Clear Sky Science · en
Xq28 duplication not F8 inversion: integrated genetic reanalysis redefines prenatal carrier diagnosis
When a Routine Baby Test Sends a Mixed Message
Expectant parents increasingly turn to genetic carrier screening to spot serious inherited diseases before a baby is born. But what happens when a trusted test gives an alarming result that later turns out to be misleading? This study follows one pregnant woman whose screening suggested she might pass on a severe bleeding disorder, only for more advanced tests to reveal a very different—and far less straightforward—change in her DNA. The case shows both the power and the pitfalls of modern genetics, and why confirmatory testing matters for real families making hard decisions.
A Bleeding Disorder That Starts in the Genes
The story centers on hemophilia A, a well-known bleeding disorder that mostly affects boys. It is caused by harmful changes in a gene on the X chromosome called F8, which is needed to make a blood-clotting protein. Many of the most severe cases come from a particular kind of rearrangement in this gene, known as an inversion in a long stretch of DNA called intron 22. Because this inversion is common and technically easy to test, many carrier screening panels for people planning a pregnancy or already pregnant include a specific assay that looks only for this single rearrangement.
When the First Test Points the Wrong Way
In this case, a healthy 27-year-old woman took an expanded carrier screening panel early in pregnancy. A specialized long-distance PCR test suggested that she carried the classic F8 inversion, marking her as a potential carrier of hemophilia A. Understandably worried about her unborn child, she underwent prenatal testing. Initial testing on the fetus appeared to show the same inversion. At first glance, it looked like a straightforward story: mother and daughter both carried a known, high-risk variant for a serious bleeding disease.
A Hidden DNA Repeat Comes to Light
Closer inspection told a more complicated tale. A genome-wide test that looks for gains and losses of DNA segments—called a SNP array—did not simply confirm an inversion. Instead, it revealed that both mother and fetus had an extra copy of a half‑megabase region at the far end of the X chromosome, an area known as distal Xq28 that includes much of the F8 gene along with several brain-related genes. Additional technologies that can see long stretches of DNA in one piece—optical genome mapping and long-read sequencing—showed that this extra copy came from a duplication between two nearly identical DNA blocks flanking the region. Crucially, the F8 gene itself was not flipped, just partly duplicated in a way that left its function intact.
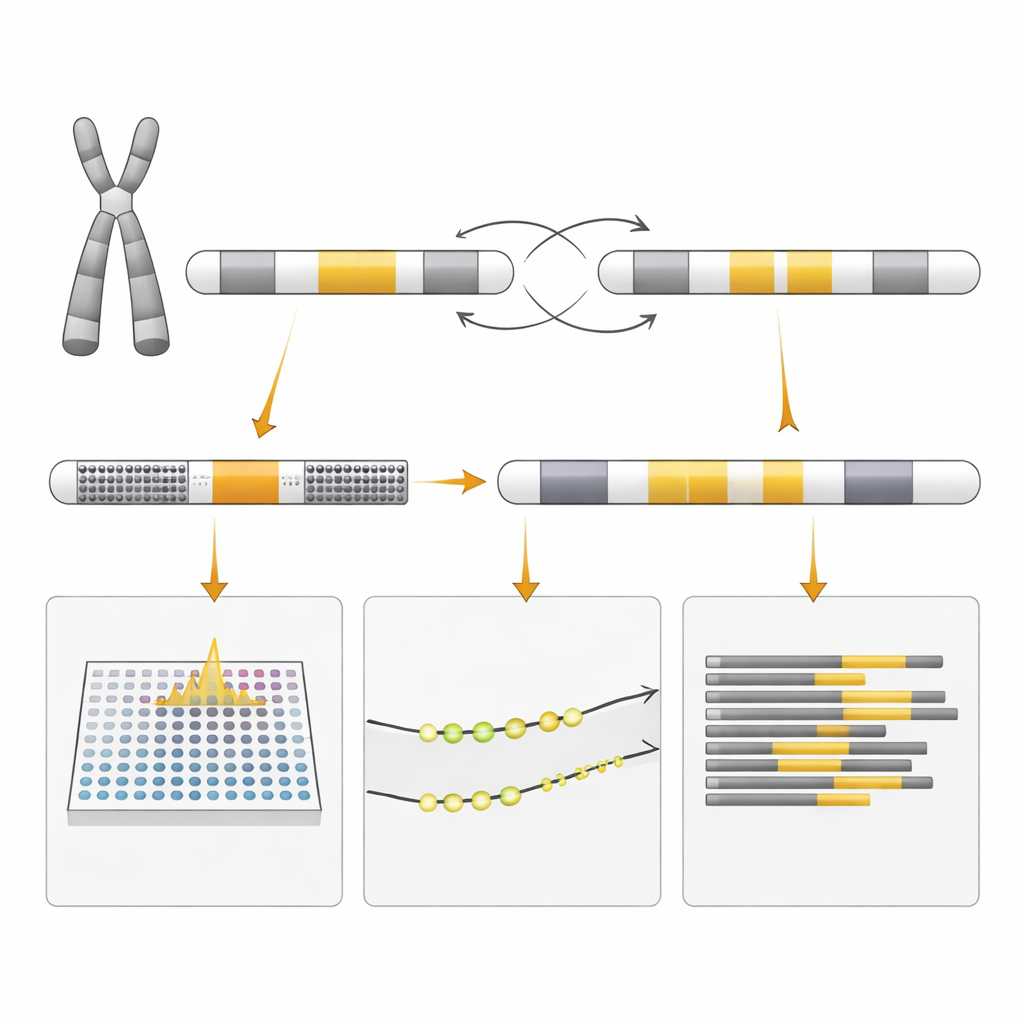
Same Signal, Different Meaning
Why did the standard inversion test appear positive if there was no inversion? The answer lies in how the test is designed. It uses pairs of DNA primers that normally produce one pattern of fragments in people without rearrangements and a different pattern when the classic inversion is present. The duplication created by mispairing between repeated DNA segments generated a mix of fragments that mimicked the inversion pattern, even though the underlying structure was different. Only the more detailed methods could disentangle whether the DNA was actually flipped, copied, or both. The researchers also examined how the two X chromosomes were switched on or off in mother and fetus, a process called X‑chromosome inactivation, to understand why the mother was healthy and to gauge possible effects on the child, though this could not predict the outcome with certainty.
What This Means for Families and Clinics
The key lesson is that even long‑standing, trusted tests can mislead when applied broadly in healthy populations rather than in patients already known to have disease. In this case, the initial assay correctly flagged an unusual rearrangement in the F8 region but could not tell whether it was the classical inversion that causes hemophilia A or a neighboring duplication linked to a different syndrome that often has mild or no effects in females. By layering simple first-line screening with newer technologies that read longer pieces of DNA, the team showed how to sort these possibilities out. They propose a stepwise approach: use the quick, inexpensive inversion test as a first pass, but follow up any positive result with optical genome mapping or long-read sequencing to clarify whether the change is truly dangerous. For families, this means that a worrying screening result should be seen as the beginning of a more careful investigation—not the final word.
Citation: Yang, X., Maimaiti, S., Qi, Q. et al. Xq28 duplication not F8 inversion: integrated genetic reanalysis redefines prenatal carrier diagnosis. Sci Rep 16, 13390 (2026). https://doi.org/10.1038/s41598-026-43654-x
Keywords: hemophilia A, prenatal genetic testing, X chromosome duplication, carrier screening, genome structural variants