Clear Sky Science · en
Biocompatibility, inflammatory response, and antimicrobial properties of single-bottle adhesives on gingival fibroblast and human dental pulp stem cells
Why Your Filling’s Glue Matters
When you get a tooth filled, the success of that treatment depends not only on the filling itself, but also on the “glue” that bonds it to your tooth. These modern bonding liquids, called universal or single-bottle adhesives, promise strong, long-lasting repairs and even some protection against cavity-causing germs. But because they sit right next to the tooth’s living tissue and the gums, they must be gentle as well as tough. This study asks a simple but important question: how kind are these glues to our cells, and how well do they keep harmful bacteria at bay?
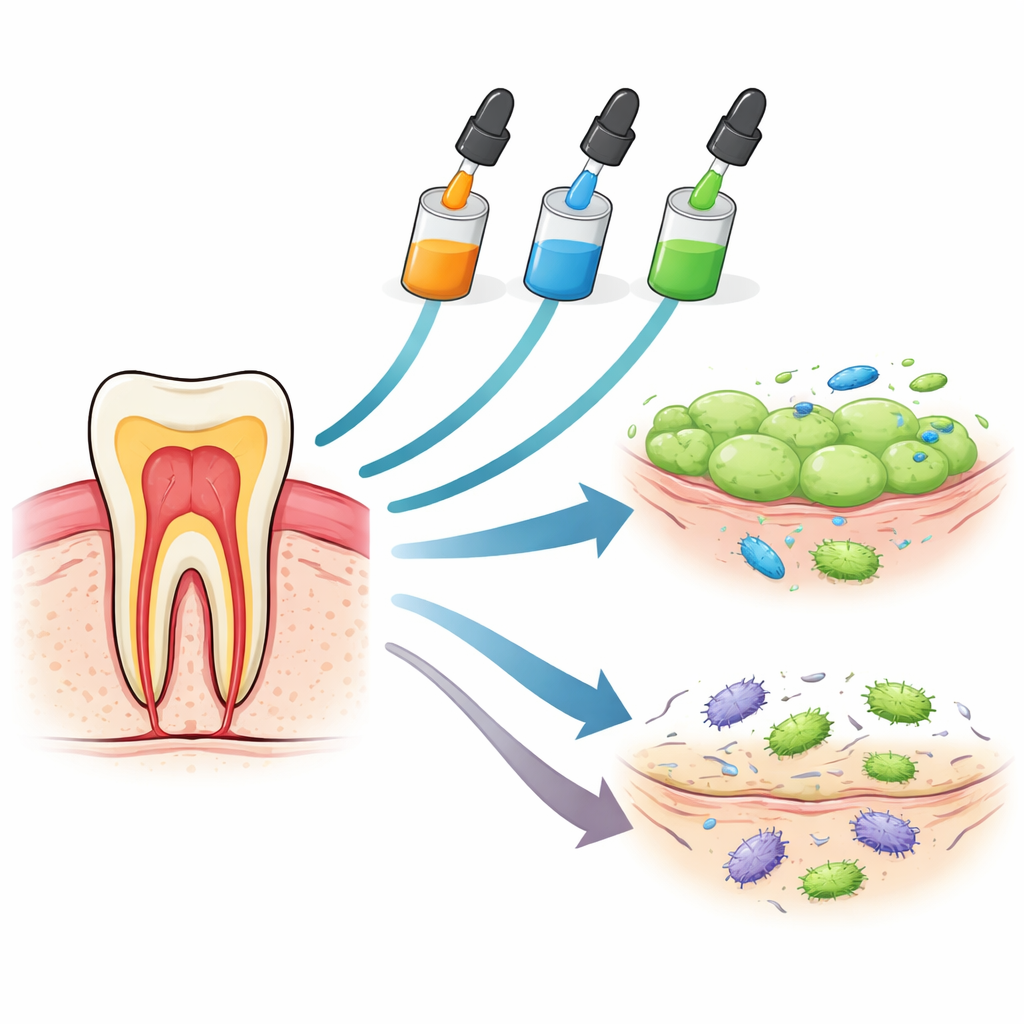
Three Glues Under the Microscope
The researchers compared three widely used one-bottle dental adhesives: Huge Bond, Single Bond Universal, and G-Premio Bond. All are designed to simplify dental work by combining several steps into one liquid that can bond to enamel, dentin, and even some ceramics and metals. In the lab, the team brought these materials into contact with two types of human cells that are directly exposed during real dental treatments: gum connective tissue cells (gingival fibroblasts) and stem cells from the soft tissue inside the tooth (dental pulp stem cells). They then tracked how many cells survived after very short contact (one minute), after an hour, and after six hours, and also measured chemical signals of irritation and tested how well the adhesives could stop common cavity bacteria from growing.
How the Cells Reacted Over Time
Right after contact, all three adhesives appeared fairly safe. At the one-minute mark, cell survival stayed close to normal for both gum and pulp cells, suggesting that a brief encounter alone is not strongly damaging. But differences emerged as time went on. After one hour, Huge Bond already showed a noticeable drop in living cells, especially among the pulp cells, while Single Bond and G-Premio caused milder changes. By six hours, the picture was clearer: both Huge Bond and Single Bond led to marked cell loss in gum and pulp cells, and G-Premio, while still reducing cell numbers, was less harmful overall. These patterns match what is known about the building blocks inside many adhesives—resin components that, if not fully hardened by light curing, can leach out and stress or kill sensitive cells.
Signals of Irritation and Battle Against Germs
Cell survival was only part of the story. The team also measured a key alarm signal that cells release when they are irritated or inflamed, a molecule often linked to early tissue damage. After six hours in contact with the adhesives, both gum and pulp cells produced much higher levels of this alarm, especially when exposed to Huge Bond, followed by Single Bond, with G-Premio again the least provocative. At the same time, the materials showed useful antibacterial behavior. When placed in wells on agar plates seeded with Lactobacilli and Streptococcus mutans—two major cavity-forming bacteria—all three created clear zones where bacteria could not grow. Huge Bond produced the largest bacteria-free zones for both species, G-Premio a bit smaller, and Single Bond the least, indicating that the same acidic and reactive components that trouble human cells can also temporarily suppress harmful oral microbes.
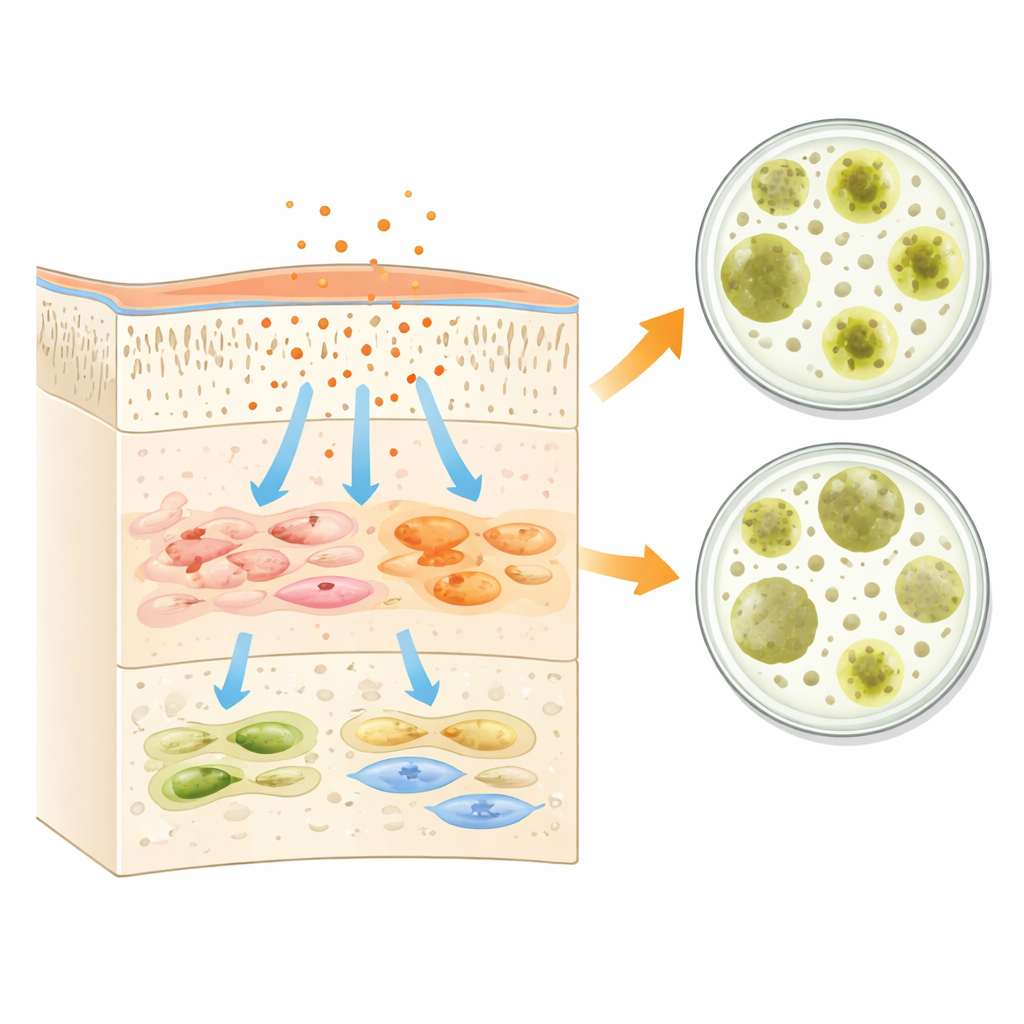
What the Ingredients Tell Us
The study connects these biological effects to what is inside each bottle. The adhesives contain different mixes of resin molecules, acidic bonding agents, solvents, and tiny fillers. Some common resin ingredients are known to be especially troublesome when they remain unreacted, as they can slip through the tooth’s hard tissues and reach the living pulp or leak onto nearby gums. The authors point out that methacrylate-type resins, widely used to create strong, durable bonds, are closely tied to cell stress, inflammation, and even programmed cell death if curing is incomplete. At the same time, the low pH of these liquids and their functional bonding components can temporarily lower bacterial activity, explaining the initial antibacterial zones seen in the lab. G-Premio’s formulation, which avoids one particularly mobile resin and relies on a tailored mix of bonding molecules, may explain why it showed the gentlest behavior toward pulp cells while still offering some antibacterial effect.
What This Means for Your Dental Visit
For patients, the takeaway is not to fear dental adhesives, but to appreciate that their safety depends on both the recipe in the bottle and how carefully they are used. Under the study’s conditions, all three products looked reasonably safe when contact with living cells was brief, as in a well-controlled clinical procedure. Over longer exposure, however, the more resin-heavy formulations caused more cell damage and stronger inflammation signals, even as they offered better short-term suppression of cavity-causing bacteria. The authors conclude that the safest path is to choose materials thoughtfully and ensure that the adhesive is thoroughly light-cured so that as few reactive leftovers as possible can escape into the tooth and gums. In short, the “invisible” layer that holds your filling in place is a delicate balance between strength, protection, and kindness to the tissues that keep your tooth alive.
Citation: Ibrahim, A.H., Mustafa, S.S., El-Khazragy, N. et al. Biocompatibility, inflammatory response, and antimicrobial properties of single-bottle adhesives on gingival fibroblast and human dental pulp stem cells. Sci Rep 16, 13886 (2026). https://doi.org/10.1038/s41598-026-49388-0
Keywords: dental adhesives, biocompatibility, dental pulp stem cells, gingival fibroblasts, antimicrobial activity