Clear Sky Science · en
A degrader of HER2 and EGFR abolishes p95HER2 and shows robust antitumor efficacy in HER2-positive breast cancer
Why this research matters
For many people with an aggressive form of breast cancer called HER2‑positive disease, modern drugs have turned a once‑deadly diagnosis into a treatable condition. Yet some tumors still find ways to escape treatment and keep growing. This study focuses on a particularly stubborn culprit—a shortened form of the HER2 protein called p95HER2—and introduces an experimental drug, PEPDG278D, that appears to wipe out this escape route and drive even highly resistant tumors into lasting remission in mice.
A tough-to-treat form of breast cancer
About one in five breast cancers carry too many copies of a gene called HER2, which makes cells grow and divide faster than they should. Several successful drugs already target HER2, including antibodies like trastuzumab and small‑molecule pills called tyrosine kinase inhibitors. However, many tumors eventually become resistant. A major reason is the appearance of p95HER2, a shortened, constantly active fragment that stays anchored in the cell membrane even after the outer portion of HER2 is cut off. This fragment lacks the usual docking sites for antibody drugs, helps tumors invade and spread, and has been linked to poor survival in patients.
Existing drugs and their limits
The authors first compared standard HER2‑targeted drugs—trastuzumab, pertuzumab, tucatinib, and lapatinib—in a range of human breast cancer cell lines grown in the lab. These cell lines overexpress full‑length HER2 and also produce varying amounts of p95HER2. While the approved drugs could slow growth in some cells, their effects were modest and inconsistent, especially in models with additional resistance factors such as mutations in growth‑signal pathways. Importantly, none of these drugs substantially reduced the actual amount of HER2 or p95HER2 protein; at best they dulled some of the activity, and in some cases they even increased levels of the troublesome p95HER2 fragment.
A new strategy: make the receptors disappear
PEPDG278D takes a different approach. Rather than merely blocking the activity of HER2, this engineered human protein binds to the outside portion of both HER2 and a related growth receptor, EGFR, and triggers their removal and breakdown inside the cell. In multiple HER2‑positive breast cancer cell lines, very low doses of PEPDG278D sharply reduced cell growth. Detailed protein analyses showed that it almost completely eliminated HER2, EGFR, and p95HER2, along with the key signaling switches (such as AKT and ERK phosphorylation) that drive proliferation and survival. Notably, PEPDG278D did not change the genes’ activity—it acted by degrading existing proteins—nor did it harm cells that lacked HER2 or EGFR, suggesting a degree of selectivity.
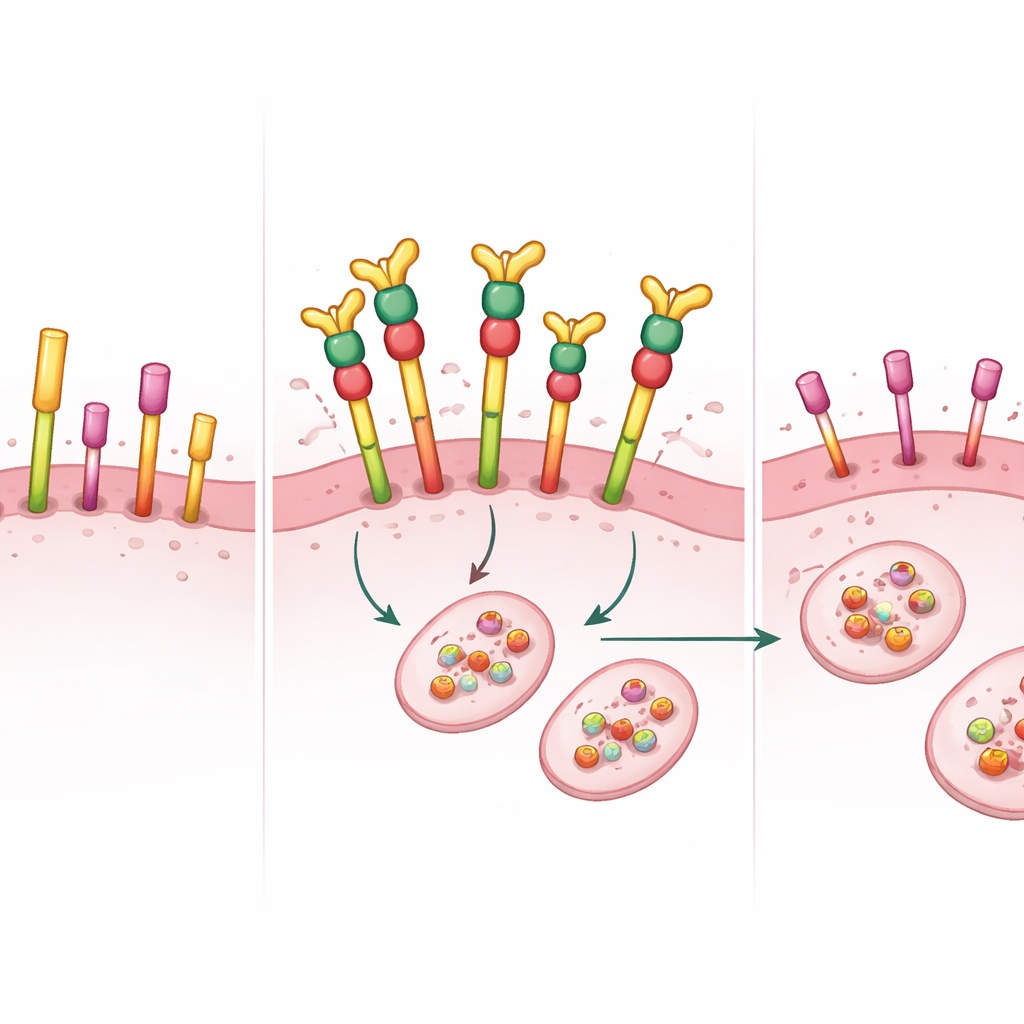
Powerful effects in mouse tumor models
The team then moved to more realistic models: tumors growing in the mammary glands of mice. In a cell‑line xenograft model and in a patient‑derived tumor model that carried amplified HER2 and EGFR and was resistant to trastuzumab and tucatinib, the standard drugs failed to halt tumor growth. In striking contrast, PEPDG278D, given together with a stabilizing antibody that prevents it from being rapidly broken down in the blood, caused rapid and often complete tumor regression. In the patient‑derived model, all tumors disappeared within about two weeks and did not return over 72 days of follow‑up after treatment stopped. Tumor samples showed that HER2, p95HER2, EGFR, and their downstream signaling partners were all essentially switched off at the protein level.
What this could mean for patients
These findings suggest that HER2‑positive breast cancers, even when they produce p95HER2 and resist current medicines, depend heavily on the presence of HER2 and EGFR proteins. By physically removing both receptors, PEPDG278D shuts down multiple growth pathways at once and eliminates the troublesome p95HER2 fragment. While this work was done in cells and mice, and safety and effectiveness in humans remain to be tested, it points to a promising new treatment strategy: instead of just blocking cancer‑driving proteins, make them vanish altogether.
Citation: Yang, L., Bhattacharya, A., Li, Y. et al. A degrader of HER2 and EGFR abolishes p95HER2 and shows robust antitumor efficacy in HER2-positive breast cancer. Sci Rep 16, 12890 (2026). https://doi.org/10.1038/s41598-026-47444-3
Keywords: HER2-positive breast cancer, p95HER2, protein degradation therapy, EGFR, drug resistance