Clear Sky Science · en
Innovative fusion models: elevating preoperative gross ETE prediction in thyroid cancer patients
Why this research matters for patients
For many people with thyroid cancer, a key question before surgery is how far the tumor has spread beyond the thyroid gland. This spread, called gross extrathyroidal extension, often dictates whether a person needs a small, targeted operation or a more extensive procedure that can affect voice, swallowing, and long‑term quality of life. The study in this paper explores whether advanced computer analysis of routine ultrasound scans can more accurately predict this kind of spread, helping surgeons tailor treatment while reducing both risk and unnecessary surgery.
Looking beyond what the eye can see
Traditionally, radiologists estimate tumor spread on ultrasound by visually judging how closely a thyroid nodule presses on or distorts surrounding tissues. This approach is subjective and misses many aggressive tumors. The authors propose using computers to measure subtle patterns in and around the tumor that the human eye cannot reliably detect. They focus on both the tumor itself and the narrow rim of tissue around it, reasoning that this “border zone” carries early clues about whether cancer cells are breaking through the thyroid capsule and infiltrating nearby muscles or organs.
Turning ultrasound images into measurable data
The team gathered ultrasound scans and clinical information from 4,542 patients with papillary thyroid carcinoma treated at multiple hospitals over five years. After carefully outlining the tumors, they used software to create thin, concentric rings of tissue around each nodule, mimicking distances from the tumor edge. From these regions they extracted hundreds of numerical image features describing size, brightness, and texture. In parallel, they used deep learning—neural networks originally trained on natural images—to automatically discover complex image patterns related to tumor behavior. By systematically varying how far these peritumoral rings extended, they could test how much information the surrounding tissue truly added.
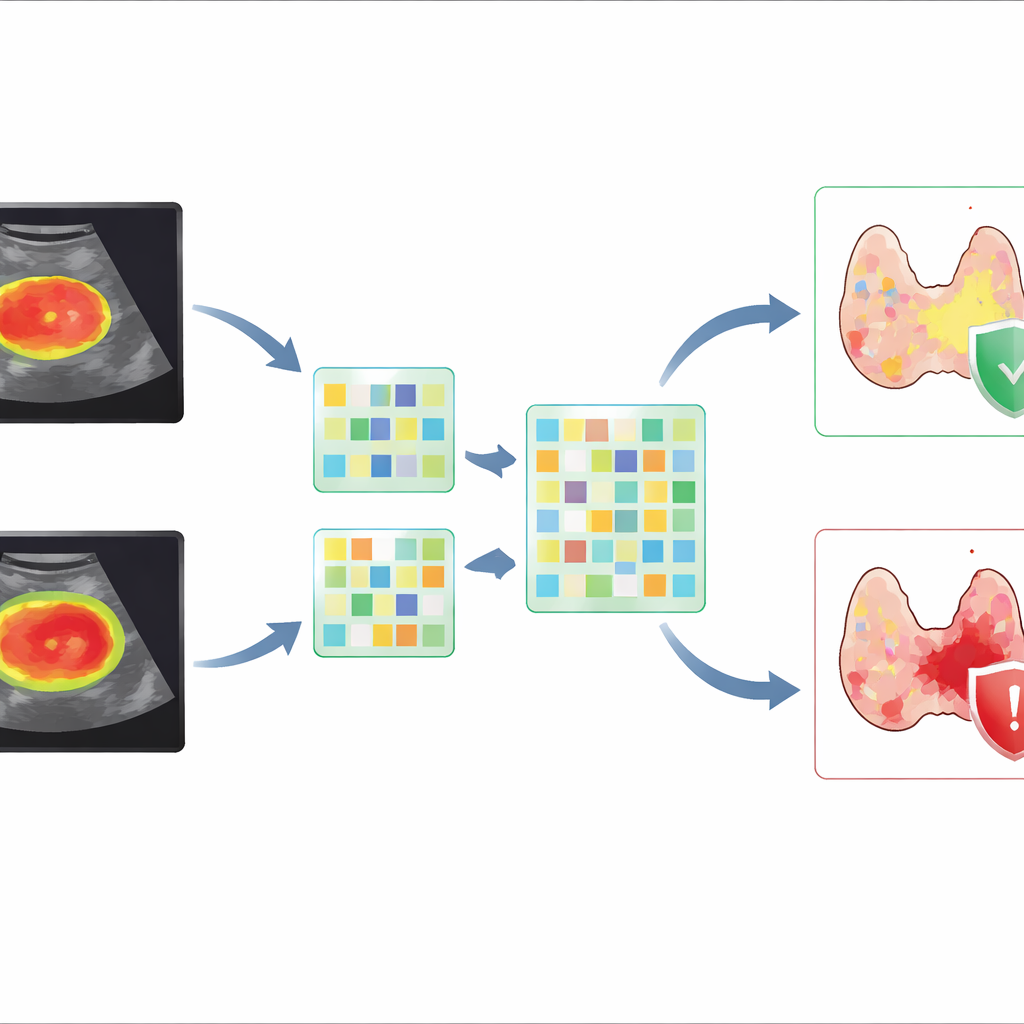
Fusing human‑designed features with deep learning
Instead of relying on a single method, the researchers created “fusion” models that combined two types of information: handcrafted radiomics features and deep learning features. They found that including a modest band of tissue around the tumor—roughly corresponding to a 15‑pixel expansion in the ultrasound image—consistently improved the ability to distinguish tumors that had broken out of the thyroid. The best deep learning model alone performed well, but when its features were fused with radiomics from the tumor plus its immediate surroundings and then combined with key clinical factors such as tumor size and margin appearance, prediction improved further. This integrated model achieved a high accuracy and strong ability to correctly rule out cases without dangerous spread, which is crucial for avoiding overly aggressive surgery.
Putting advanced models into a single simple tool
To make these complex models usable in practice, the authors built a visual scoring chart known as a nomogram. Clinicians can read this chart by locating a patient’s tumor size, margin characteristics, and the model‑derived image score, then summing the points to estimate the probability of gross extrathyroidal extension. The team validated this tool on an independent group of patients from different centers and ultrasound machines, showing that its performance held up across varied real‑world conditions. They also used visualization methods to highlight which image regions and features drove the model’s predictions, helping doctors see that the computer was focusing on clinically meaningful details at the tumor’s edge.
What this could mean for future thyroid surgery
In plain terms, this study shows that carefully analyzing both the cancer and its immediate surroundings on standard ultrasound images can help predict which thyroid tumors are likely to have pushed beyond the gland. By blending traditional image measurements, powerful deep learning, and basic clinical information into a single, easy‑to‑use chart, the authors offer a noninvasive way to better plan surgery and stratify risk. While the method still misses some deeply invading tumors and needs further prospective testing, it points toward a future in which routine scans are paired with intelligent software to guide more precise, individualized care for thyroid cancer patients.
Citation: Pan, T., Wu, F., Cai, J. et al. Innovative fusion models: elevating preoperative gross ETE prediction in thyroid cancer patients. Sci Rep 16, 13070 (2026). https://doi.org/10.1038/s41598-026-43230-3
Keywords: thyroid cancer, ultrasound imaging, deep learning, radiomics, surgical planning