Clear Sky Science · en
Hypoxia-induced gene expression changes in N. vectensis embryos
Why oxygen matters for tiny sea embryos
Earth’s first animals arose in ancient seas where oxygen levels rose and fell over time. This study asks a simple but far-reaching question: how do very young animal embryos cope when the oxygen they depend on suddenly drops? By watching the embryos of a small sea anemone, the researchers uncovered how early life can temporarily pause its own development during low-oxygen episodes and then restart once conditions improve. These insights help us imagine what early animal life faced hundreds of millions of years ago—and how today’s animals still carry molecular traces of those challenges.
Ancient oceans with shifting breath
Geologists think that during the Neoproterozoic era, more than half a billion years ago, oxygen in the oceans did not simply rise and stay high. Instead, especially in shallow coastal seas where early animals lived, oxygen likely swung between richer and poorer levels over days and seasons. For any developing animal, such swings would pose a serious problem: building a complex body from a single cell is energy-hungry work, and that energy usually comes from oxygen-based metabolism. The team reasoned that embryos of early-branching animals might reveal how the first metazoans adapted to such unstable conditions.
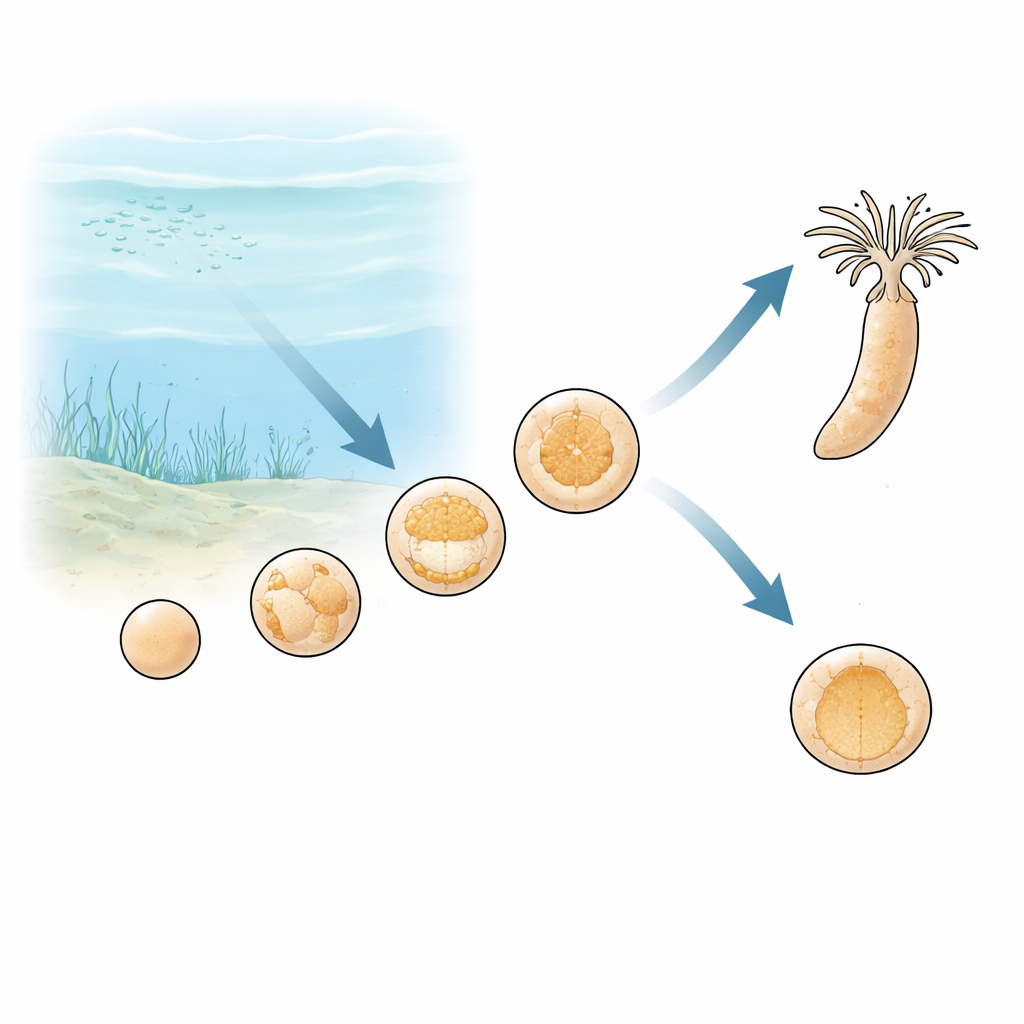
A sea anemone embryo under stress
The researchers turned to Nematostella vectensis, a starlet sea anemone that represents one of the earliest branches of the animal family tree. They built a tightly controlled culture system in which seawater flowing over embryos could be held at normal oxygen or driven to extremely low levels. Under normal conditions, the embryos move from a loose ball of cells to a hollow sphere, then on to a stage called gastrulation, when cells fold inward to form the basic body layers. When oxygen was stripped from the water before or during this folding stage, development did not collapse outright. Instead, embryos stopped progressing and remained as simple hollow spheres, yet stayed alive and structurally intact.
Paused, not broken
To see whether this halt was permanent damage or a controlled pause, the team restored oxygen after several hours of deprivation. Embryos that had stalled before gastrulation restarted development after a delay: cell layers began to fold inward again, and most embryos eventually reached the more advanced gastrula stage. Tests that label dividing cells showed what was happening inside. In low oxygen, DNA copying and cell division almost completely shut down. Within just one to two hours of re-oxygenation, cell division rebounded rapidly, even before visible shape changes returned. This pattern shows that the embryos enter a reversible “quiescent” state—turning down growth and movement until enough oxygen is available to power the next steps.
Genes that sense and adapt to low oxygen
The team then examined which genes turned on or off in embryos exposed to hypoxia at different stages. Thousands of genes changed their activity, with the strongest shifts in younger embryos that were most sensitive to oxygen loss. The affected genes formed distinct sets depending on developmental time: early on, many were tied to cell structures, chromosome handling, and other components needed for tissue shaping; later, more were involved in managing energy use, protein quality control, and cell metabolism. Classic low-oxygen pathways known from more complex animals also appeared. Key marker genes associated with the Hypoxia Inducible Factor system and with stress-response networks such as AMPK signaling, the unfolded protein response, and antioxidant production were activated, even though some core switch genes themselves did not rise in abundance. This pattern suggests that the molecular toolkit animals use today to endure low oxygen was already present in very ancient lineages.
What this means for the story of animals
Seen together, the results portray the anemone embryo as a tiny survival artist. When oxygen drops, it does not simply die; it actively slows its cell cycle, reshapes its gene activity, and waits in a safe holding pattern until conditions improve, at which point development resumes. Because these strategies closely resemble those seen in worms, insects, and vertebrates, the study argues that a shared, oxygen-sensitive genetic program was likely in place in the common ancestor of modern animals. For a lay observer, the takeaway is that the ability of embryos to pause and restart growth in response to changing oxygen may have been a key innovation that allowed complex animal life to thrive in Earth’s unstable ancient seas.
Citation: Hadife, S., Wang, H., Hongo, Y. et al. Hypoxia-induced gene expression changes in N. vectensis embryos. Sci Rep 16, 14315 (2026). https://doi.org/10.1038/s41598-026-44143-x
Keywords: hypoxia, embryo development, sea anemone, oxygen evolution, stress response genes