Clear Sky Science · en
Dissecting the differentiation origins of intestinal metaplasia and early intestinal-type gastric cancer in gastric antrum by single-cell RNA profiling
Why changes in stomach lining cells matter
Stomach cancer is one of the world’s deadliest cancers, in part because it is often discovered late. Yet before a tumor appears, the cells lining the lower part of the stomach (the antrum) pass through a series of subtle changes that can unfold over years. This study asks a simple but crucial question: which exact cells along this path first start to behave like cancer, and how do ordinary stomach cells get reprogrammed into intestinal‑like and then cancerous cells? By tracking individual cells one by one, the researchers map this hidden journey and point to new ways to spot and possibly stop cancer earlier. 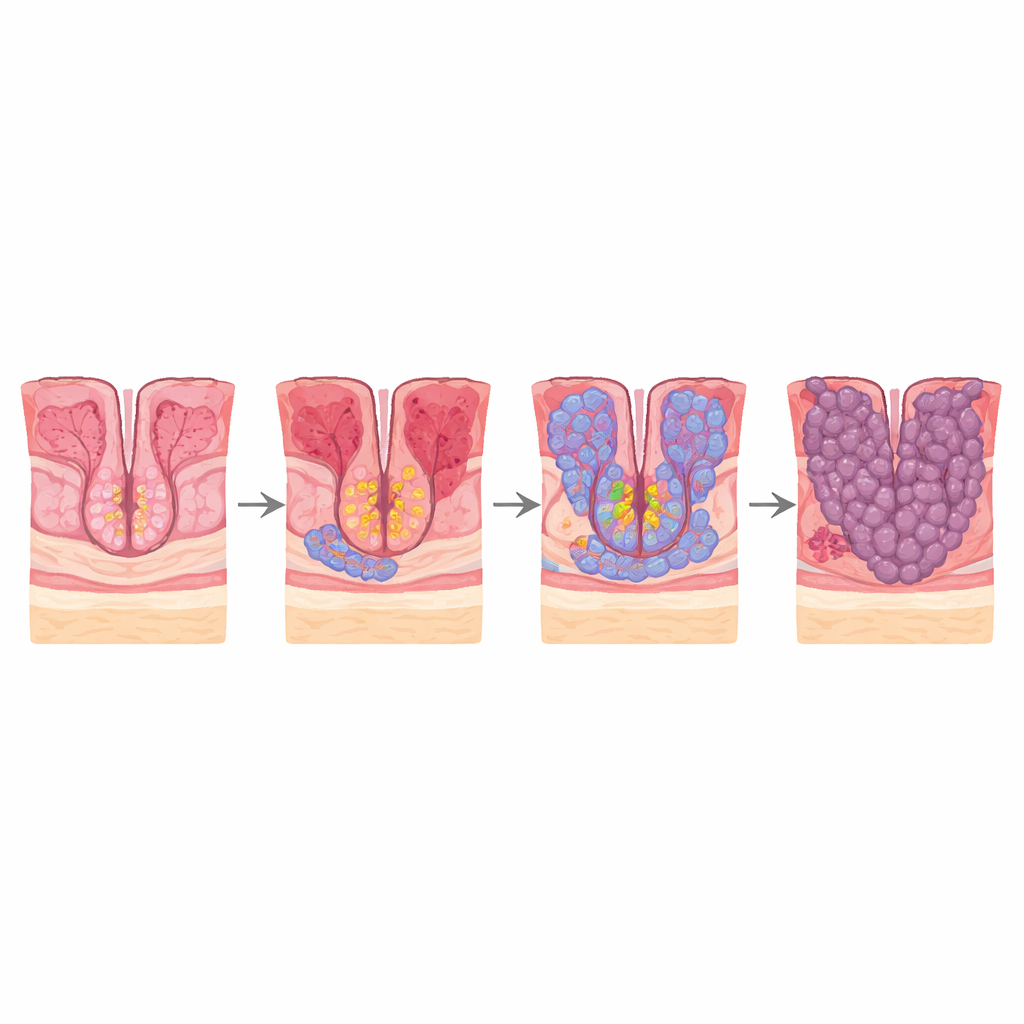
Following the journey from irritation to early cancer
Most intestinal‑type gastric cancers arise through a well‑described sequence: chronic gastritis, loss of normal glands (atrophy), replacement of stomach cells by intestine‑like cells (intestinal metaplasia), and finally early cancer. The authors re‑analyzed single‑cell RNA sequencing data from biopsies that spanned this entire path—non‑atrophic gastritis, atrophic gastritis, intestinal metaplasia, and early gastric cancer. This technology reads out which genes are active in thousands of individual cells, allowing each cell to be classified and its likely “family tree” inferred. They combined these data with spatial transcriptomics, which adds back where each cell sits in the tissue, and with staining of patient samples to confirm key patterns.
The key players in a healthy yet inflamed stomach
In the early, non‑atrophic gastritis stage, the antral lining is dominated by three main epithelial cell types: pit mucous cells that line the surface, gland mucous cells deeper in the glands, and a smaller pool of progenitor cells sitting between them. By analyzing gene activity and developmental “scores,” the team shows that these progenitor cells sit at the root of the system, giving rise to both pit and gland mucous cells through an intermediate, less committed state. Specific control proteins, especially one called TAGLN2, are switched on as progenitor cells become pit mucous cells, helping drive this normal renewal process during inflammation.
When repair turns into intestinal‑like tissue
As disease progresses to chronic atrophic gastritis and then intestinal metaplasia, the balance of lineages shifts. Pit mucous cells decline, while gland mucous cells and new intestine‑like cell types expand. In intestinal metaplasia, the researchers find that both progenitor cells and gland mucous cells can acquire features of intestinal‑type stem cells and then mature into absorptive enterocytes, the hallmark cells of the intestine. They trace two parallel paths that converge on an intermediate intestinal‑like stem cell state before ending in fully developed enterocytes. Another secretory progenitor branch gives rise to goblet cells, the mucus‑secreting cells normally found in the intestine. A set of metabolic control proteins, especially HNF4A and ESRRA, become progressively more active along these paths and appear to steer the gastric‑to‑intestinal transformation. Spatial maps of human tissue show a smooth gradient of markers along glands, confirming that these transitions occur in a continuous, layered fashion. 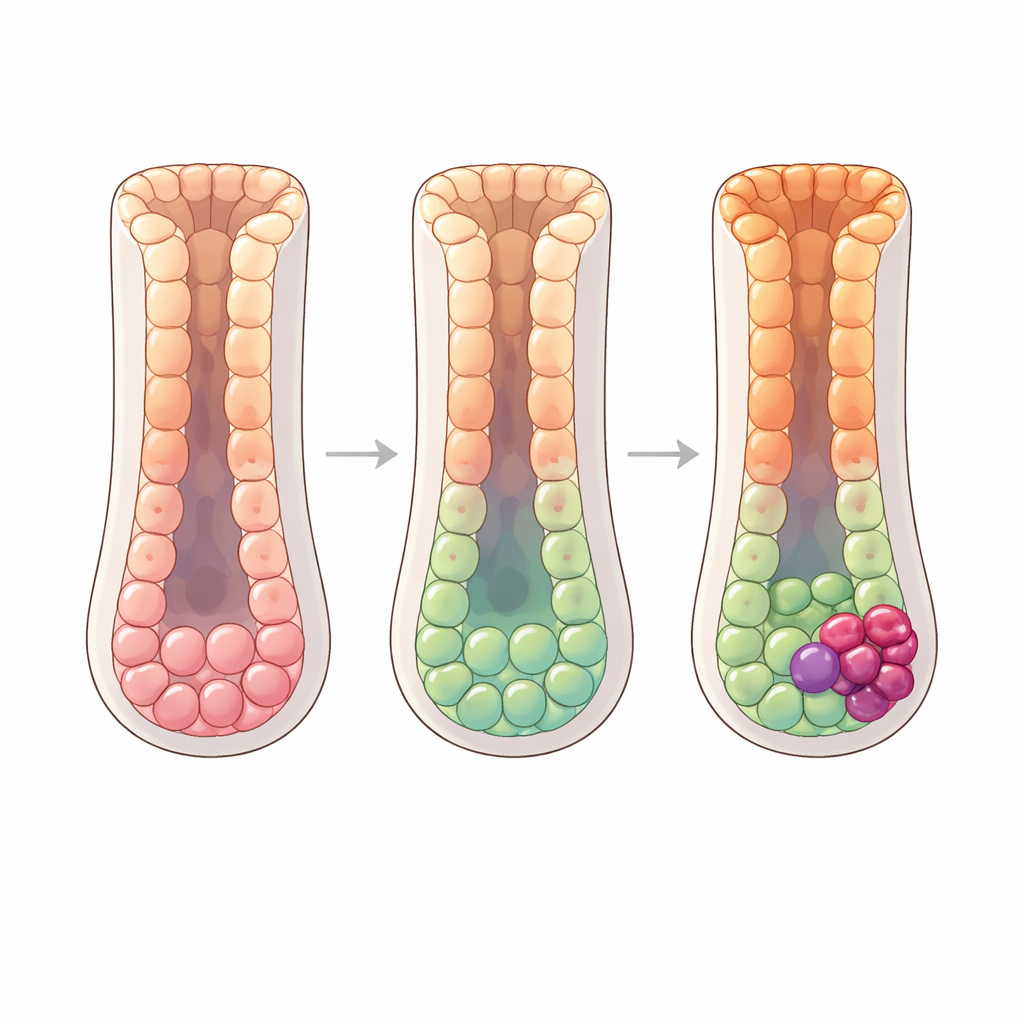
Pinpointing the likely starting cells of early tumors
In early gastric cancer samples, the team asks which non‑cancerous cells most closely resemble the emerging cancer cells in both gene expression and chromosomal alterations. Progenitor cells show the highest similarity, including matching patterns of copy‑number changes, suggesting that at least some cancers arise directly from this pool. Within progenitor cells, they identify a subset marked by two genes, KIAA0101 and PRAP1, that are strongly enriched in cancer tissue and barely detectable elsewhere. These double‑positive cells cluster within malignant regions in spatial maps and are highlighted by fluorescent staining, supporting the idea that they are immediate precursors of early tumors. Additional regulatory proteins, such as SIRT6 and XRCC4, become activated as these progenitors transition into cancer cells and may help support DNA damage tolerance, inflammation, and survival during this dangerous change.
What this means for prevention and early detection
To a lay observer, this work turns a fuzzy picture—“stomach irritation can lead to cancer”—into a detailed roadmap of who does what, when, and where inside the glands of the gastric antrum. It shows that a small pool of progenitor and gland mucous cells acts as a central engine that can be redirected from normal repair toward building intestinal‑like tissue and, eventually, early cancer. By flagging specific molecular markers of high‑risk progenitors (such as KIAA0101 and PRAP1) and key control switches (HNF4A, ESRRA, SIRT6), the study suggests new tests that could identify dangerous changes in routine biopsies and new targets for treatments designed to interrupt the process before a visible tumor forms.
Citation: Yin, H., Zhang, H., Zheng, S. et al. Dissecting the differentiation origins of intestinal metaplasia and early intestinal-type gastric cancer in gastric antrum by single-cell RNA profiling. npj Precis. Onc. 10, 167 (2026). https://doi.org/10.1038/s41698-026-01355-8
Keywords: gastric cancer, intestinal metaplasia, single-cell RNA sequencing, cell lineage, precancerous lesions