Clear Sky Science · en
Tumor-suppressing multi-enterobacteria enhance the anti-PD-1/PD-L1 efficacy in microsatellite stable colorectal cancer
Why Your Gut Bacteria Matter for Colon Cancer Treatment
Immunotherapy has transformed care for some cancers, but for most people with a common form of colorectal (colon) cancer, these drugs still do not work well. This study explores a simple but powerful idea: can we boost the body’s cancer-fighting immune response by deliberately reshaping the bacteria that live in the gut using a tailored mix of probiotics, so that standard immunotherapy becomes much more effective?
A Tough-To-Treat Type of Colon Cancer
Colorectal cancer is one of the leading causes of cancer death worldwide. A group of drugs called immune checkpoint inhibitors helps the immune system recognize and attack tumors and has been very successful in some cancers such as melanoma and lung cancer. In colon cancer, however, only a small subset of patients whose tumors have a specific genetic feature called microsatellite instability benefit strongly from these drugs. Most patients have microsatellite-stable tumors that usually respond poorly. Finding ways to “wake up” the immune system in these resistant tumors is a major challenge in oncology.
A Custom Probiotic Team Designed for Battle
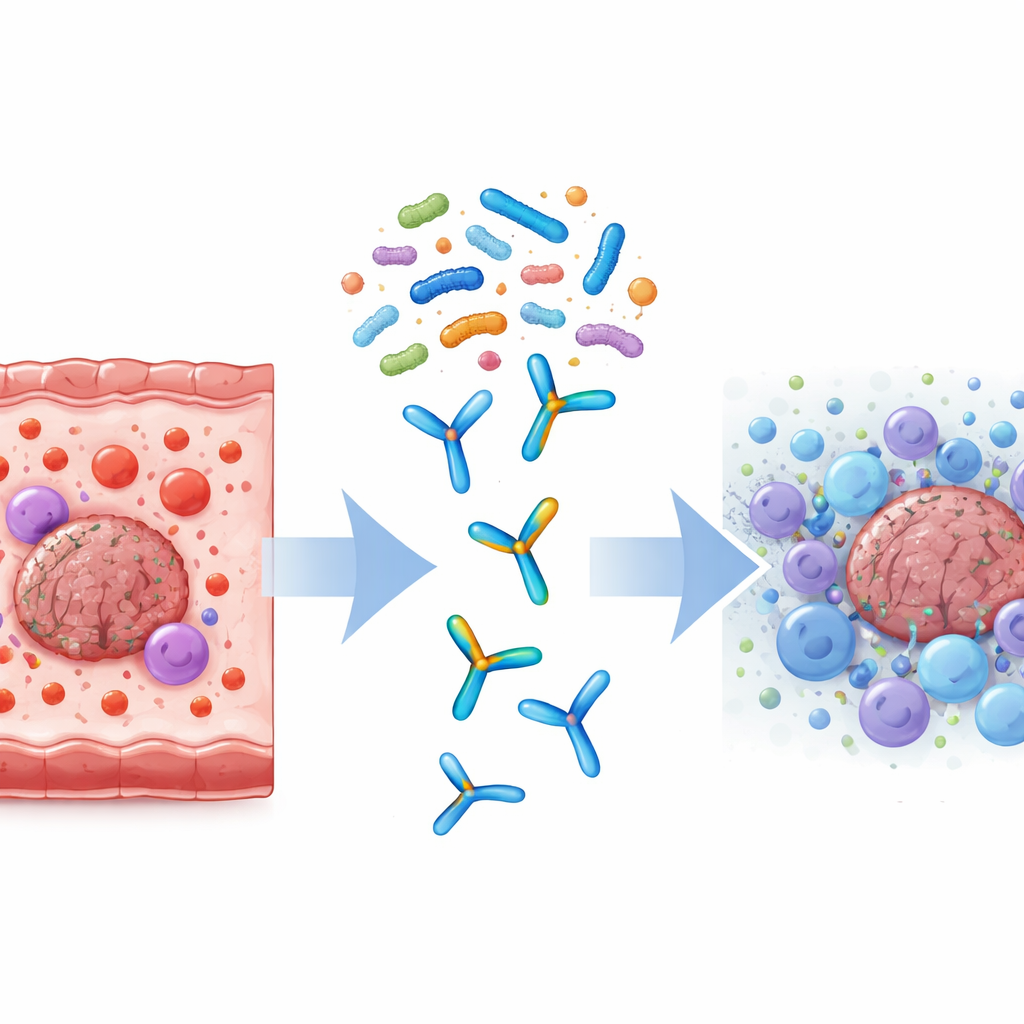
The researchers created a probiotic cocktail called Tumor-Suppressing Multi-Enterobacteria (TSME), made of nine well-known, food-grade strains of Bifidobacterium, Lactobacillus and Streptococcus. These strains were chosen because they can survive the harsh journey through the stomach, are commonly found in healthy people, and have evidence of supporting immune health or slowing tumor growth. In mice implanted with microsatellite-stable colorectal tumors, the team compared several treatments: probiotics alone, immunotherapy drugs that block PD-1 or PD-L1 alone, or the combination of probiotics plus immunotherapy.
Stronger Tumor Control and More Immune Soldiers
When mice received both TSME and anti-PD-1 or anti-PD-L1 drugs, their tumors grew much more slowly than in animals treated with the drugs alone. The mice maintained healthy body weight, suggesting the probiotic mix was well tolerated. Inside the tumors, the combination treatment drew in many more CD8+ T cells—the “killer” immune cells that can directly destroy cancer cells—than immunotherapy on its own. At the same time, blood tests and tissue analyses showed a healthier balance of signaling proteins called cytokines: harmful, pro-inflammatory molecules such as IL-1β, IL-6, IL-17 and TNF-α decreased, while IFN-γ, which supports anti-tumor activity, increased. Together, these changes indicate that the tumors shifted from an immune-poor, “cold” state to a more active, “hot” state that responds better to checkpoint blockade.
Remodeling the Microbial Neighborhood
The scientists also examined how the probiotic mix changed the gut ecosystem itself. Using genetic sequencing of bacterial DNA from mouse stool, they found that the combination of TSME with anti-PD-L1 therapy produced a distinct and more balanced microbial community compared with control animals or those receiving immunotherapy alone. Notably, the combination group had higher levels of bacteria such as Akkermansia and Alistipes, which have been linked in other studies to better metabolism, stronger gut barrier function, and improved responses to cancer immunotherapy. Computational analysis suggested that the new microbial makeup was associated with functions and pathways that can support a more effective immune response against tumors.
Switching On Key Immune Pathways Inside the Tumor
To see what was happening inside the tumor cells and their surroundings, the team measured gene activity in tumor tissue using RNA sequencing. Compared with immunotherapy alone, tumors from mice treated with both TSME and anti-PD-L1 showed hundreds of genes turned up or down, especially those involved in immune responses. Important communication routes, including TNF signaling, cytokine–receptor interactions, and the JAK–STAT pathway, were more active. Several genes tied to immune cell activation and tumor cell death were boosted, supporting the idea that reshaping the gut bacteria helps reprogram the tumor environment into one that is more vulnerable to immune attack.
What This Could Mean for Future Cancer Care
This work, done in mice, suggests that a carefully designed probiotic mix can make stubborn colon tumors far more sensitive to existing immunotherapy drugs, by both rebalancing gut bacteria and rewiring immune activity in and around the tumor. While more research and clinical trials are needed before this approach can be used in patients, the study points toward a future where simple, safe bacterial blends become part of personalized cancer treatment plans—helping the immune system recognize and clear tumors that previously resisted some of our most promising drugs.
Citation: Su, X., Jin, J., Huang, Y. et al. Tumor-suppressing multi-enterobacteria enhance the anti-PD-1/PD-L1 efficacy in microsatellite stable colorectal cancer. Sci Rep 16, 14069 (2026). https://doi.org/10.1038/s41598-026-44494-5
Keywords: colorectal cancer, immunotherapy, gut microbiome, probiotics, PD-1 PD-L1