Clear Sky Science · en
Predicting surgical outcome in drug-resistant epilepsy by combining interictal biomarkers within a machine learning framework
Why this research matters for families with epilepsy
For children whose seizures do not respond to medication, brain surgery can offer the hope of a seizure free life. Yet surgeons must make a critical decision: which tiny regions of brain tissue to remove while sparing healthy areas. This study explores how computer algorithms can sift through routine brain recordings taken between seizures to better guide that choice and help predict which children are most likely to benefit from surgery.
Looking for clues between seizures
Traditionally, doctors rely on brain activity recorded during an actual seizure to estimate the epileptogenic zone, the area thought to trigger seizures. Capturing enough seizures, however, can require days of hospital monitoring and may still miss crucial regions. The authors instead focus on interictal data, the electrical signals recorded when no seizure is happening. These quiet intervals contain brief abnormal events called spikes and ripples that may hint at where seizures begin, but each type alone is often too imprecise to guide surgery. The team set out to see whether combining many properties of spikes and ripples with machine learning could sharpen this picture.
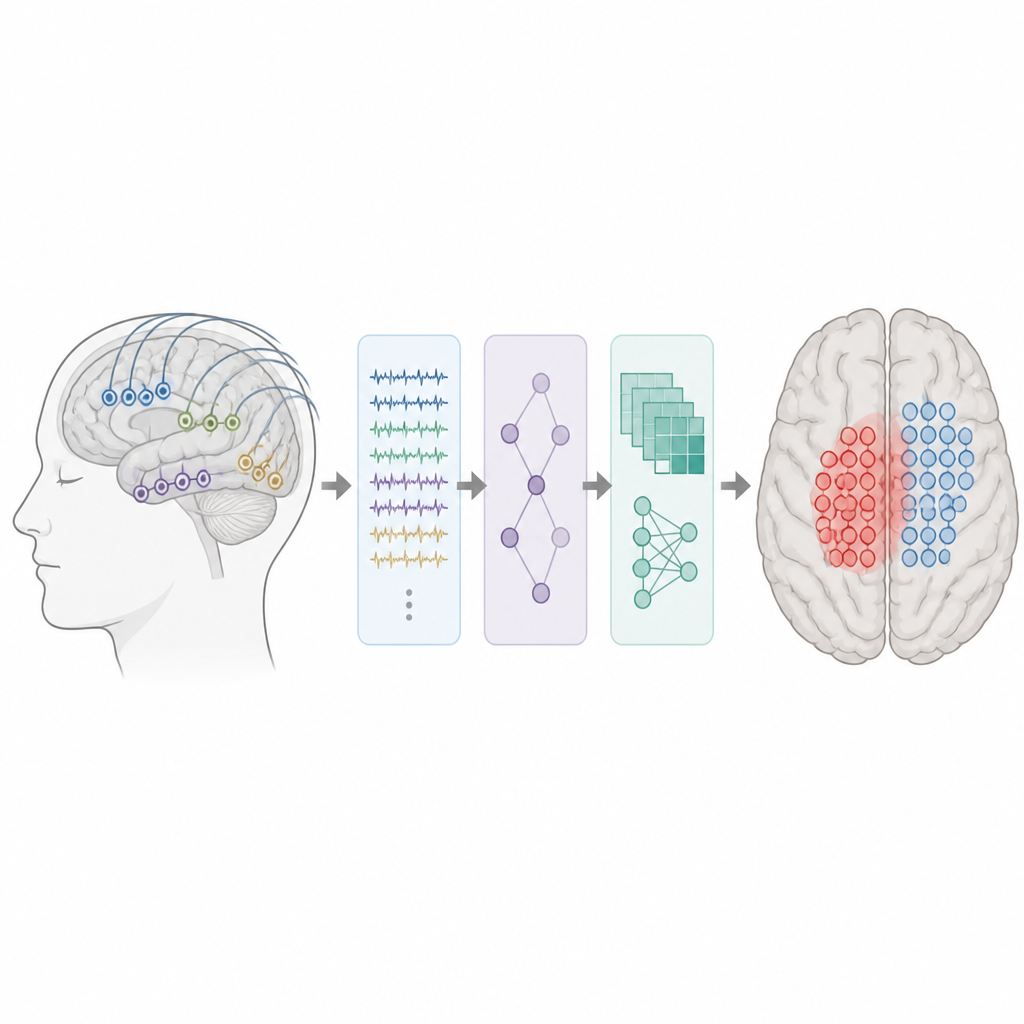
Turning complex brain signals into usable features
The researchers analyzed brain recordings from 62 children and young adults with drug resistant focal epilepsy who underwent invasive monitoring with electrodes placed directly on or in the brain. All had at least five minutes of clean, interictal data and at least one year of follow up after surgery. Automated detectors first scanned the recordings for spikes and ripples. For each electrode, the team then measured how often these events occurred, how strong they were, how they spread across neighboring electrodes, and how close each contact lay to event onsets and hotspots. In total, they created a set of temporal, spectral, and spatial features describing local excitability and how abnormal activity propagated through the brain.
Training machines on successful surgeries
To teach their system what truly epileptogenic tissue looks like, the authors used only patients who became seizure free after surgery. For these children, the tissue that had been removed or ablated most likely contained the critical seizure generating zone. Each electrode inside this resection was labeled as epileptogenic, and those outside as non epileptogenic. Using these labels, the team trained Random Forest classifiers, a type of machine learning model, on three different inputs: spike based features alone, ripple based features alone, or a combination of all spike and ripple features. The models learned patterns that distinguish epileptogenic contacts from the rest and then produced a predicted epileptogenic zone for each patient.
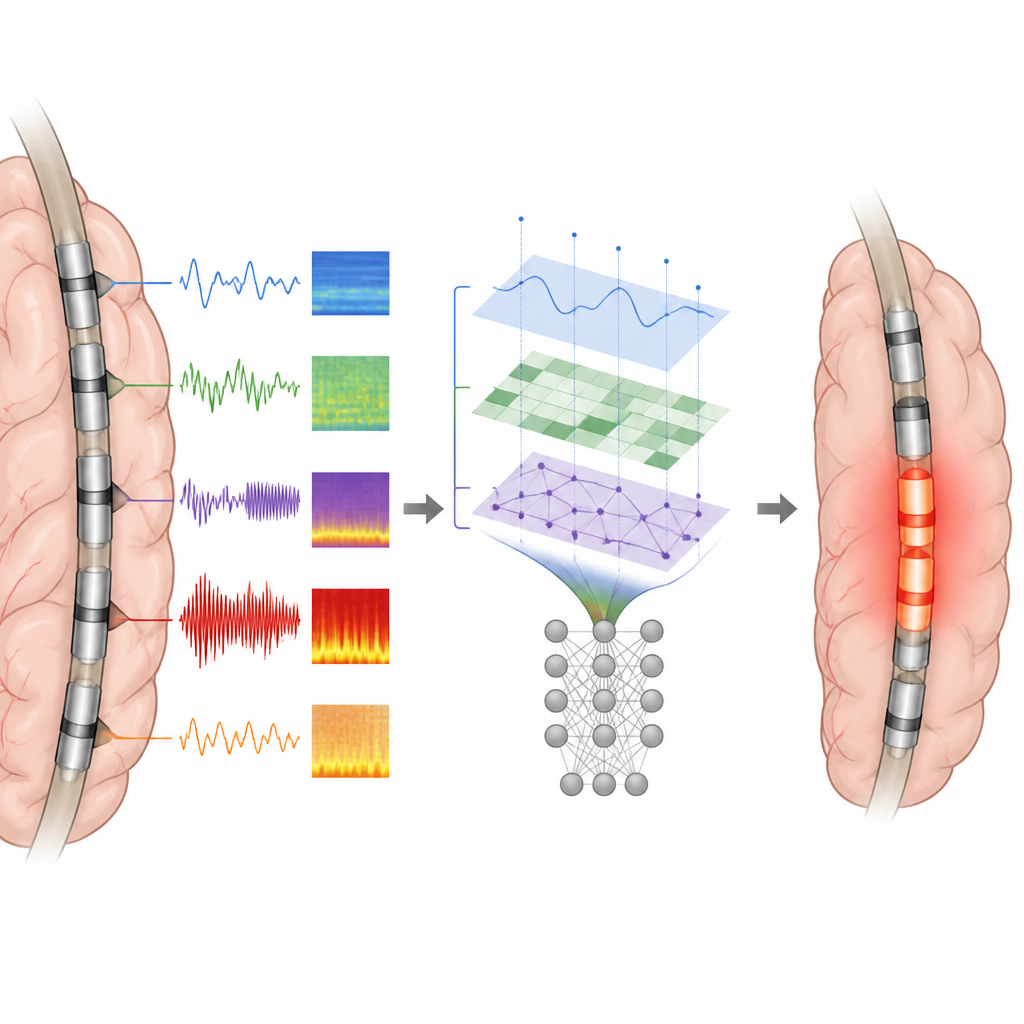
How well the models matched real surgeries
When tested, models based on spike features and on combined spike plus ripple features performed best. They correctly separated epileptogenic from non epileptogenic contacts with high accuracy, and their predicted zones overlapped about three quarters of the actual resection in seizure free patients. By contrast, most single measurements, such as spike rate alone, were less reliable. An important finding was that the exact spatial arrangement of spikes, especially how close an electrode was to the earliest or strongest activity, provided richer information than counts of events. Ripple features added only modest gains beyond spikes, perhaps because ripples can also appear in healthy brain regions.
Connecting predictions to real life outcomes
The team then asked a practical question: if at least half of the predicted epileptogenic zone lay inside the tissue a surgeon removed, did the patient tend to do well? Using the combined feature model, this simple rule identified seizure free children with a sensitivity of 88 percent and specificity of 68 percent, outperforming the traditional seizure onset zone defined by specialists. In patients who continued to have seizures, the predicted zone often lay partly or largely outside the resected region, suggesting that important epileptogenic tissue had been missed or could not be safely removed.
What this could mean for future epilepsy care
For families facing the prospect of epilepsy surgery, this work suggests that a few minutes of brain recording between seizures may someday help surgeons plan operations more precisely and estimate the chances of success. While the approach still needs prospective testing in larger and more uniform groups of patients, it shows that combining multiple subtle features of brain activity with machine learning can give a clearer, more objective view of where seizures arise and how completely surgery has targeted those regions.
Citation: Partamian, H., Jahromi, S., Perry, M.S. et al. Predicting surgical outcome in drug-resistant epilepsy by combining interictal biomarkers within a machine learning framework. Sci Rep 16, 15166 (2026). https://doi.org/10.1038/s41598-026-45177-x
Keywords: epilepsy surgery, interictal EEG, machine learning, pediatric epilepsy, epileptogenic zone