Clear Sky Science · en
Fractal-fractional modeling of chronic myelogenous leukemia immune dynamics using Laguerre wavelets method
Why this matters for blood cancer and the immune system
Chronic myelogenous leukemia (CML) is a blood cancer that unfolds over years, while the immune system constantly tries to keep it in check. Doctors now have powerful drugs, but predicting how a patient’s cancer and immune cells will interact over time is still difficult. This paper introduces a new kind of mathematical model that treats the tumor and immune response as a coupled dynamic system with “memory” and complex structure. The goal is to build more realistic tools that can ultimately help design smarter therapies and long-term treatment plans.
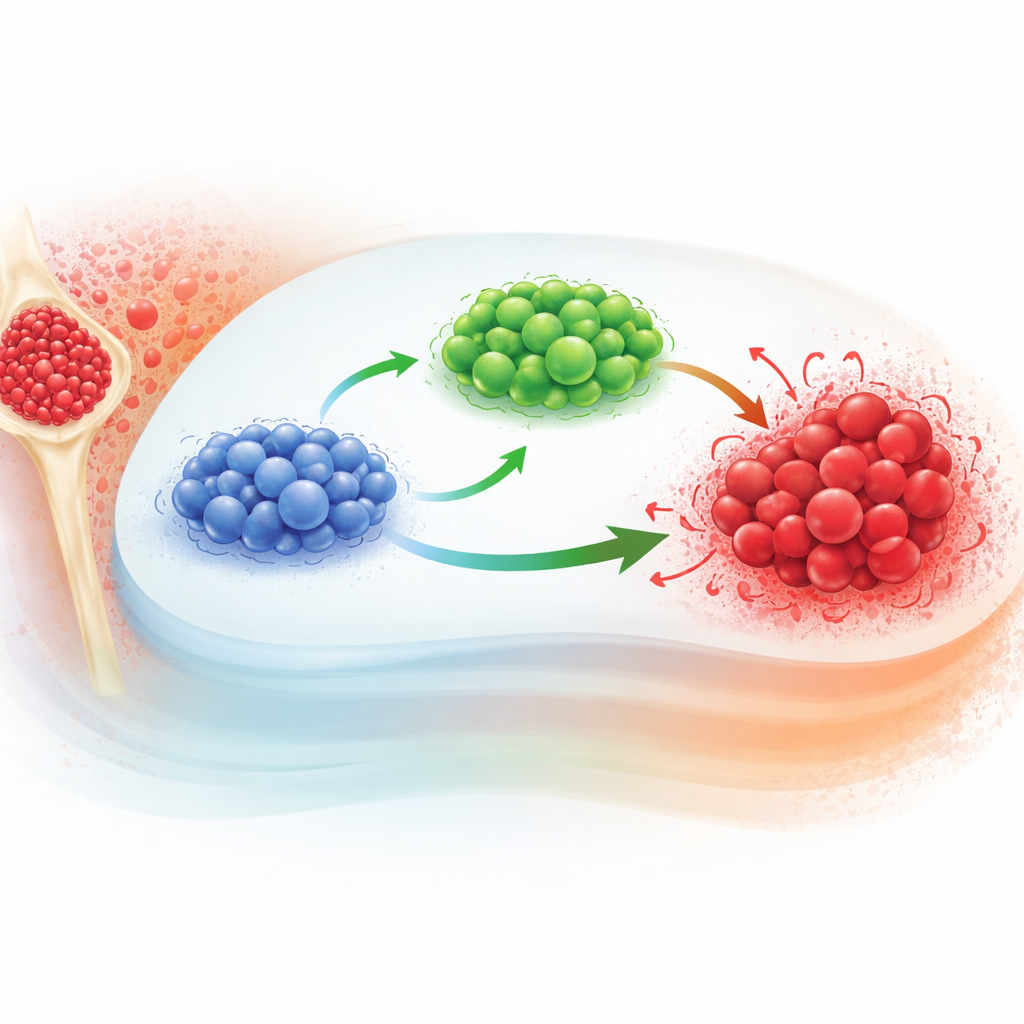
How the cancer and immune cells interact
The authors start from an existing model that tracks three key players in CML: naive T cells, effector T cells, and leukemia cells. Naive T cells are the inexperienced recruits; when they encounter leukemia signals, some become effector T cells, the front-line fighters that attack cancer cells. The cancer cells themselves grow following a saturating law: they expand quickly when small, then slow as they approach a maximum size set by their environment. Standard versions of this model use ordinary differential equations, which assume that what happens next depends only on the current state, not on how the system got there.
Adding memory and rough structure to the model
Real biology is not memoryless or smooth. Immune responses depend on past encounters, and tumors grow in irregular, patchy patterns rather than neat balls. To capture both effects at once, the authors rebuild the CML model using what they call fractal–fractional operators. One part, controlled by a parameter called the fractional order, encodes memory: the system’s future depends on a weighted history of its past. The other part, controlled by a fractal dimension, captures how irregular or “rough” the tumor’s environment is. When both parameters are set to one, the model behaves like the classical version; when they are reduced, history and geometric complexity become more important.
Ensuring the new equations behave sensibly
Before trusting such a model, one must show that its solutions make biological sense. The authors prove that, under reasonable conditions, their equations have a unique solution that stays positive and bounded over time—cell counts never become negative or blow up to infinity. They then analyze the system’s steady states. In this framework, a completely tumor-free state is mathematically unstable: any small introduction of leukemia cells will grow. Instead, the system is drawn toward a “tumor-present” equilibrium where cancer and immune cells coexist. Using tools from stability theory, they show that this coexistence state is not just locally stable but globally attractive: all realistic starting conditions evolve toward it.
Fast numerical methods to solve a slow disease
Because the new equations involve memory and fractal effects, they are harder to solve on a computer than standard ones. The authors develop a numerical technique based on Laguerre wavelets, a family of building-block functions that can efficiently approximate behavior over long times. This method turns the original problem into a smaller system of algebraic equations that can be solved quickly and accurately. When they compare it to more familiar time‑stepping schemes, the wavelet method achieves the same accuracy with far fewer computations, which is important for running many simulations or fitting the model to patient data.
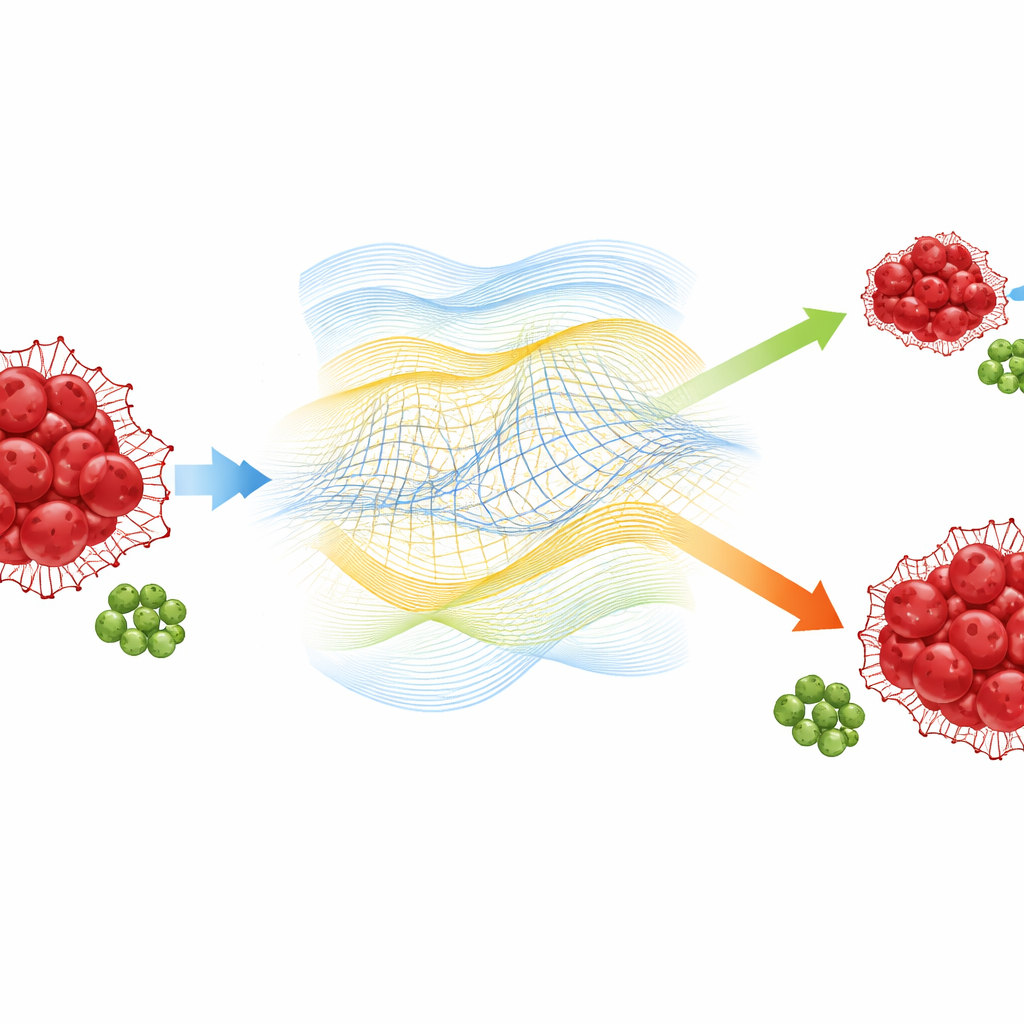
What the simulations reveal about disease control
With these tools in hand, the authors explore how changing the memory and fractal parameters shapes disease outcomes. Lowering the fractional order (stronger memory) or lowering the fractal dimension (more irregular tumor geometry) both drive the leukemia population to decay faster and settle at a smaller long‑term level. In other words, a system that “remembers” its past and a tumor that grows in a more intricate environment are easier for the immune system to restrain. A sensitivity analysis shows that two biological quantities dominate the final tumor size: the cancer’s own growth rate and the rate at which effector T cells kill cancer cells. This aligns with current therapy strategies that combine targeted drugs to slow cancer growth with immunotherapies that boost immune killing.
What this means for patients and future work
In plain terms, the study concludes that CML is unlikely to vanish on its own; the natural tendency of the system is to settle into a balance where cancer and immune cells coexist. However, by building a model that includes memory and structural complexity, the authors can describe scenarios where that balance favors the patient, with a lower long‑term cancer load. Their fractal–fractional framework, coupled with efficient numerical methods, lays groundwork for future “digital experiments” on treatment timing and combinations. With clinical data, the same approach could be personalized, tuning the memory and geometry parameters for individual patients to forecast disease trajectories and optimize therapy.
Citation: Khirsariya, S.R., Noori, N. Fractal-fractional modeling of chronic myelogenous leukemia immune dynamics using Laguerre wavelets method. Sci Rep 16, 14106 (2026). https://doi.org/10.1038/s41598-026-43767-3
Keywords: chronic myelogenous leukemia, tumor immune dynamics, fractional calculus, fractal tumor modeling, numerical wavelet methods