Clear Sky Science · en
An improved ICEEMDAN–depth hybrid network model integrating multimodal data for the screening of diabetic peripheral neuropathy
Why Nerve Damage in Diabetes Needs a Gentler Test
For many people with diabetes, damage to the nerves in the feet and legs creeps in silently, only becoming obvious when pain, numbness, or even ulcers and amputations appear. Today’s tests for this condition, called diabetic peripheral neuropathy, can be invasive, expensive, or hard to repeat in everyday clinics. This study explores a new way to screen for early nerve damage using only harmless light and electrical sensors on the skin, combined with smart signal processing and a tailored deep‑learning model.
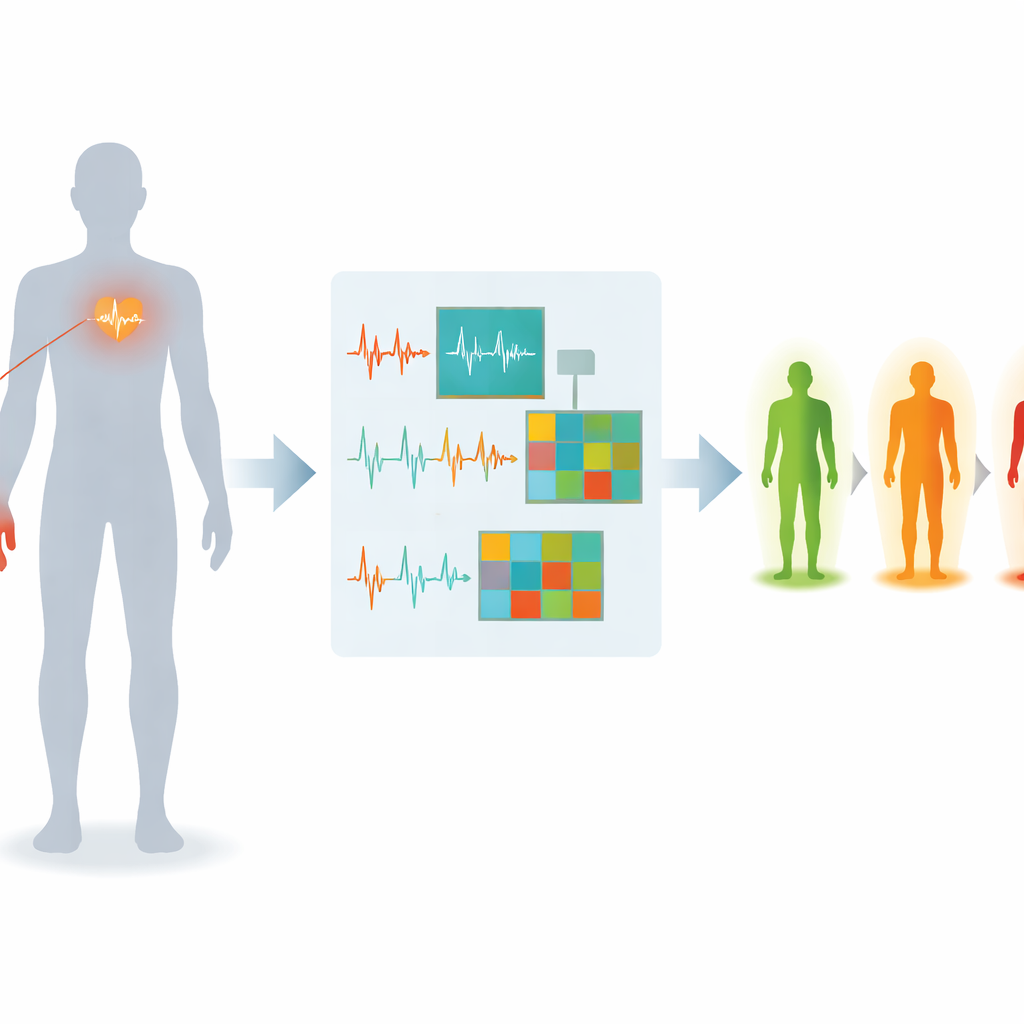
A New Way to Listen to the Heart and Blood Flow
The researchers focus on two signals that are already common in hospitals and wearable devices. One is the electrocardiogram, or ECG, which records the heart’s electrical activity. The other is the photoplethysmogram, or PPG, an optical signal from a fingertip sensor that tracks blood flow with each heartbeat. Each signal tells part of the story of how diabetes affects blood vessels and nerves; together, they can paint a richer picture of early nerve damage than either signal alone. The team recruited 120 people, including healthy volunteers, people with diabetes but no diagnosed nerve damage, and people with confirmed neuropathy. For each participant, ECG and PPG were recorded at rest under controlled conditions to reduce everyday sources of noise such as movement and temperature changes.
Cleaning Up Messy Biological Signals
Real‑world heart and pulse recordings are full of unwanted wiggles from muscle activity, breathing, and electrical interference. If these distortions are not removed carefully, they can hide the subtle changes that point to early nerve problems. The study introduces an improved denoising method that breaks each signal into many simpler “building blocks,” decides which parts are mostly noise and which carry genuine physiology, and then reconstructs a cleaner version. To do this, the authors combine a flexible decomposition technique with a swarm‑based search strategy that automatically tunes key parameters, and then apply wavelet‑based filtering only where it helps most. Tests against several established methods show that this approach better boosts the useful part of both ECG and PPG signals while keeping distortion to a minimum.
Turning Waves into Pictures the Computer Can See
Instead of feeding raw one‑dimensional waves into a neural network, the researchers convert short segments of the ECG and PPG into small color images. Each color channel encodes a different aspect of how the signal evolves over time: overall position relationships between points in the heartbeat cycle, changes in phase and rhythm, and patterns that repeat in complex, non‑linear ways. When these three views are stacked together, they form a compact “portrait” of each multi‑beat window that emphasizes dynamic features linked to nerve and vessel health. By combining several consecutive cycles rather than a single beat, the method captures more stable patterns and long‑range interactions between the heart’s electrical activity and the pulse wave traveling through the vessels.
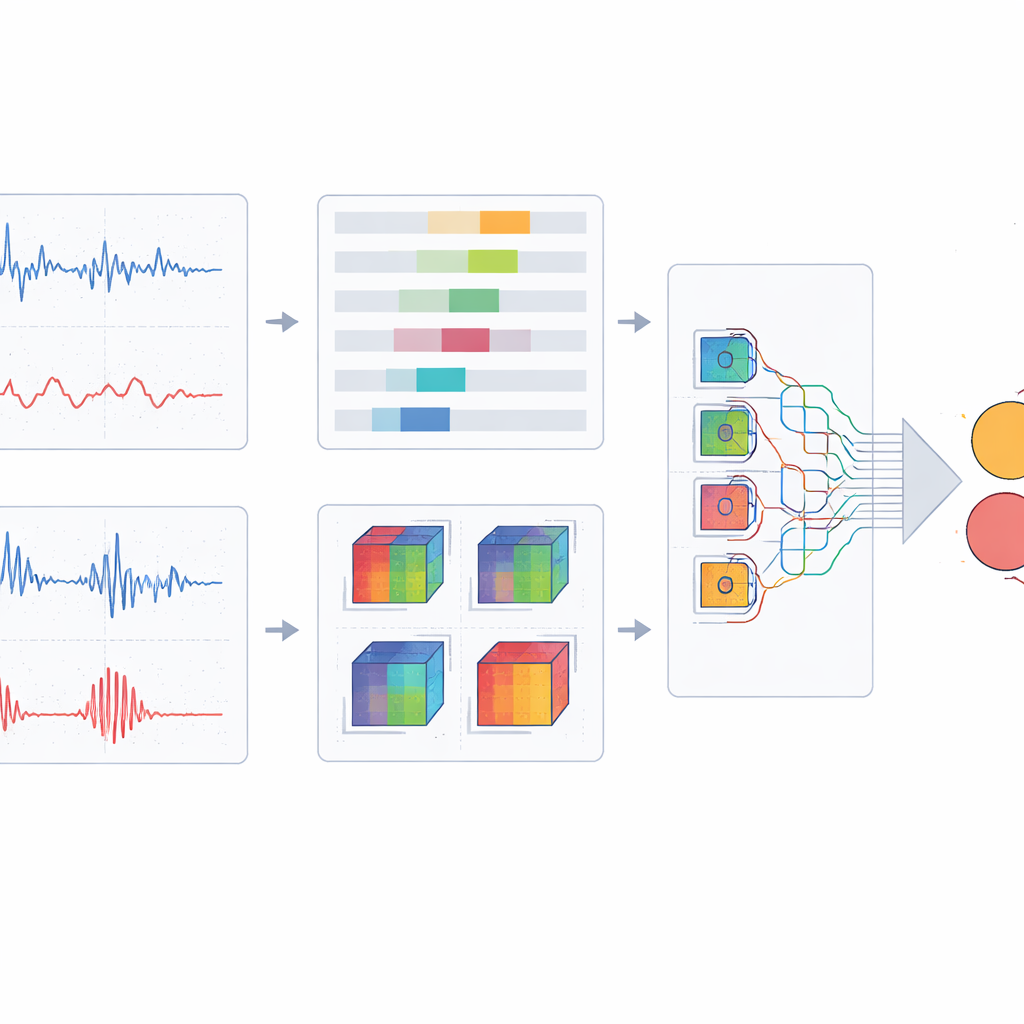
A Lightweight Network That Focuses on What Matters
To classify these fused images into healthy, at‑risk, or neuropathy cases, the team designs a customized deep‑learning architecture built on a modern, efficient backbone. They replace standard convolution blocks with a multi‑branch design that looks at each image patch with kernels of different shapes and sizes, capturing both fine details and broader trends without a large increase in computation. On top of this, they add a spatial attention mechanism that lets the network emphasize informative regions and patterns while downplaying redundant background. The model is trained and tuned using careful cross‑validation, and its performance is compared with a range of well‑known image‑classification networks, including several popular convolutional and transformer‑based designs.
What the Results Mean for People with Diabetes
On data built from multiple heartbeat cycles, the proposed system correctly classifies neuropathy status in nearly 94% of cases, with similarly high sensitivity and precision. It clearly outperforms both traditional signal‑processing setups and strong modern deep‑learning baselines, indicating that the combination of smarter denoising, richer signal‑to‑image encoding, and tailored network design adds real value. While the study is based on data from a single center and will need broader testing, it points toward a future in which routine, painless measurements from simple clips and electrodes—possibly even in primary‑care clinics or at home—could flag early nerve damage in people with diabetes long before serious complications arise.
Citation: Xiao, M., Wang, F., Fang, S. et al. An improved ICEEMDAN–depth hybrid network model integrating multimodal data for the screening of diabetic peripheral neuropathy. Sci Rep 16, 10954 (2026). https://doi.org/10.1038/s41598-026-45862-x
Keywords: diabetic peripheral neuropathy, noninvasive screening, ECG, PPG, deep learning