Clear Sky Science · en
Challenges in the standardization of in vitro cytotoxicity assays for comparative risk assessment of cold atmospheric pressure plasma devices
Why this matters for future wound treatments
Cold atmospheric pressure plasma—essentially an energized gas at room temperature—is being tested as a new way to help stubborn wounds heal and to fight germs. Several medical devices using this technology are already on the market, but they differ greatly in how they produce and deliver plasma. This study asks a deceptively simple question with big implications: can lab tests fairly compare the safety and strength of these different devices, or are we mixing apples and oranges without realizing it?
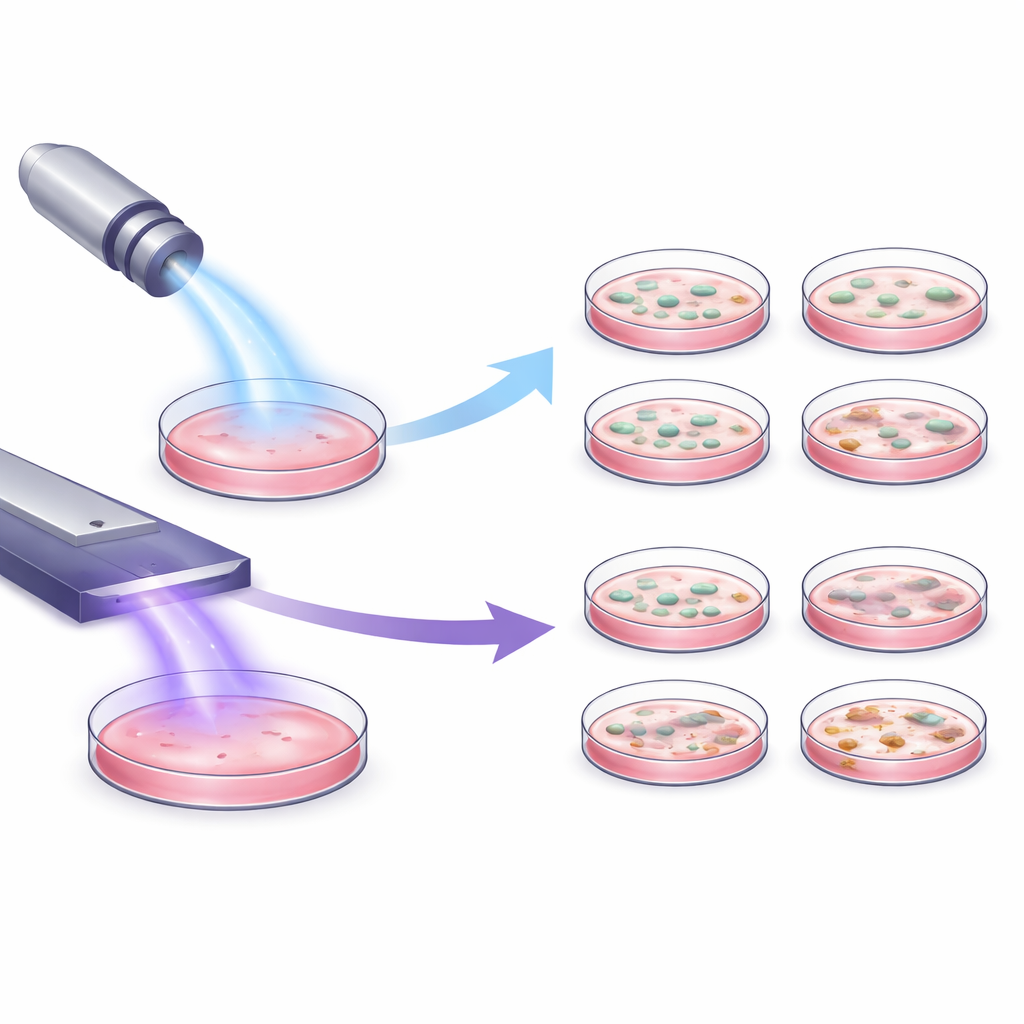
Different tools, different impacts on cells
The researchers examined two medically approved plasma devices that work in very different ways: a pen-like jet that blows a focused stream of plasma, and a flat device that creates plasma directly above the surface it treats. Using three types of skin-related cells grown in dishes, they exposed the cells to each device under what looked like identical settings in the lab. Despite these shared conditions, the cells reacted very differently. The flat device caused little to no loss of cell activity, while the jet device reduced cell metabolism in a clear, time-dependent manner—meaning longer exposure led to stronger effects. Even the flow of inert gas from the jet, without active plasma, measurably affected cell health, showing that gas movement alone can stress cells.
How you move the device changes the outcome
To explore how treatment geometry matters, the team varied the way the plasma jet was moved over the dish. They compared a tight circular path near the center with a larger circle that covered more of the dish surface. Both patterns lowered cell activity over time, but the larger circle did so much more quickly. In other words, the size of the area actually swept by the jet—something that might not be reported in much detail—dramatically altered how strongly the cells were affected, even when exposure time and device settings were the same. This highlights that simple lab parameters like “treatment time” can hide important differences in how plasma truly interacts with cells.
Trying a shortcut with plasma-treated liquid
Because directly exposing cells to plasma jets brings complications—such as liquid being pushed around by gas flow and mismatched device and dish sizes—the researchers also tested an indirect approach. They used a metal grid and a buffer solution (PBS) to create “plasma-conditioned” liquid, which was then transferred onto cells. At first glance, this seemed promising: treating a small volume of liquid through the grid produced a solution that clearly reduced cell activity once applied. But when they treated a much larger volume under nearly the same conditions and then used only a small portion of it on cells, the effect largely disappeared. The team expanded this idea further using larger and smaller culture wells with different liquid depths and treatment times, and again found that simply scaling treatment time with volume did not restore equal strength.
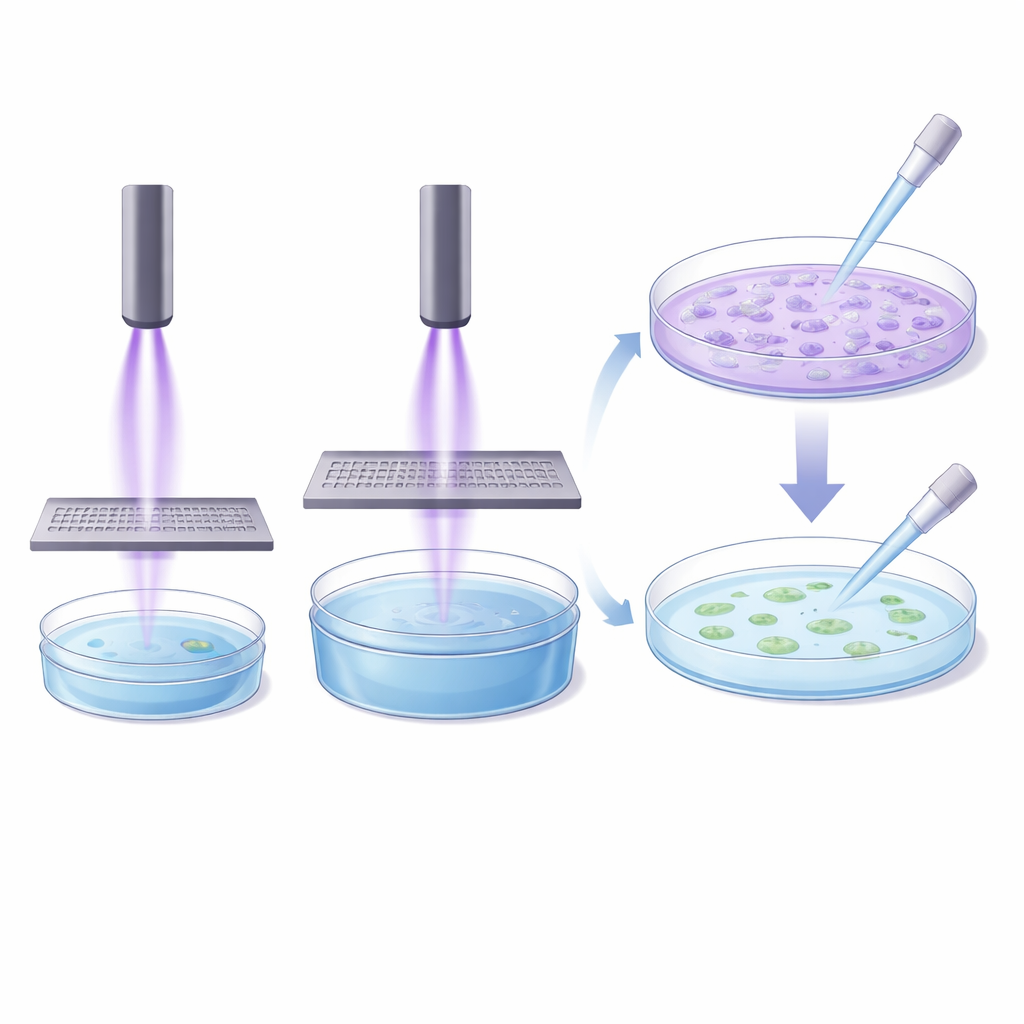
Why simple scaling fails in the lab
The indirect experiments revealed that the relationship between how much liquid is treated, how long it is exposed, and how strongly it affects cells is not straightforward. Larger volumes did not just need proportionally more time; they needed much longer, and even then they often failed to match the impact of smaller volumes. The authors point out that indirect methods also miss short-lived reactive components of plasma, as well as electric and electromagnetic effects that likely contribute to its biological action. Together with the findings from direct treatments, this shows that no single “universal” lab protocol can reliably compare all plasma devices.
What this means for patients and researchers
For people hoping that plasma will become a routine, safe, and effective treatment for wounds or infections, the message is cautious but constructive. The study shows that small differences in device design and lab handling can lead to big differences in how cells respond, making direct head-to-head comparisons misleading if those details are ignored. Instead of forcing all devices into one rigid test, the authors argue that researchers should thoroughly and transparently report key technical and experimental details every time they run an experiment. With this richer context, results from different labs and devices can still be meaningfully compared and combined, building a more reliable evidence base before new plasma tools are tried in rigorous clinical trials.
Citation: Boeckmann, L., Ficht, PK., Bernhardt, T. et al. Challenges in the standardization of in vitro cytotoxicity assays for comparative risk assessment of cold atmospheric pressure plasma devices. Sci Rep 16, 10503 (2026). https://doi.org/10.1038/s41598-026-45037-8
Keywords: cold atmospheric plasma, wound healing, cell culture assays, medical device comparison, treatment parameters