Clear Sky Science · en
Temperature-dependent gating pathways in TRPV3
How Our Skin Senses Hot and Cold
Our skin can tell a warm mug from an icy glass in an instant, thanks to tiny protein gates in cell membranes that open and close with temperature. One such gate, called TRPV3, helps detect warmth and participates in skin biology. This study asks a simple but deep question: do the same basic rules explain how this protein senses both heat and cold, even when its activity later shuts itself off?
Two Temperature Roads to One Sensor
TRPV3 is a four-part protein channel that sits in the cell membrane and lets charged atoms flow when it opens. Earlier work suggested that its response to heat could be described by a classic thermodynamic equation that links temperature changes to shifts in energy and heat capacity. That equation predicts that, under the right conditions, the same channel should also respond to cooling. However, TRPV3 is tricky because once activated it can inactivate, meaning the channel stops conducting even if the stimulus remains. The author set out to see whether both the initial hot and cold responses still follow the same energetic rule, despite these later changes.
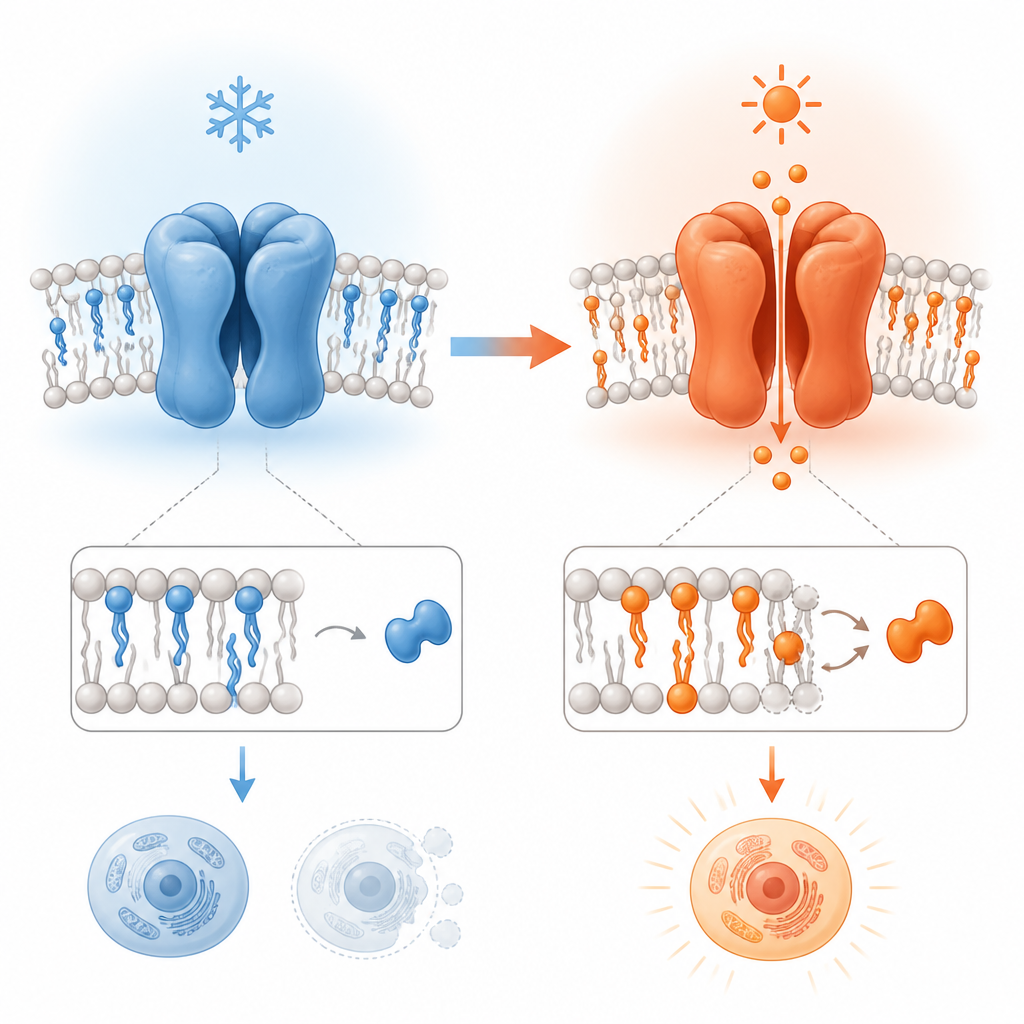
Watching the Channel Change Shape
To probe this, the study compared high-resolution cryo-electron microscopy structures of TRPV3 from mouse and human samples at different temperatures. The channel was examined in membrane-like discs that include natural fats and a special plant-derived molecule, tetrahydrocannabivarin (THCV), which can bind at a site normally occupied by a lipid. By looking at TRPV3 with and without THCV at cold temperatures, and comparing those structures to a heat-activated form, the work mapped the stepwise structural changes that accompany opening, inactivation and an unusual “pore-dilated” state in which the channel widens and changes its assembly.
Hidden Heat Rules in a Cold Response
The key analysis treats the protein as a network of small noncovalent links between amino acids. These links form loops, or “thermorings,” whose size and strength reflect how stable each part of the structure is. The least stable link in the largest ring acts like a thermal weak spot that sets the temperature at which a major rearrangement occurs. By tracking how these weak spots and loops shift between closed, open and inactivated states, the author could estimate the temperature thresholds for each transition. Importantly, when a membrane lipid at the vanilloid site was displaced by THCV, the initial cold-induced opening showed a structural temperature sensitivity that closely matched the known heat sensitivity measured for TRPV3 under warming. This mirrored behavior supports the idea that a single heat capacity change can underlie both hot and cold sensing.
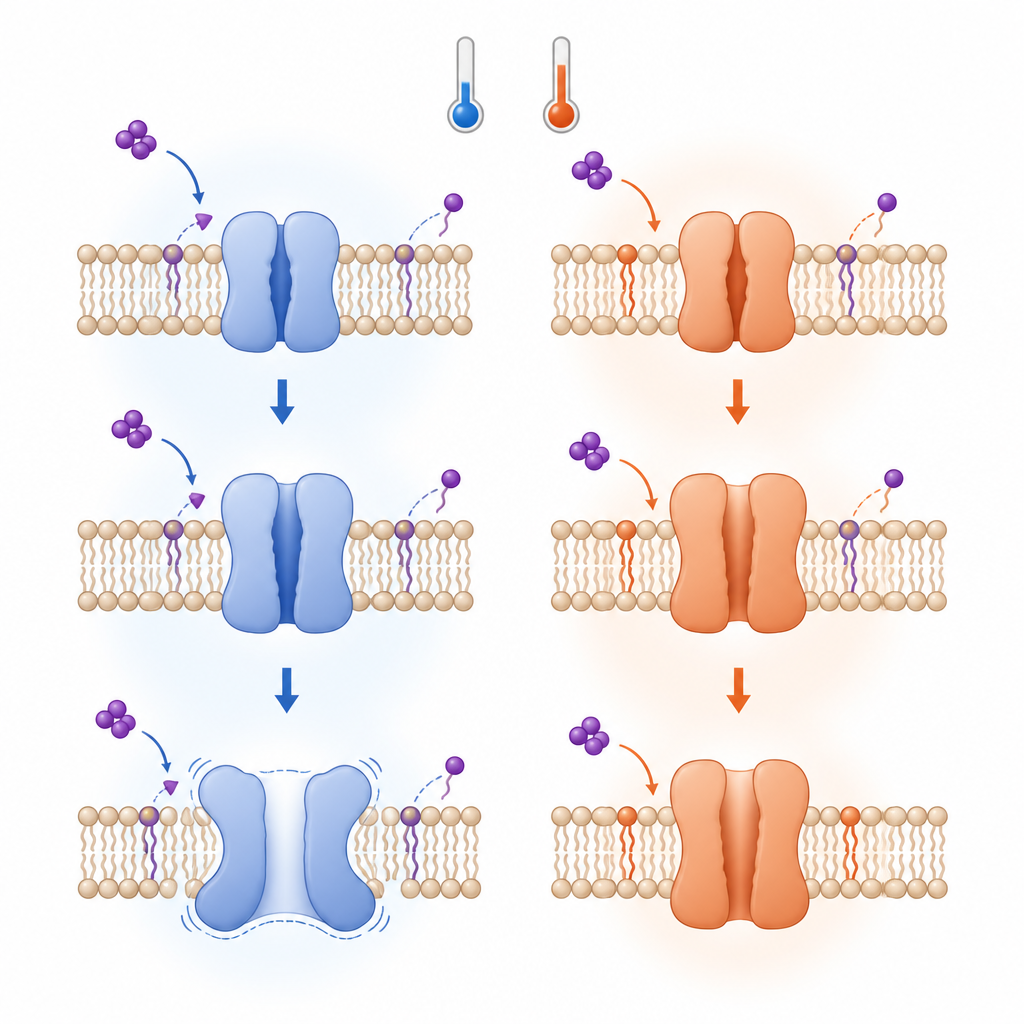
Why Inactivation Looks So Different
Although the early steps of hot and cold activation obey the same thermodynamic rule, the later steps diverge. At low temperatures, after TRPV3 opens, further rearrangements stabilize an inactivated state and can even drive a shift from the usual four-part assembly to a five-part, pore-dilated form with a widened opening. These changes involve the breaking of specific contacts that normally couple different segments of the channel, making the structure more stable in some respects but also less predictable. Above about 30 degrees Celsius, by contrast, once the lipid has been displaced and the channel opens, it tends to remain open without passing through the same inactivation pathway.
What This Means for Sensing Heat and Cold
For a non-specialist, the central message is that TRPV3 behaves like a thermal gadget whose first reaction to hot and cold is governed by the same underlying energy rule, even though its later behavior can branch into different structural routes. The work shows that both warming and cooling can start from the same molecular “starter” at a lipid binding site and produce openings with similar sensitivity to temperature. After that, especially at lower temperatures, the channel may shut itself off or even remodel into a different assembly. This supports a general model in which a single thermodynamic framework can explain how one sensor protein contributes to both hot and cold feeling, while also highlighting how added layers of structural change shape what our cells ultimately do with that signal.
Citation: Wang, G. Temperature-dependent gating pathways in TRPV3. Sci Rep 16, 15030 (2026). https://doi.org/10.1038/s41598-026-44194-0
Keywords: TRPV3, temperature sensing, ion channels, thermodynamics, cryo electron microscopy