Clear Sky Science · en
A transverse picoNewton force revealed in anisotropic Womersley flow
Why tiny sideways pushes in blood flow matter
Our arteries are lined with a thin layer of cells that constantly feel the push and pull of flowing blood. For decades, researchers have focused on the drag of blood rubbing along the vessel wall, called shear stress, as the main mechanical signal that these cells sense. Yet this measure has struggled to reliably predict where dangerous conditions like atherosclerosis or aneurysms will appear. This study proposes a different way to think about how blood pokes at the vessel wall: not just as a smooth rubbing along the wall, but as tiny sideways nudges—on the order of a trillionth of a newton—that arise from the way blood’s internal motion twists and turns near the wall.
Looking beyond a simple rubbing force
Traditionally, scientists have tried to link the health of blood vessels to wall shear stress, a single number describing how strongly blood drags along the surface. But real blood flow is far from simple. Pulsing with each heartbeat, it forms swirling structures and complex patterns that change in time and space. Experiments and computer simulations have shown that two flows with the same shear stress at the wall can still produce very different responses in the cells, suggesting that important information is being lost when everything is compressed into a single scalar measure.
Blood as a direction-sensitive fluid
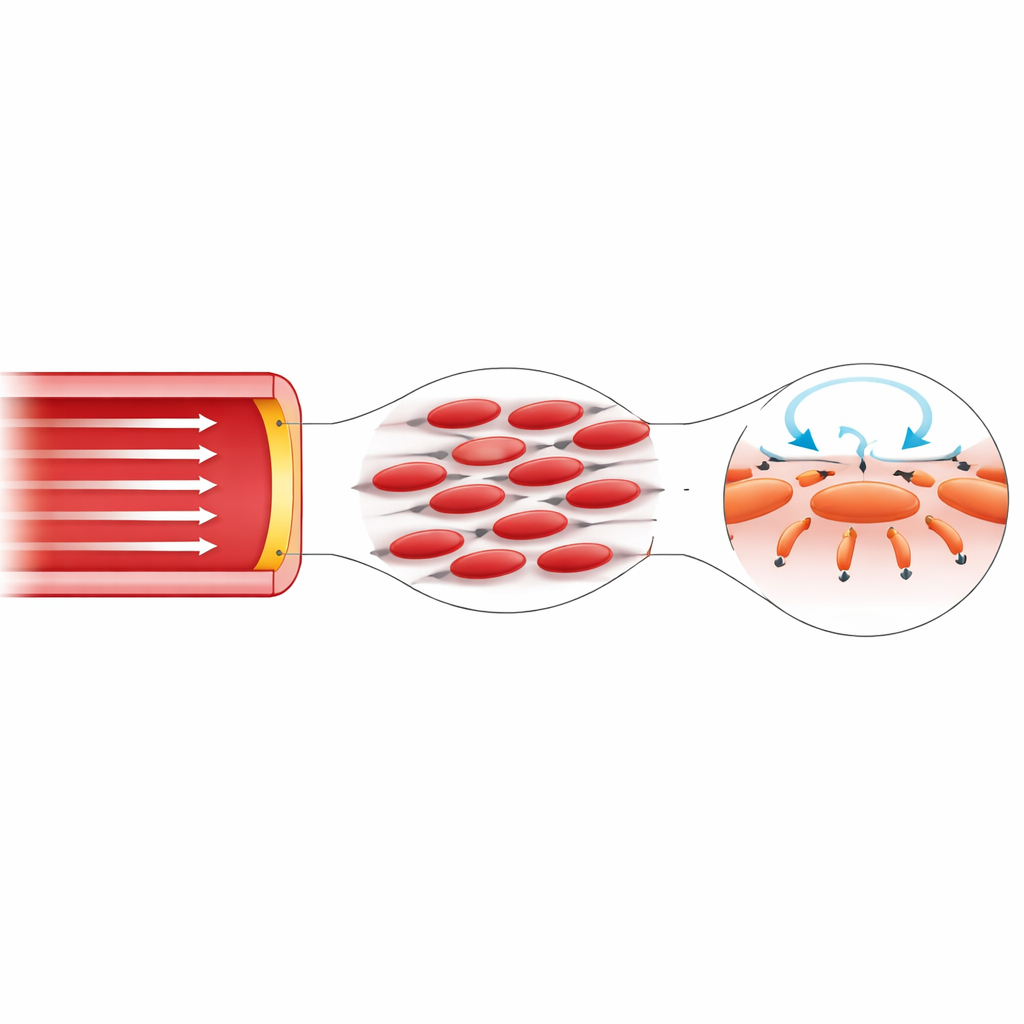
Part of the puzzle lies in the nature of blood itself. Blood is not a uniform liquid; nearly half of its volume is made up of red blood cells that deform, align, and migrate under flow. Near the vessel wall, these cells tend to move away, leaving a thin layer that has different properties from the core. As a result, the internal stresses in blood depend on direction—they cannot be fully described by a single viscosity value. In this work, the author builds this direction-sensitivity directly into the governing equations by replacing the usual scalar viscosity with a small matrix that can couple motion along the vessel to motion around it. This mathematically simple change unlocks behaviours that are impossible in the classical model.
Uncovering hidden sideways forces
Using an idealized straight, rigid tube as a clean testbed, the study revisits a classic description of pulsatile flow known as Womersley flow. In the standard picture, fluid moves only along the tube, with no swirl and no sideways inertial push other than a gentle centripetal effect. When the new direction-dependent viscosity is introduced, the solution changes qualitatively: the flow develops a slight azimuthal swirl, the vorticity gains new components, and their interaction creates a radial inertial term—the Lamb vector—that points toward or away from the wall. By solving these equations numerically with high accuracy, the author shows that this transverse inertial force is sharply concentrated within a thin near-wall layer and, when integrated over the footprint of a single endothelial cell, reaches picoNewton levels comparable to the forces known to open mechanosensitive ion channels.
How these forces vary across the arterial tree
The analysis is then extended from a single ideal heartbeat to realistic pressure waveforms measured in six different human arteries, ranging from the large aortic root to smaller peripheral vessels. A key control parameter is the Womersley number, which compares unsteady inertia to viscosity and grows with vessel size. In large, inertia-dominated arteries, the induced swirl is confined to a very thin boundary layer near the wall, and the resulting sideways force changes sign and direction several times over a single heartbeat, producing a rich, high-frequency pattern. In smaller arteries, the force profile is smoother, penetrates deeper toward the vessel core, and tends to maintain a more consistent inward direction. Statistical analysis confirms that the pattern and strength of these picoNewton forces differ systematically from one artery type to another.
Competing with curvature and shaping fast signals
Real arteries are not straight, and vessel curvature also generates transverse forces through centrifugal effects and secondary swirling motions. The study compares this geometry-driven loading with the new anisotropy-driven mechanism in the frequency domain. At the fundamental heart rate, curvature-induced forces are typically stronger—often by about an order of magnitude. However, as one looks at higher harmonics of the pulse, the large-scale geometric contribution is strongly filtered out by inertia, while the near-wall anisotropic forces retain substantial power. This means that at higher frequencies, the sideways forcing felt by endothelial cells is dominated not by the shape of the vessel, but by the direction-dependent nature of blood itself.
What this means for vessel health
By showing that realistic levels of blood anisotropy naturally produce picoNewton-scale sideways forces concentrated at the vessel wall, this work introduces a new, geometry-independent baseline for how pulsatile blood flow can mechanically stimulate endothelial cells. Instead of relying solely on wall shear stress, it points to a volumetric quantity—the Lamb vector, capturing how velocity and vorticity interact—as a more complete descriptor of the mechanical environment. The study suggests that high-frequency, multidirectional forcing arising from blood’s internal structure may help explain why cells in different arteries respond so differently, even under similar average shear levels, and offers a theoretical foundation for future experiments that probe endothelial mechanobiology in the spectral and directional domains.
Citation: Saqr, K.M. A transverse picoNewton force revealed in anisotropic Womersley flow. Sci Rep 16, 12584 (2026). https://doi.org/10.1038/s41598-026-47474-x
Keywords: blood flow mechanics, endothelial mechanotransduction, pulsatile hemodynamics, anisotropic viscosity, arterial wall forces