Clear Sky Science · en
Purification, characterization, and dehairing properties of alkaline and thermo-stable keratinase by Penicillium citrinum AUMC 14742
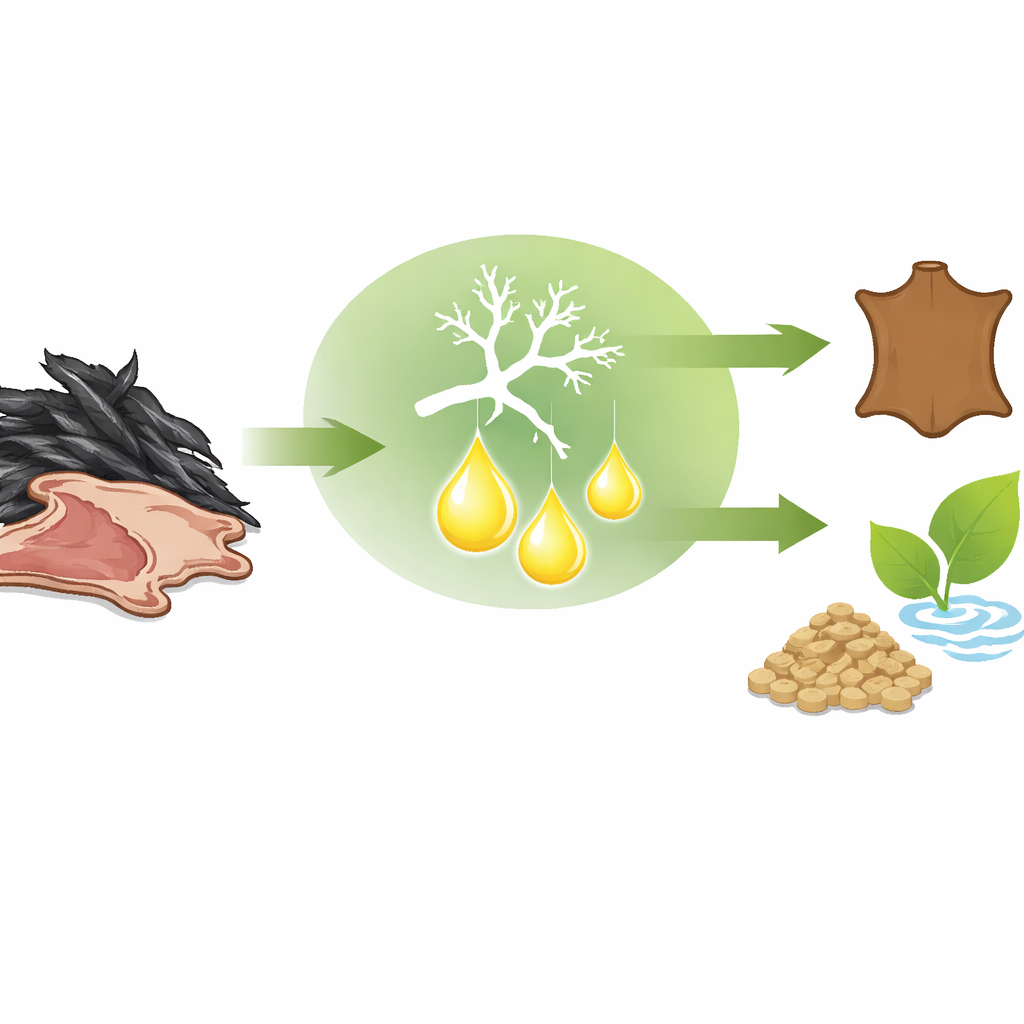
Turning Waste Feathers into Useful Goods
Modern poultry farms generate mountains of feathers and other tough, hair-like scraps that are hard to break down and often end up burned or dumped. This study shows how a naturally occurring fungus can make a powerful enzyme that turns this stubborn waste into something valuable, while also offering a cleaner way to remove hair from animal hides in the leather industry.
Why Feathers Are So Hard to Handle
Feathers, wool, and animal hair are built from keratin, a protein arranged like tightly knotted ropes reinforced with many chemical "bridges." This design makes feathers light, strong, and almost indestructible, but it also means they resist ordinary decay and most common protein-cutting enzymes. As a result, keratin-rich waste accumulates around farms and tanneries, creating pollution and wasting a potentially rich source of nutrients and raw materials that could be used for animal feed, fertilizers, or specialty products.
A Helpful Fungus Joins the Team
The researchers focused on a strain of the fungus Penicillium citrinum, originally collected and mapped in detail to confirm its identity. This strain produces keratinase, an enzyme tailor-made to attack the tough structure of keratin. Using a statistical optimization approach called a Box–Behnken design, the team systematically adjusted growth temperature, acidity (pH), and the amount of added peptone, a nitrogen-rich nutrient. They discovered that a relatively cool temperature of 26 °C, mildly alkaline conditions (pH 8), and a low peptone concentration gave the highest enzyme output. Under these conditions, the fungus produced an impressively active keratinase, which was then carefully purified and shown to be a single, well-defined protein.
An Enzyme Built for Harsh Jobs
Once purified, the keratinase revealed a set of traits that make it especially promising for industrial use. It works best at a strongly alkaline pH of 10 and at a moderately high temperature of 55 °C, showing that it can stay active where many other proteins would fall apart. Tests with various salts, detergents, and solvents showed that the enzyme is remarkably tolerant of conditions similar to those found in cleaning products and processing baths. Certain metal ions and additives, and especially a reducing chemical that helps open up keratin’s internal bridges, boosted its activity dramatically. The enzyme also showed high catalytic efficiency, meaning it can process keratin quickly once it encounters it.
From Lab Bench to Leather Shop
To see whether these favorable properties translate into real-world performance, the team tested the crude enzyme mixture on goat skins. Instead of using harsh, smelly chemicals normally employed to remove hair, they simply soaked skins in the keratinase solution at 30 °C. Within 15 hours, the hair lifted off completely, leaving smooth, flexible skin without visible damage. This gentle yet effective dehairing demonstrates that the enzyme can do the same job as traditional chemicals, but under milder conditions and with far fewer environmental downsides.
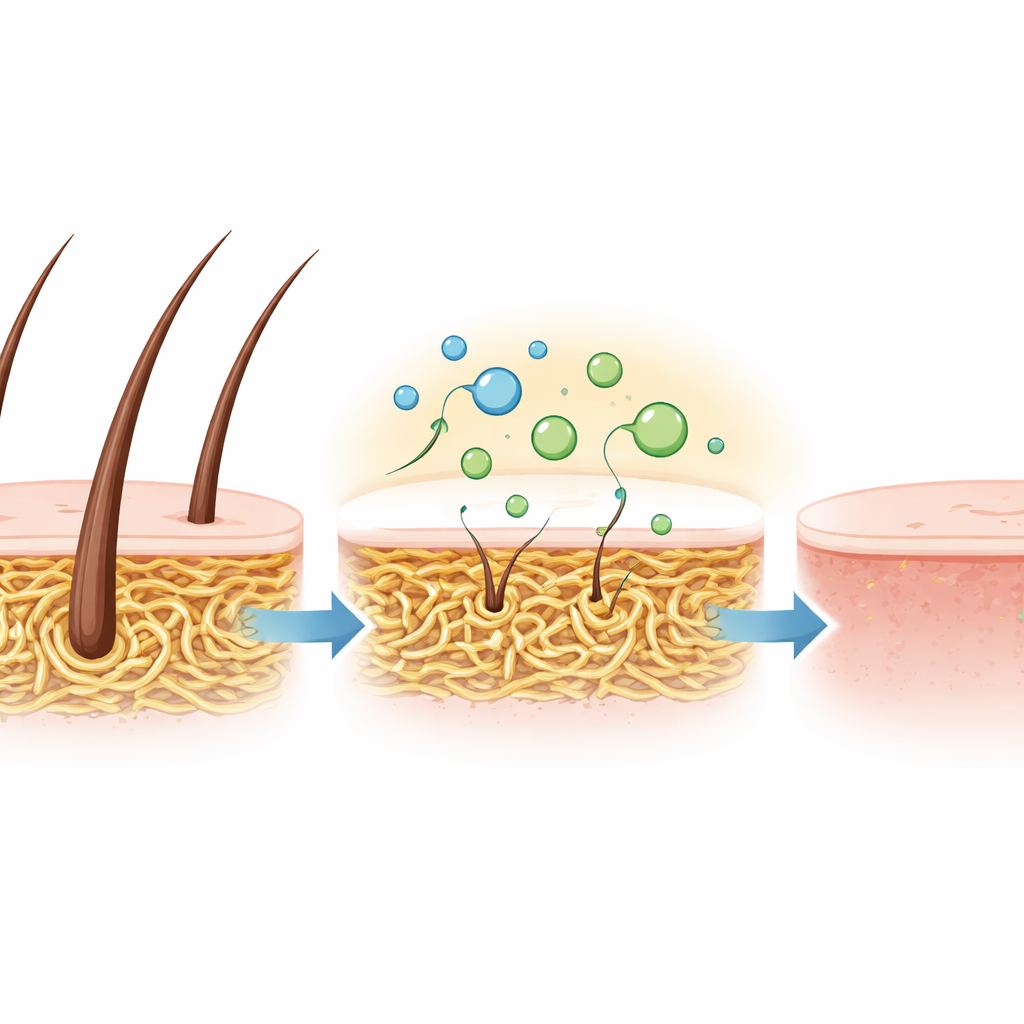
A Greener Path for Tough Protein Waste
In simple terms, this work uncovers a fungus-made enzyme that is unusually strong in alkaline and warm environments, stands up to detergents and solvents, and can tear apart keratin efficiently. That combination makes it a powerful tool for turning stubborn feather and hair waste into useful products and for replacing toxic chemicals in leather processing. For readers, the takeaway is that what we currently treat as a troublesome waste stream could instead become a valuable resource, thanks to a microscopic partner and its finely tuned molecular scissors.
Citation: Al-Bedak, O.A.M., Abdel-Latif, A.M.A., Abo-Dahab, N.F. et al. Purification, characterization, and dehairing properties of alkaline and thermo-stable keratinase by Penicillium citrinum AUMC 14742. Sci Rep 16, 13025 (2026). https://doi.org/10.1038/s41598-026-48471-w
Keywords: keratinase, poultry feather waste, fungal enzymes, eco-friendly leather processing, bioconversion