Clear Sky Science · en
A fractional-order vaccination model to analyze the dynamics of Mpox Clade I and II with real data
Why this matters for everyday health
Mpox, once a rare virus seen mostly in parts of Africa, has recently made global headlines. Two distinct forms, or clades, now circulate: one more severe, one milder but widespread. This study asks a practical question with big public-health consequences: given limited vaccines and complex transmission between people and animals, can we use mathematics to understand how mpox spreads and how vaccination might finally bring it under control?
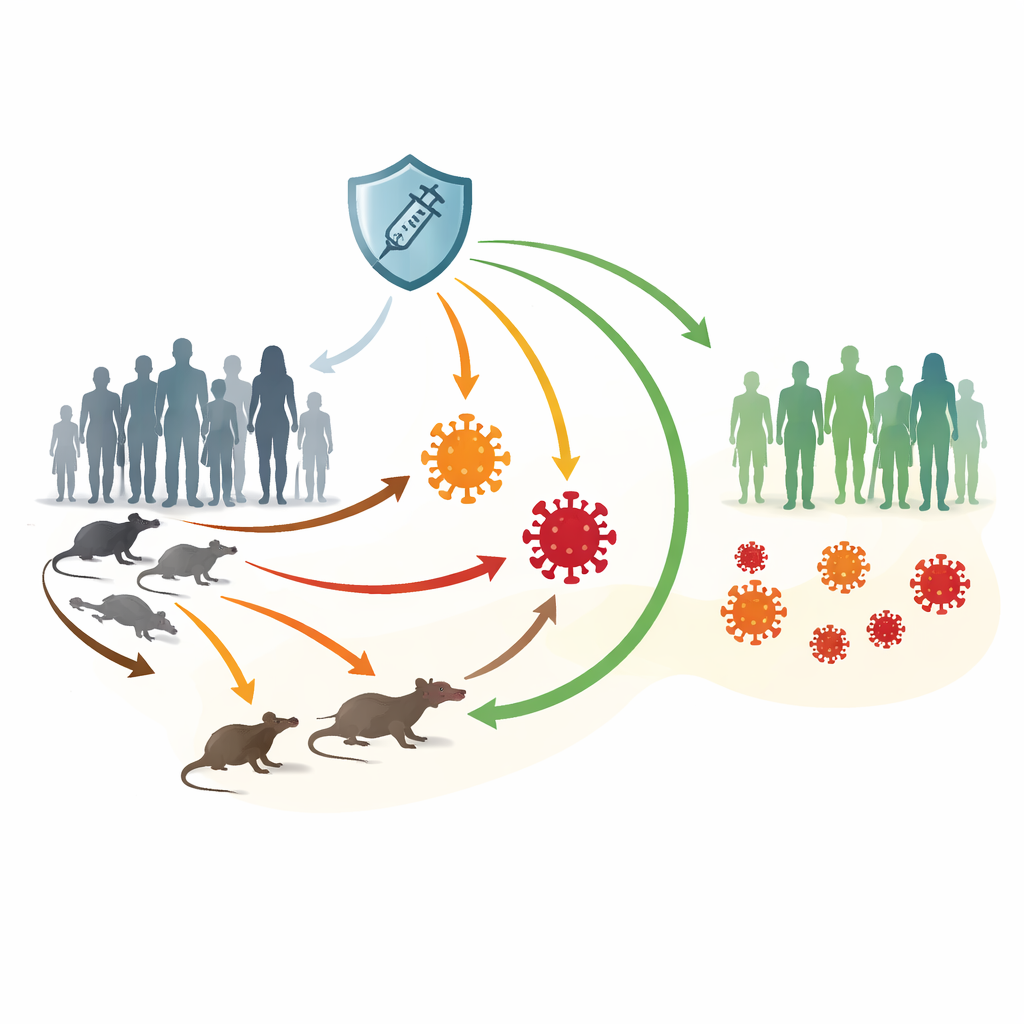
Two kinds of virus and many paths of spread
The researchers focus on Clade I and Clade II of the mpox virus. Clade I, still common in some African countries, causes more severe illness and higher death rates. Clade II, responsible for the 2022 global outbreak, is usually milder but has spread widely through close and intimate contact, especially among specific at-risk communities. Mpox can jump between humans and rodents and circulate quietly in animal populations, making it harder to eliminate. Vaccines such as JYNNEOS offer strong protection, but supplies are uneven worldwide and immunity can fade over time.
Turning mpox into a flow of people and animals
To untangle this complexity, the authors build a compartment model that tracks how individuals move between health states. People start out as susceptible, can receive a vaccine that offers temporary but strong protection, become exposed after contact with infection, then progress to being sick with either Clade I or Clade II. Some require hospital care, while others recover directly. At the same time, rodents are divided into healthy and infected groups. Arrows in the model represent all major routes of spread: human-to-human, animal-to-human, human-to-animal, and animal-to-animal. This framework lets the team test how changes in contact, vaccination, and loss of immunity ripple through both human and animal communities.
Adding memory to the math
Instead of using only standard equations, which assume infections and recoveries follow simple, uniform timelines, the authors use a fractional-order approach. In plain terms, this adds a built-in "memory" to the model: past events continue to influence current infection levels in a gradual way. That better reflects real outbreaks, where incubation times, recovery, and behavior vary widely between individuals. The team first writes a traditional version of the model, then extends it to the fractional framework and proves that its solutions stay biologically realistic (no negative populations, no explosions to infinity) and are mathematically well behaved and unique.
What the model says about spread and control
Using U.S. data on Clade II mpox cases from January to July 2025, the researchers fit their model and estimate the basic reproduction number—the average number of new infections caused by one infected person in a fully susceptible population. They find a value of about 1.33, meaning each case produces more than one new case and the virus can persist. They also derive a "vaccination reproduction number" that accounts for immunization. If this value falls below one, the infection-free state becomes stable in theory. However, the model also reveals a phenomenon called backward bifurcation: even if the reproduction number is pushed slightly below one, mpox can still linger at low but steady levels, especially when multiple transmission routes and waning immunity are present.
Which levers matter most
To see which factors most strongly influence spread, the team varies parameters such as contact rates and animal death rates across realistic ranges and measures how the reproduction number responds. In this analysis, frequent contact between infected and susceptible rodents, and between rodents and humans, strongly increases transmission, while faster natural turnover in rodent populations helps suppress it. Human-to-human contact in both clades, and contacts between infected rodents and people, also play a major role. By contrast, some clinical details, like exact hospitalization and recovery rates, matter less for the overall ability of the virus to persist.
What this means for mpox control
The study concludes that vaccination can substantially reduce mpox cases and hospitalizations, particularly when coverage is high and immunity is maintained, but it is not a magic off switch. Because the virus can hide in animals and because of backward bifurcation, small improvements may not be enough: strong, sustained efforts are needed. In everyday terms, that means combining widespread vaccination in high-risk groups with measures that reduce close-contact transmission and address animal reservoirs where feasible. The work also shows that realistic, memory-aware models can better match real case data and help health agencies plan strategies that keep mpox from re-emerging, rather than simply chasing the next outbreak.
Citation: Khan, M.A., DarAssi, M.H., Tasqeeruddin, S. et al. A fractional-order vaccination model to analyze the dynamics of Mpox Clade I and II with real data. Sci Rep 16, 11093 (2026). https://doi.org/10.1038/s41598-026-41453-y
Keywords: mpox, vaccination, mathematical modeling, zoonotic disease, fractional calculus