed an enormous variety of microbes, many of which have never been formally described before. Despite this diversity, goats and sheep shared a common core of gut residents, dominated by bacteria that thrive in herbivores and help them break down plant-rich diets.
Hidden stockpiles of resistance and harm
Within these microbial genomes, the team searched for two kinds of worrying genes: those that protect bacteria from antibiotics and those that help them infect or damage hosts. They found 2,440 antibiotic resistance genes spread across about one in twelve of the assembled genomes, with especially strong defenses against tetracycline and multiple antibiotic types at once. They also uncovered more than 5,400 virulence-related genes in about one in six genomes. Some strains of Escherichia coli and Pseudomonas aeruginosa stood out as heavy “gene hoarders,” each carrying dozens of resistance and virulence traits packed into single genomes.
Genes on the move
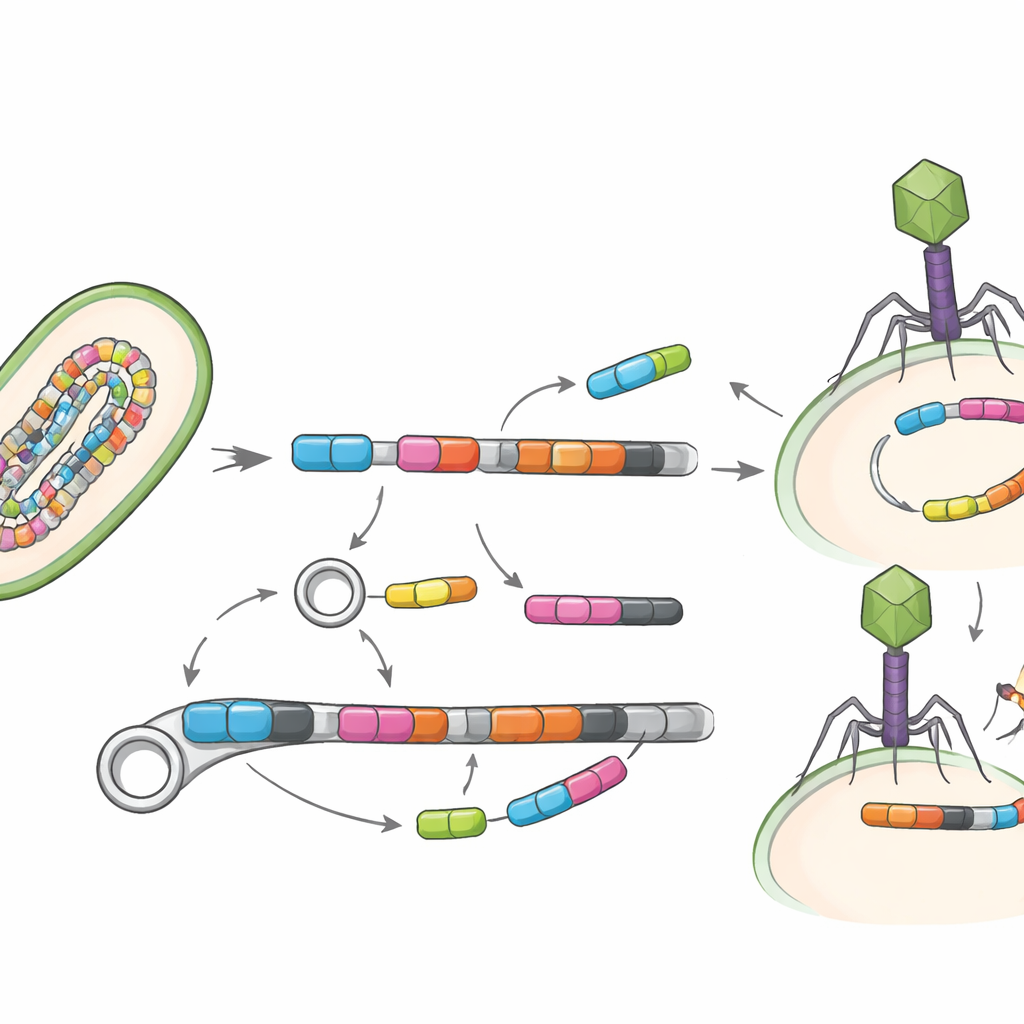
A key question is not just what genes are present, but how easily they can move. The study therefore looked for mobile genetic elements—DNA segments such as transposases and other “jumping” pieces that can shuttle genes between bacteria—and for viruses that infect bacteria, known as bacteriophages. Only a small fraction of genomes clearly carried these mobile elements, likely because such regions are hard to reconstruct, but where they did appear they often sat right beside resistance genes. The team identified 19 direct pairings of mobile elements with resistance genes, and even found three resistance genes embedded in viral genomes, suggesting that both DNA shuttles and gut viruses could help spread these traits.
Links between livestock and people
To understand what this means beyond the barn, the researchers compared resistance genes in Caprinae gut microbes with those found in thousands of human gut genomes. They discovered 184 types of resistance genes shared between animals and people, including ones tied to antibiotics considered last-resort treatments in hospitals, such as tigecycline and vancomycin. While this study does not prove that genes are moving directly from goats and sheep into human pathogens, the overlap shows that both hosts draw from a common pool of resistance. Given that humans, livestock, and the environment often mix closely—through manure, soil, water, and food—these shared genes represent possible routes for cross-species spread.
What this means for everyday life
Overall, the work paints Caprinae gut microbes as an important, and until now underappreciated, reservoir of antibiotic resistance and virulence potential. It shows that these genes cluster in certain bacterial strains, are often linked to mobile DNA, and can even be carried by viruses, all of which can help them persist and travel between hosts. For the lay reader, the takeaway is clear: how we manage antibiotics and livestock does not just affect farm animals; it shapes a shared genetic landscape that can influence the future effectiveness of life-saving drugs. The authors argue that goats, sheep, and related animals should be included in integrated “One Health” surveillance efforts that jointly consider human, animal, and environmental sources of antimicrobial resistance.
Citation: Su, JW., Elsheikha, H.M., Guo, L. et al. Metagenomic analysis of antimicrobial resistance, virulence, and mobile genetic elements in the gut microbiota of Caprinae species. Commun Biol 9, 456 (2026). https://doi.org/10.1038/s42003-026-09726-4
Keywords: antimicrobial resistance, gut microbiome, livestock, mobile genetic elements, bacteriophages