Clear Sky Science · en
Identification of immunogenic neoantigens from intron retention in colorectal cancer
Turning Hidden Genetic Glitches into Cancer Targets
Colorectal cancer is one of the most common cancers worldwide, and for many patients, especially those with tumors that carry few DNA mutations, today’s personalized cancer vaccines have little to work with. This study reveals a surprising new source of vaccine targets hiding not in classic gene mutations, but in the way tumor cells process their RNA. By focusing on a process called intron retention—where pieces of raw genetic messages are mistakenly left in—researchers uncover a rich supply of unusual protein fragments that the immune system can recognize and potentially attack.
Why Cancer Vaccines Need New Targets
Most experimental cancer vaccines today are built from “neoantigens”—small protein fragments that appear only on tumor cells but not on healthy tissues. Traditionally, scientists hunt for these targets by scanning a patient’s tumor DNA for point mutations and other small changes, then predicting which altered protein pieces might be shown to immune cells. The problem is that many tumors, including the common microsatellite-stable form of colorectal cancer, simply do not carry enough DNA mutations to generate many strong neoantigens. That scarcity makes it difficult to design vaccines for large groups of patients and limits how effective these treatments can be.
When the Cell’s Editing Machinery Slips
Instead of looking only at DNA changes, the authors turned to RNA, the intermediate messages cells read to make proteins. Normally, cells remove non-coding segments called introns from these messages before translation. In many cancers, however, this editing process becomes sloppy, and introns are accidentally left in. The team analyzed RNA sequencing data from tumors and nearby healthy tissue from 23 colorectal cancer patients using a specialized software pipeline. They found that tumor samples consistently showed more intron retention events than healthy tissue, with hundreds of such events per tumor and thousands of introns that were more frequently retained across the patient group. Many of the affected genes were involved in cell division and DNA handling—key processes that are often distorted in cancer.
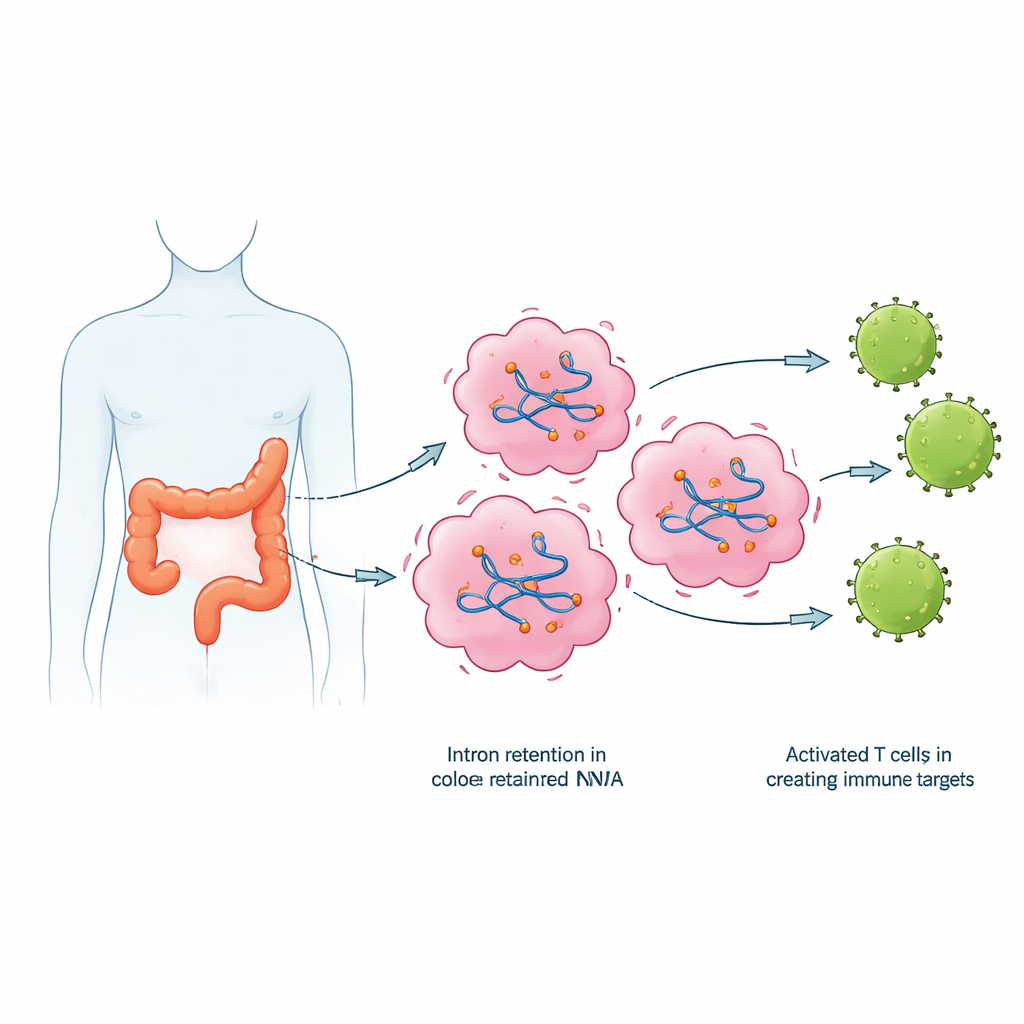
From Faulty Messages to New Immune Flags
Retained introns do more than clutter RNA; they can be turned into abnormal protein fragments that the immune system has never seen before. The researchers translated retained intron sequences into short peptides and used computer tools to predict which ones could bind tightly to common immune display molecules (HLA proteins) on cell surfaces. They then cross-checked public protein datasets to keep only peptides seen in colorectal cancer samples but not in healthy tissues. From this screen, they selected a set of candidate peptides and confirmed, using sensitive PCR tests, that many of the underlying intron-containing RNAs were indeed present at higher levels in tumors than in matched normal tissue.
Teaching T Cells to Recognize the New Targets
To see whether these intron-derived fragments could truly rouse the immune system, the team trained human CD8 T cells from healthy donors using lab-grown immune cells loaded with the candidate peptides. Several peptides triggered strong responses: trained T cells secreted immune signals, turned on activation markers, and showed signs of killing when they encountered cells displaying the matching peptide. Additional mass-spectrometry evidence suggested that at least some of these peptides are naturally produced inside colorectal tumors. Strikingly, the authors also identified 24 intron-derived neoantigens that appeared in roughly 30% of patients and were predicted to bind well to the most common HLA types in the group, hinting at the possibility of shared, rather than purely one-off, vaccine targets.
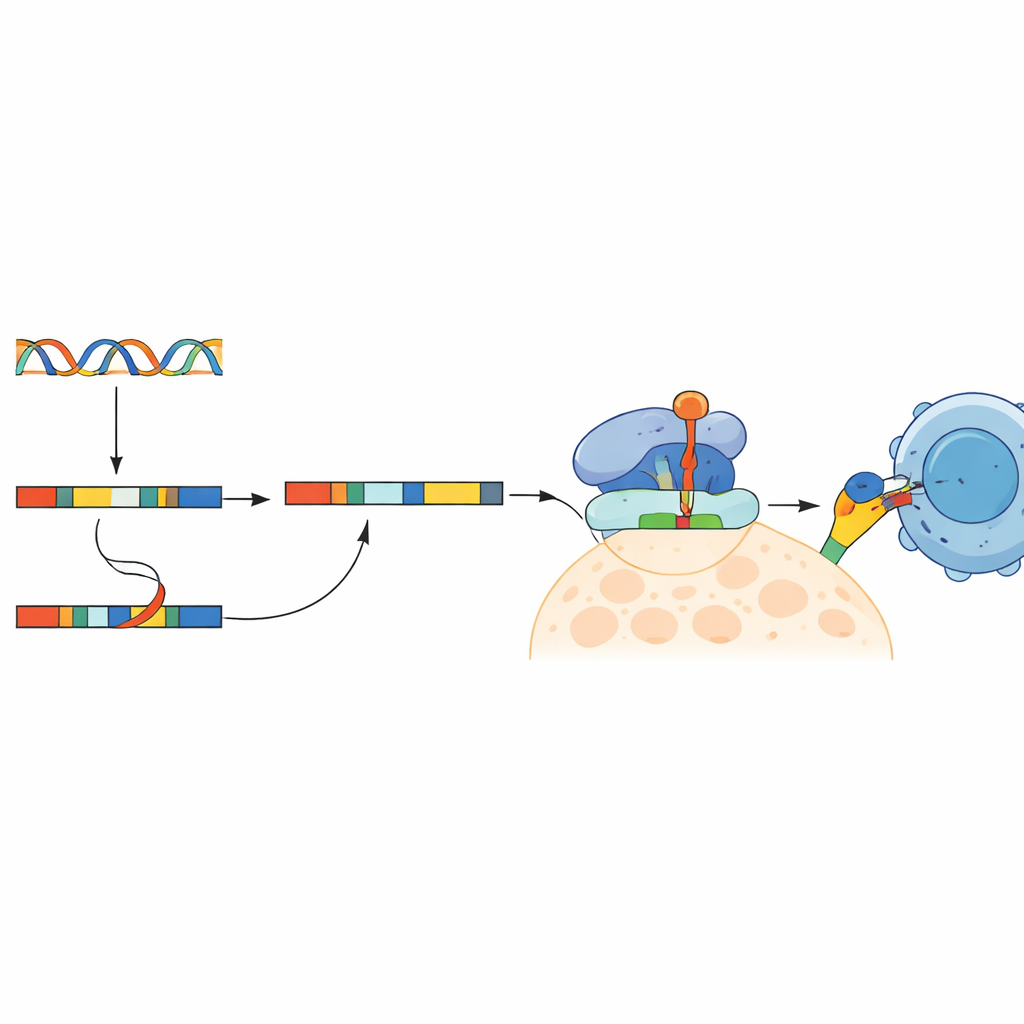
New Paths Toward Broader Cancer Vaccines
This work shows that intron retention—a once-overlooked quirk of RNA processing—can generate real, immune-visible targets on colorectal cancer cells. By proving that these intron-based neoantigens are present, sometimes shared between patients, and capable of activating killer T cells in the lab, the study opens the door to a new class of cancer vaccine ingredients. For patients whose tumors lack many classic DNA mutations, especially those with microsatellite-stable colorectal cancer, tapping into this hidden layer of abnormal protein fragments could greatly expand future options for both personalized and off-the-shelf immunotherapies.
Citation: Manoharan, T., Kee, B.B.R., Cheng, C.Z.M. et al. Identification of immunogenic neoantigens from intron retention in colorectal cancer. Sci Rep 16, 12796 (2026). https://doi.org/10.1038/s41598-026-43687-2
Keywords: colorectal cancer, cancer vaccines, neoantigens, intron retention, immunotherapy