Clear Sky Science · en
Lack of evidence for cargo release of CD63-EVs into recipient cells
How tiny cell packages caught scientists by surprise
Our cells constantly send out nanosized bubbles packed with proteins and genetic material, often described as little parcels for cell to cell messages. Many researchers have suggested that these parcels deliver their contents directly into other cells, shaping health and disease in ways we are only beginning to grasp. This study takes a hard look at that idea and finds that, at least for a common type of vesicle, the delivery of cargo into recipient cells may be far less efficient than many have assumed. 
Small bubbles that carry big expectations
Cells release tiny membrane bound bubbles called extracellular vesicles into their surroundings. A major subclass, often called exosomes, buds from internal compartments and carries molecules such as proteins and RNA. Because these vesicles can be taken up by distant cells, they have been widely proposed as a means for long range cell communication and as potential vehicles for future therapies. A key open question, however, is whether their cargo truly escapes into the interior of the recipient cell, where it could change how that cell behaves.
A molecular light switch to track real entry
To address this, the researchers used a very sensitive reporting system that behaves like a molecular light switch. One half of a light producing enzyme, called HiBiT, was placed inside vesicles by fusing it to CD63, a standard marker protein that is abundant in these bubbles. The matching half, LgBiT, was produced inside recipient cells, either freely in the fluid interior or attached to endosomal membranes, which are thought to be major landing spots for incoming vesicles. If a vesicle truly fused with a cell membrane and released its inner contents, the two halves would meet, lock together, and emit a strong glow that can be accurately measured.
Vesicles enter cells but keep their secrets
First, the team confirmed that their engineered CD63 molecules were correctly built into vesicles without changing their size or shape. When these vesicles were mixed with cells, imaging and biochemical tests showed that they could bind to cells and likely be pulled inside by standard uptake routes. Yet, despite this clear interaction, the expected light signal did not increase over time. Even when LgBiT was concentrated on endosomes, or when another common vesicle cargo protein, HSP70, was used instead of CD63, the readout remained at background levels. Only when both vesicles and cells were artificially permeabilized with detergents did the latent light signal appear, indicating that the cargo stayed trapped behind intact membranes.
Viral fusion machinery changes the story
To be sure the assay was capable of detecting rare fusion events, the scientists equipped some vesicles with VSV G, a viral fusion protein known to help viruses merge with cell membranes. This time, when VSV G bearing vesicles carrying the HiBiT tag were added to LgBiT cells, luminescence rose clearly over a few hours, proving that the system can report cargo release when fusion truly occurs. Interestingly, only a small fraction of vesicles carried this viral protein, yet the signal was robust, highlighting the sensitivity of the method. Vesicles with VSV G were also taken up somewhat more efficiently, but the much larger jump in light output showed that fusion, not just uptake, was the critical missing step in the unmodified vesicles. 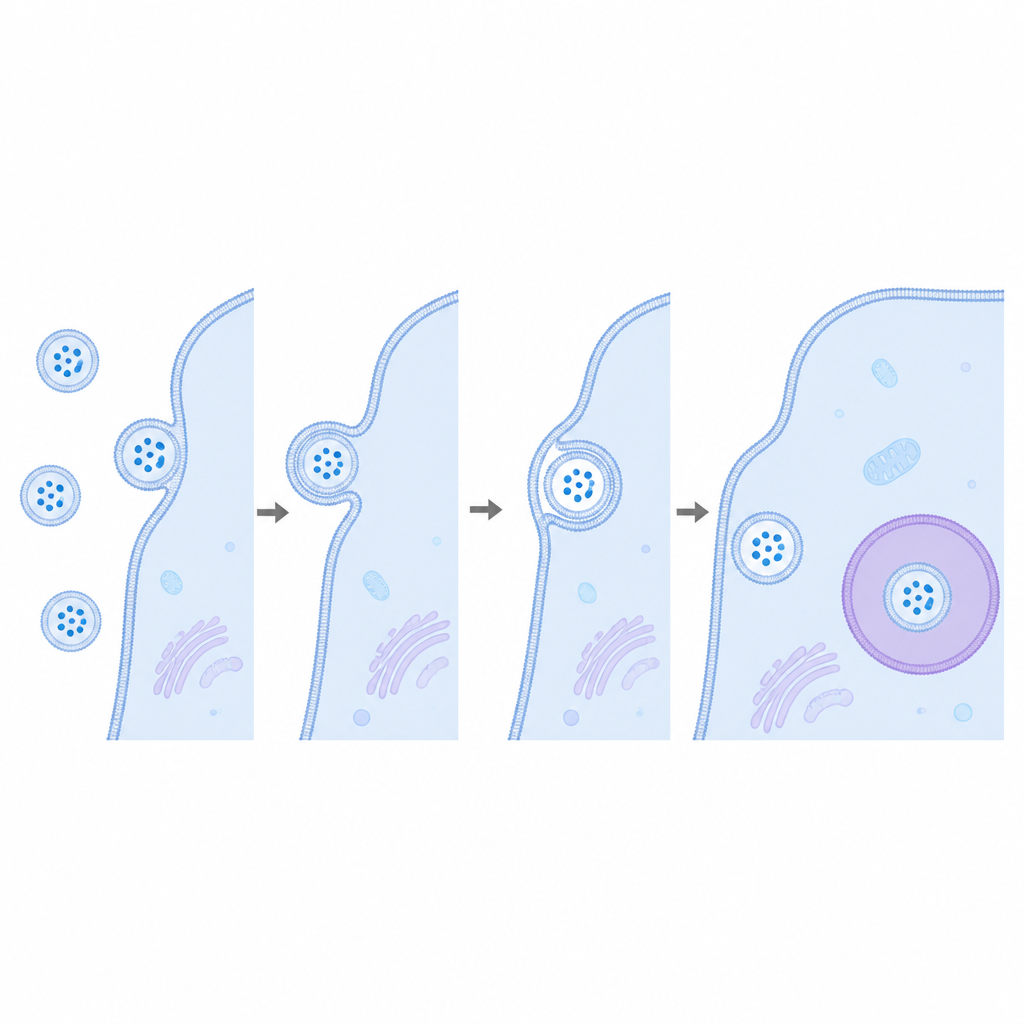
Rethinking how these bubbles talk between cells
Taken together, the work suggests that under standard lab conditions, CD63 positive vesicles from human kidney cells rarely, if ever, fuse with recipient cell membranes to spill their inner cargo into the cell interior. They may still bind, be swallowed into internal compartments, or signal through surface interactions, but direct delivery of their internal load appears inefficient without added fusion helpers such as viral proteins. For a general reader, the message is that these natural cell made parcels may not be the straightforward message carriers many had imagined, and that scientists will need to rethink both how cells communicate with them and how best to harness them for future treatments.
Citation: Askarian-Amiri, S., Weissenhorn, W., Sadoul, R. et al. Lack of evidence for cargo release of CD63-EVs into recipient cells. Sci Rep 16, 15164 (2026). https://doi.org/10.1038/s41598-026-45021-2
Keywords: extracellular vesicles, exosomes, cell communication, membrane fusion, nanoluciferase assay