Clear Sky Science · en
Causal relationship between autoimmune diseases and iron deficiency anemia: a two-sample mendelian randomization study
Why this matters for everyday health
Iron deficiency anemia is one of the most common causes of tiredness and poor health worldwide, yet doctors do not always know why a particular person becomes anemic. This study asks a simple but important question: do certain autoimmune diseases actually cause a higher risk of iron deficiency anemia, rather than just happening to occur in the same people? By using large genetic datasets instead of traditional clinical trials, the authors try to untangle cause and effect in a way that could eventually change how high‑risk patients are monitored and treated.
Hidden links between immunity and low iron
Autoimmune diseases arise when the body’s defense system mistakenly attacks its own tissues, leading to long‑lasting inflammation in organs such as joints, intestines, kidneys, and connective tissue. Separately, iron deficiency anemia develops when the body does not have enough usable iron to make healthy red blood cells, leaving people fatigued, short of breath, and less able to concentrate. Observational studies have long noted that people with conditions like rheumatoid arthritis or inflammatory bowel disease often also have anemia, but these snapshots in time cannot show whether the autoimmune disease truly increases the risk of iron deficiency, or whether other shared factors—such as diet, medications, or blood loss—are to blame.
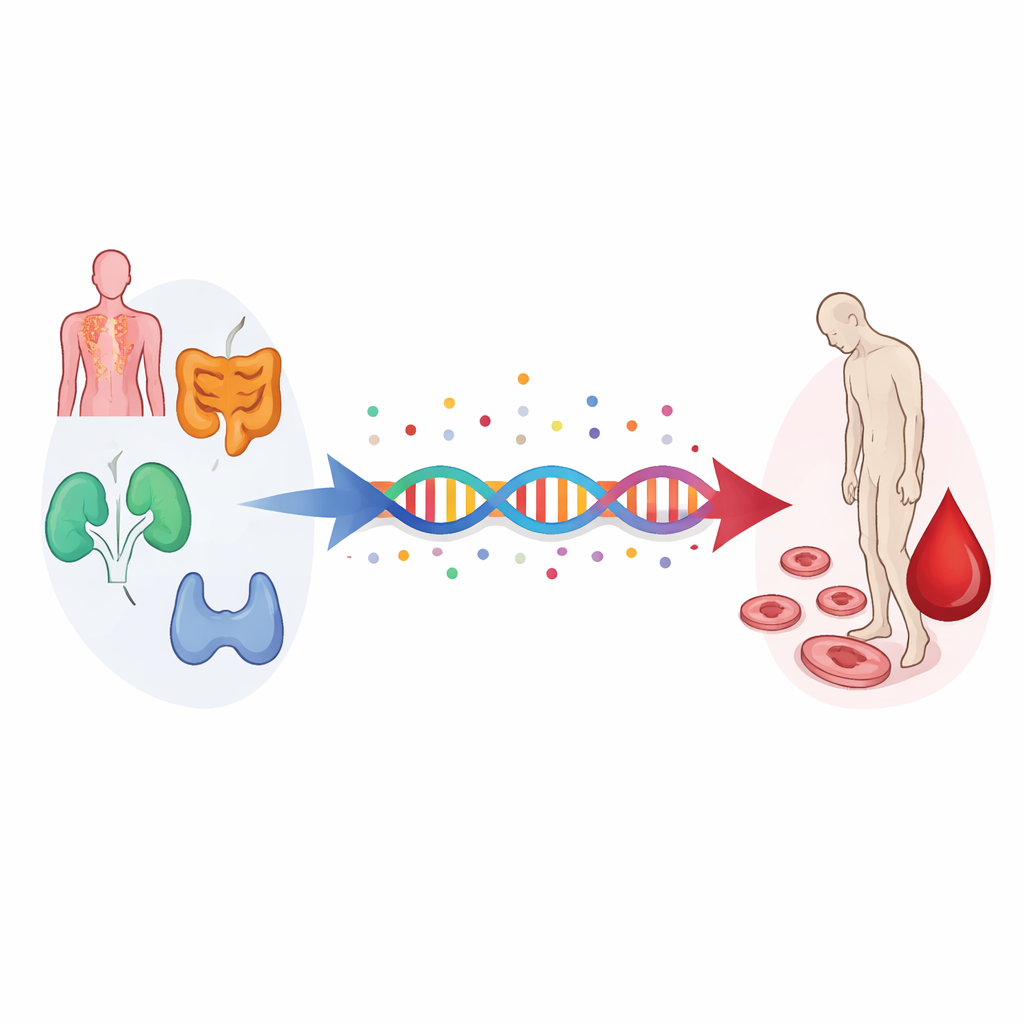
Using genes as a natural experiment
The researchers turned to a method called Mendelian randomization, which treats naturally occurring genetic differences as a kind of lifelong “assignment” to higher or lower risk of a given disease. Because genes are set at conception and are not changed by lifestyle, they can help separate cause from coincidence. The team gathered summary data from very large genetic studies of eight autoimmune diseases—rheumatoid arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, celiac disease, systemic lupus erythematosus, ankylosing spondylitis, and membranous nephropathy—all in people of European ancestry. They then compared these with genetic data on more than 15,000 people with iron deficiency anemia and almost 400,000 without it from the Finnish FinnGen project.
What the genetic evidence shows
Using several layers of quality checks, the authors selected genetic variants strongly linked to each autoimmune disease, while trying to avoid variants that might influence anemia through other routes. Their main statistical approach, supported by multiple sensitivity tests, found that people who were genetically predisposed to rheumatoid arthritis, inflammatory bowel disease, and ulcerative colitis had a small but clear increase in risk of iron deficiency anemia. Signals for celiac disease, ankylosing spondylitis, Crohn’s disease, systemic lupus, and membranous nephropathy were weaker: some appeared linked to anemia at first, but the strength of these links dropped or disappeared after the researchers removed variants from a complex immune‑related region of the genome that can distort such analyses.
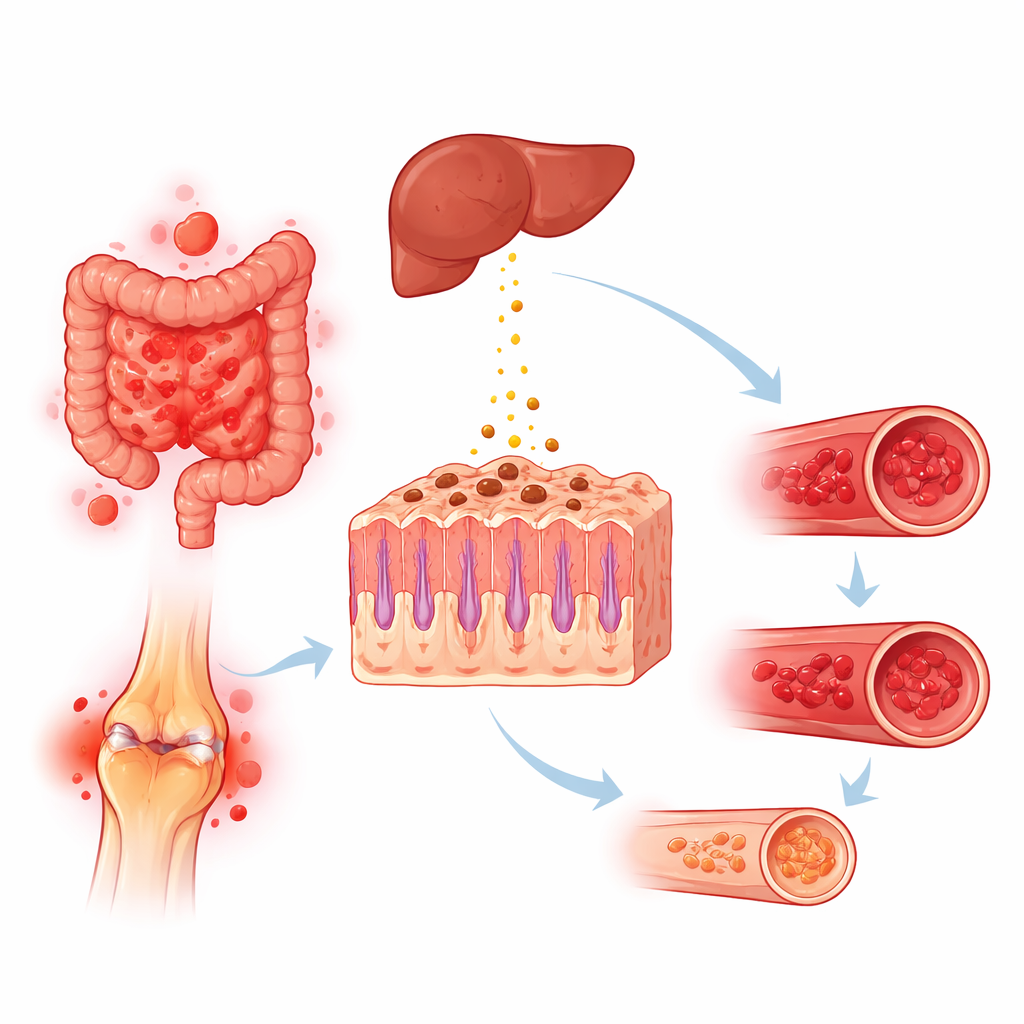
How autoimmune activity may drain iron
Several biological pathways could explain why autoimmune diseases, especially those involving the gut and chronic inflammation, push people toward iron deficiency. Intestinal disorders can cause slow internal bleeding, poor absorption of nutrients, or damage to the upper small intestine where most iron uptake occurs. Long‑term use of pain‑relieving drugs in joint disease can also increase bleeding from the stomach and intestines. At the same time, inflammatory signals common in autoimmunity stimulate the liver to release hepcidin, a hormone that locks iron inside storage cells and reduces its passage from the gut into the bloodstream. The result can be either absolute iron loss, functional blockage of iron use, or both, producing anemia even when overall body iron stores are not completely depleted.
What this means going forward
For a person living with rheumatoid arthritis or inflammatory bowel disease, the study suggests that their condition does more than merely coexist with anemia—it likely helps cause it, even if the increase in risk for each individual is modest. Because both autoimmune diseases and iron deficiency anemia are common, a small shift in risk can translate into many additional cases at the population level. Clinicians may therefore want to monitor iron status more proactively in these patients and address subtle drops before they lead to severe symptoms. At the same time, the uncertain findings for other autoimmune diseases highlight that not every statistical signal is clinically large or fully understood; more work in diverse populations and across age and sex groups will be needed to clarify who is most vulnerable and why.
Citation: Chen, W., Wang, Y., Long, H. et al. Causal relationship between autoimmune diseases and iron deficiency anemia: a two-sample mendelian randomization study. Sci Rep 16, 12935 (2026). https://doi.org/10.1038/s41598-026-42356-8
Keywords: autoimmune disease, iron deficiency anemia, inflammatory bowel disease, rheumatoid arthritis, genetic epidemiology