Clear Sky Science · en
Comparative study of armchair and zigzag graphene quantum dots as HIV-1 protease inhibitors
A Tiny Carbon Tool in the Fight Against HIV
Medicines that block HIV from making copies of itself have turned a deadly infection into a manageable condition, but the virus can still evolve resistance to existing drugs. This study explores a new kind of candidate drug built from graphene quantum dots—ultra-small flakes of carbon only a few billionths of a meter across—to see whether they could latch onto a key viral enzyme, HIV-1 protease, and potentially shut it down.
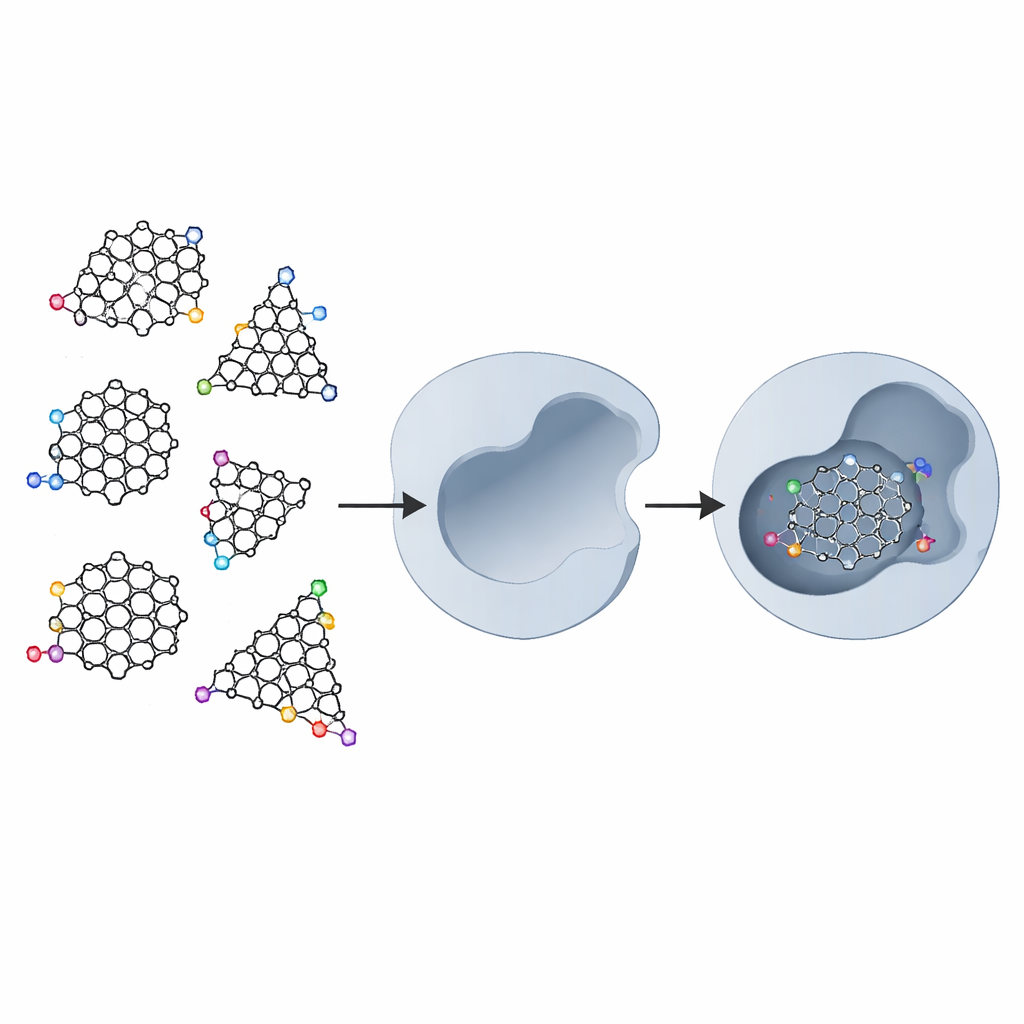
What Makes These Carbon Dots Special
Graphene is a sheet of carbon atoms arranged like chicken wire. When that sheet is cut into very small pieces, called graphene quantum dots, it gains new behaviors because electrons are confined in such a tiny space and because the edges of the pieces become chemically active. The dots can be cut into triangular or hexagonal shapes, and their borders can have different patterns—"armchair" or "zigzag" arrangements of atoms. On top of that, chemists can attach extra chemical groups to the edges to improve how the dots dissolve in water and how they interact with biological molecules. These combined features make graphene quantum dots promising tools not just for electronics and imaging, but also for medicine.
Designing Carbon-Based Protease Blockers
The researchers examined five basic types of graphene quantum dots: a plain sheet, triangular and hexagonal pieces with armchair edges, and triangular and hexagonal pieces with zigzag edges. They then "decorated" each of these with a small ring (pyrrolidine), a benzene ring, and finally two hydroxymethylcarbonyl (HMC) groups. The HMC groups were chosen because they can form hydrogen bonds with two aspartic acid units that sit at the heart of the HIV-1 protease active site. In the real enzyme, almost all nearby amino acids are water-hating (hydrophobic), but these two aspartic acids are water-loving (hydrophilic), creating a natural docking spot for carefully placed chemical hooks.
Testing Reactivity on the Computer
Instead of working in a wet lab, the team used high-level quantum chemistry calculations to predict how these designer dots behave. They optimized each structure and calculated quantities that signal chemical reactivity, such as the total dipole moment (a measure of how unevenly charge is spread out) and the energy gap between the highest occupied and lowest unoccupied electron states. A large dipole moment combined with a small gap usually means a molecule is more eager to interact. Among all designs, a zigzag-edged triangular dot with attached pyrrolidine and HMC groups stood out, displaying the highest polarity and the smallest gap. The scientists also examined how the electron cloud spreads across each dot and how many electronic states are available, further confirming that certain shapes and edge patterns make the material more responsive.
Seeing How Well the Dots Grip the Enzyme
To understand whether these quantum dots could really clasp onto HIV-1 protease, the researchers simulated their interaction with two aspartic acid units that mimic the enzyme’s active site. Using a method called quantum theory of atoms in molecules, they inspected the fine details of the electron density where bonds might form. All of the modified dots formed stable complexes, but the zigzag triangular design with HMC groups showed especially strong interactions, with some contacts taking on a partially covalent character—closer to a true chemical bond than a fleeting attraction. Another important factor was size: the best-fitting structure, based on a pristine sheet modified with pyrrolidine, benzene, and two HMC groups, measured about 9.3 angstroms across, nicely matching the roughly 10-angstrom-wide cavity of the real protease active site.
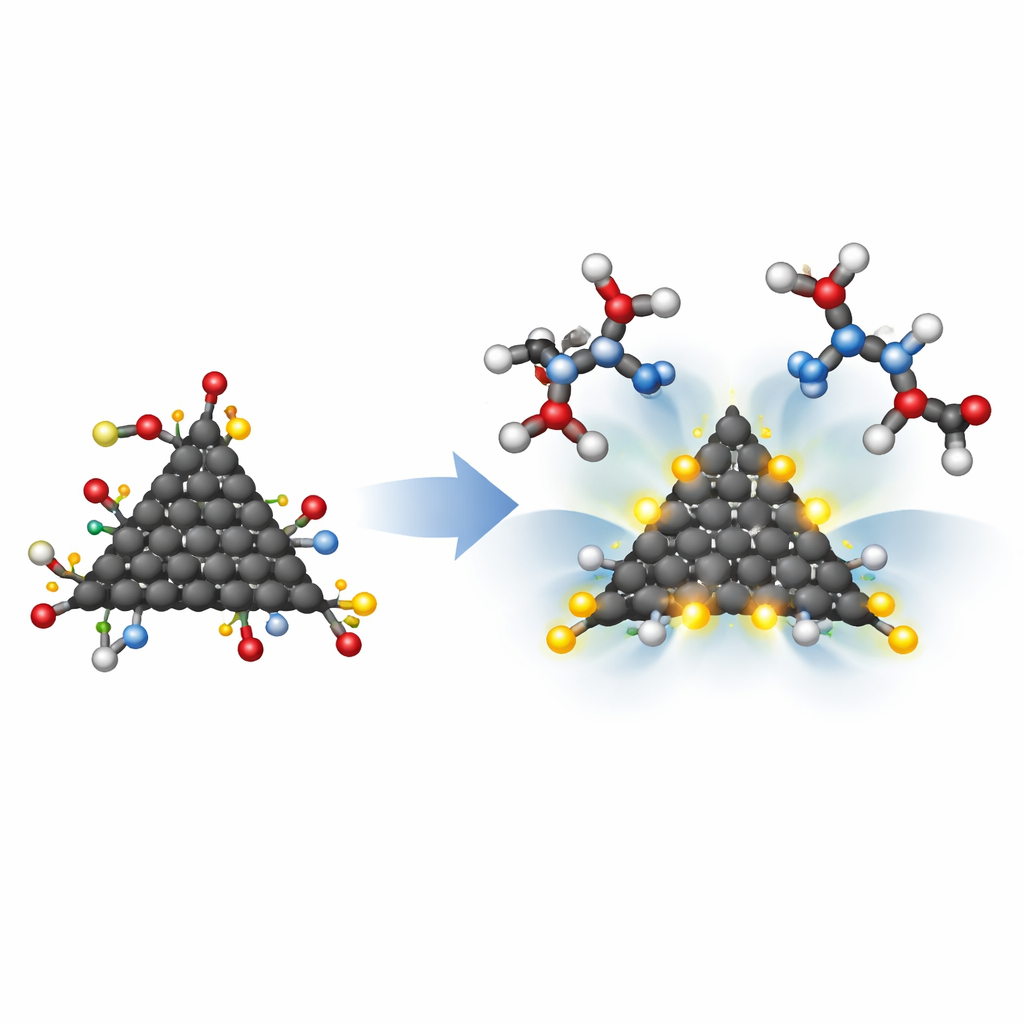
Why This Matters for Future HIV Treatments
By combining clever nanoengineering with detailed computer modeling, this work shows that tiny carbon dots can be tailored to both fit into the HIV-1 protease pocket and grip its key aspartic acid residues firmly. The most promising versions are small enough to enter the enzyme cavity, polar and reactive enough to form strong hydrogen bonds, and electronically stable once bound. While these are early, theoretical results rather than ready-made drugs, they map out how the shape, edge pattern, and attached chemical groups of graphene quantum dots jointly control their ability to act as HIV-1 protease blockers. That roadmap could guide the design of a new class of carbon-based antiviral materials in the future.
Citation: Ibrahim, A., Elhaes, H. & Ibrahim, M.A. Comparative study of armchair and zigzag graphene quantum dots as HIV-1 protease inhibitors. Sci Rep 16, 14650 (2026). https://doi.org/10.1038/s41598-026-48709-7
Keywords: graphene quantum dots, HIV-1 protease, nanomedicine, computational drug design, carbon nanomaterials