Clear Sky Science · en
Imaging mass cytometry unveils functional and spatial remodeling of peri-lesional cells in jaw osteonecrosis
Why jaw healing sometimes fails
Some people who take powerful drugs to protect their bones develop a puzzling problem: parts of their jawbone die and the overlying gum never fully heals. This condition, called medication‑related osteonecrosis of the jaw, can cause pain, infection, and open sores that linger for months. Doctors know which drugs are involved, but not why only a fraction of patients are affected or why the wounds refuse to close. This study peeks inside the living edge of these jaw lesions, mapping millions of cells and their locations to understand how normal repair processes go off track.
A close look at damaged jaw tissue
The researchers focused on the “peri‑lesional” zone—the rim of still‑living tissue that surrounds dead bone in affected jaws. They compared samples from patients with jaw osteonecrosis to healthy oral tissue taken during routine dental surgery. Using a technology called imaging mass cytometry, they stained each thin slice of tissue with a large panel of antibodies and then read out dozens of proteins in every single cell while preserving its exact position. This allowed them to separate the tissue into three main regions—surface lining (epithelium), supporting connective tissue (stroma), and blood and lymph vessels—and to identify immune cells, structural cells, and their activity states all at once. 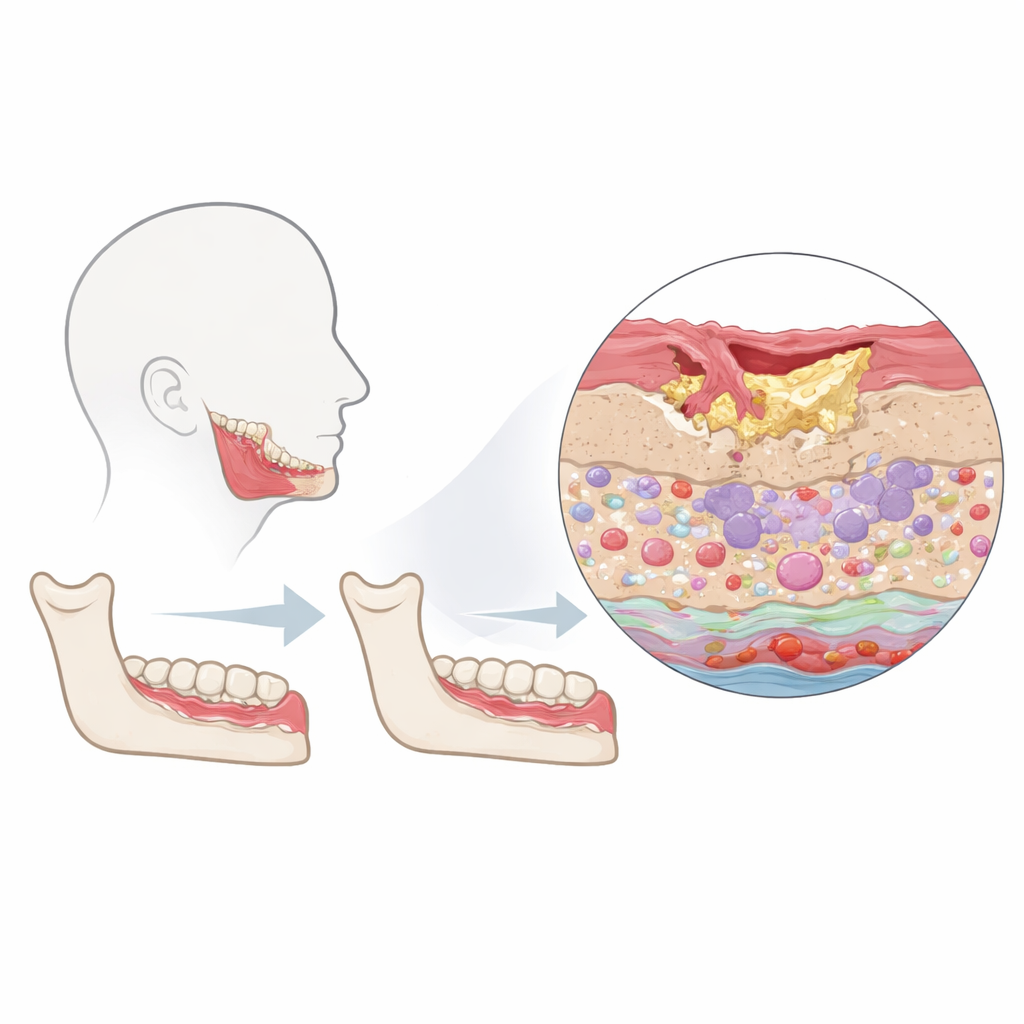
Immune cells gather but lose their guidance
In diseased jaws, immune cells of many types flooded all regions of the tissue—surface, stroma, and vessels—far more than in healthy controls. However, their organization changed in telling ways. Under normal conditions, many immune cells are scattered and show only gentle preferences for one another and for nearby tissues, forming a balanced surveillance network. In osteonecrosis, several cell types linked to immune suppression and chronic inflammation—regulatory T cells, so‑called “exhausted” T cells, and M2‑like macrophages—shifted from avoiding one another to forming tight local clusters, especially around vessels. Other immune pairings that normally worked closely with the epithelium became more distant. This rearrangement suggests that instead of coordinating an efficient clean‑up, the immune system becomes patchy and locally suppressive, potentially allowing infection and damage to persist.
Stressed tissues trying, and failing, to repair
The lining of the mouth over osteonecrotic bone showed fewer typical epithelial cells and weaker expression of the structural markers that normally hold this barrier together. At the same time, many remaining lining cells produced high levels of proteins associated with cell division and repair, as well as markers of stress, inflammation, and programmed cell death. This mix points to a frantic but disorganized attempt to regrow the surface. In the supporting tissue beneath, fibroblast cells became more numerous and activated, and the usually collagen‑rich matrix was replaced by densely packed cells. Blood and lymph vessels appeared enlarged and their lining cells showed signs of proliferation, inflammation, and oxygen shortage. Together, these changes paint a picture of a microenvironment caught between growth and injury: many cells are dividing, yet just as many are stressed or dying. 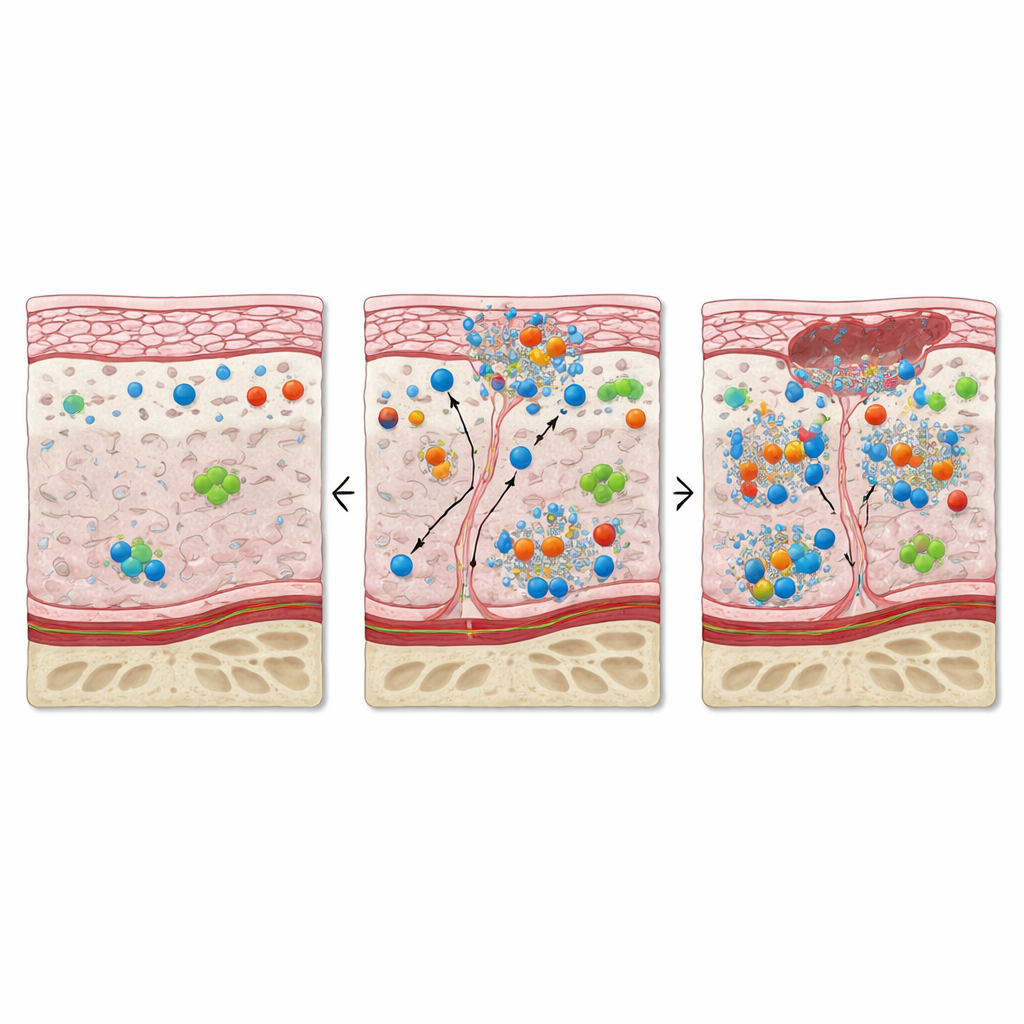
A tangled web of signals and spaces
By combining their spatial maps with more traditional staining methods, the team confirmed that key immune populations and signaling molecules were enriched and repositioned in diseased tissue. Proteins such as PD‑1, which normally help keep immune responses in check, were strongly expressed not only on immune cells but also on epithelial cells, hinting at widespread “brakes” on inflammation that may be over‑applied. Markers linked to survival pathways and migration were elevated across many cell types, consistent with cells struggling to adapt to a harsh, chronically inflamed setting. Yet when these data were compressed into global patterns, healthy and diseased samples still overlapped substantially, underscoring that the crucial differences lie as much in spatial arrangement and functional imbalance as in which cell types are present.
What this means for patients
Rather than a simple failure of bone or a single “bad” cell type, this work shows jaw osteonecrosis as a systems problem: immune cells, blood vessels, connective tissue, and surface lining are all reshaped and locked into a stressed, poorly coordinated state. The tissues around the lesion are packed with cells attempting to repair damage, but they are at the same time exhausted, crowded, and mispositioned. By providing a detailed atlas of who is where and what they are doing, this study offers a roadmap for future therapies—ones that might, for example, loosen suppressive immune clusters, calm damaging inflammation, and restore orderly regeneration so that the jaw can heal.
Citation: Cai, J., Xue, Y., Tornaas, S. et al. Imaging mass cytometry unveils functional and spatial remodeling of peri-lesional cells in jaw osteonecrosis. Commun Biol 9, 442 (2026). https://doi.org/10.1038/s42003-026-09696-7
Keywords: jaw osteonecrosis, bone‑protective drugs, oral wound healing, immune microenvironment, spatial tissue mapping