Clear Sky Science · en
Immunophenotypic skewing of B cells toward IgD⁻CD27⁻IgG⁺ subtype and metabolic attenuation in colorectal cancer
Why the body’s own defenders can falter in colon cancer
Colorectal cancer is not just a mass of rogue cells; it is a complex community where tumor cells and immune cells constantly interact. Among these immune cells are B cells, better known for making antibodies and helping us fight infections. This study explores what happens to B cells when they enter the environment of a colon tumor, revealing that many of them become both functionally and energetically weakened. Understanding this hidden side of the immune response could eventually help doctors design treatments that coax these cells back into the fight against cancer.
Uncovering a strange B cell shift inside tumors
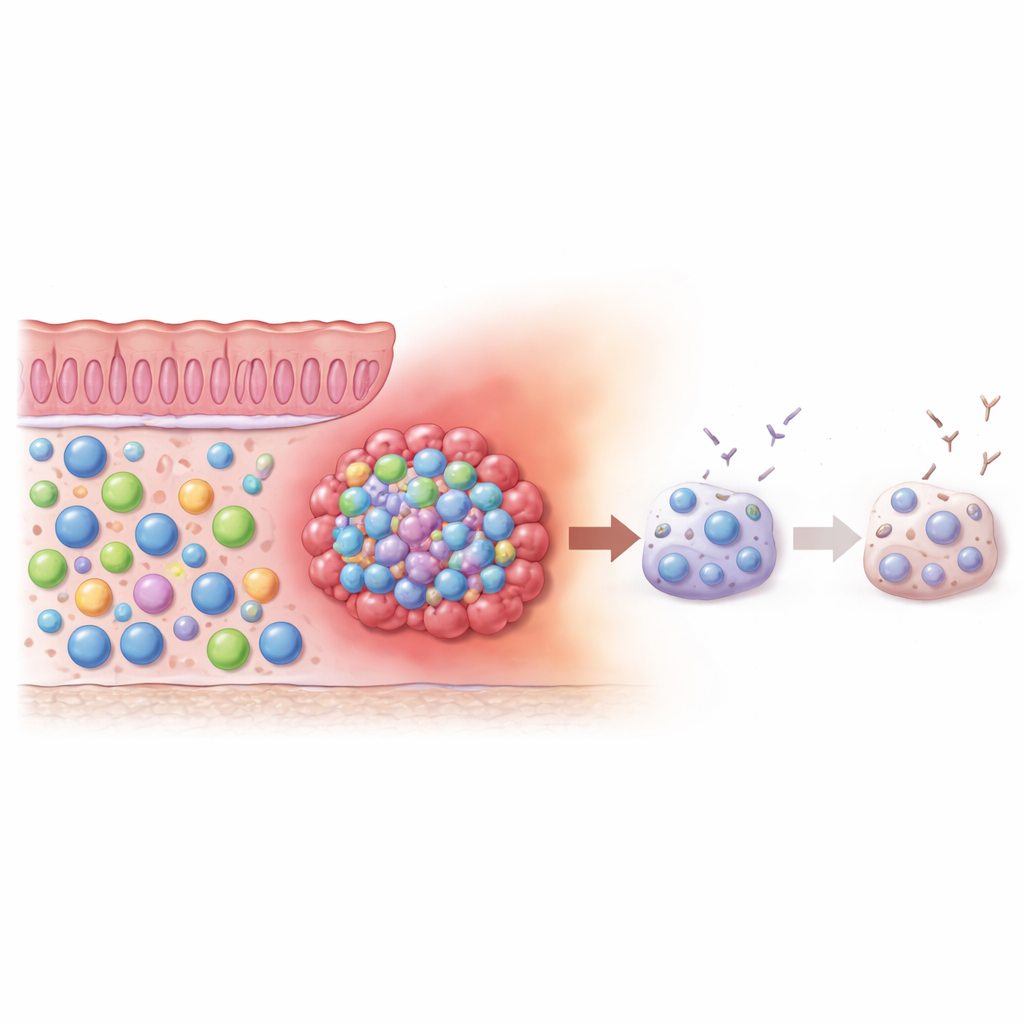
The researchers began by comparing B cells found in colorectal cancer tissue with those in nearby normal intestine from the same patients. In healthy gut tissue, most B cells were in active, antibody-producing stages. Inside tumors, however, a very different pattern emerged: an unusual subtype called “double-negative” B cells (lacking two typical surface markers) became the dominant group. Normally rare, these cells are known to expand during chronic infections and some autoimmune diseases, where they often show signs of being worn out or exhausted. In the tumor samples, these double-negative B cells also displayed low levels of a surface protein associated with healthy function, reinforcing the idea that they were in a fatigued state.
From protective antibodies to a different response
In the healthy intestine, B cells usually produce IgA antibodies, which help protect the gut lining and keep peace with the resident microbiota. In the tumor setting, the authors found that the dominant B cell subsets shifted away from IgA and toward IgG antibodies. This altered antibody balance suggests that the local environment nudges B cells away from their normal barrier-protective role and toward a more inflammation-linked response. Yet, when the team stimulated B cells taken from the tumor with signals that normally drive them to become full-fledged antibody factories, these cells responded poorly compared with B cells from healthy tissue. Together, these observations point to a sizable pool of B cells in colorectal tumors that have been redirected and then partly disabled.
Silent engines: low-activity B cells in the tumor
Because cell behavior is tightly linked to how cells use energy, the researchers next examined B cell metabolism inside tumors. They measured protein synthesis and mitochondrial activity, common readouts of how “switched on” a cell is. Surprisingly, the double-negative B cells in tumors looked metabolically quiet, resembling resting B cells rather than highly active defenders. This low-gear profile offers a plausible explanation for why they struggle to mature into robust antibody-secreting cells. Instead of fueling a vigorous immune attack, their internal engines appear tuned down, suggesting that the tumor environment may be pushing them into a sluggish, less effective state.
How tumor mini-organs rewire B cells’ fuel use
To probe how tumor tissue creates this effect, the team turned to mouse models. They grew three-dimensional “organoids” from normal intestine and from chemically induced colon tumors, then co-cultured these structures with healthy mouse B cells. B cells exposed to tumor organoids divided less and produced fewer antibody-secreting cells than those grown alone or with healthy organoids. When the researchers profiled key metabolic pathways, they found that B cells near tumor organoids gradually lost their ability to take up glucose efficiently and showed reduced glycolysis, the rapid sugar-burning route that often powers immune responses. At the same time, the cells became more reliant on their mitochondria, yet their mitochondrial machinery declined over time, with lowered membrane potential, reduced oxygen consumption, and diminished capacity to produce energy.
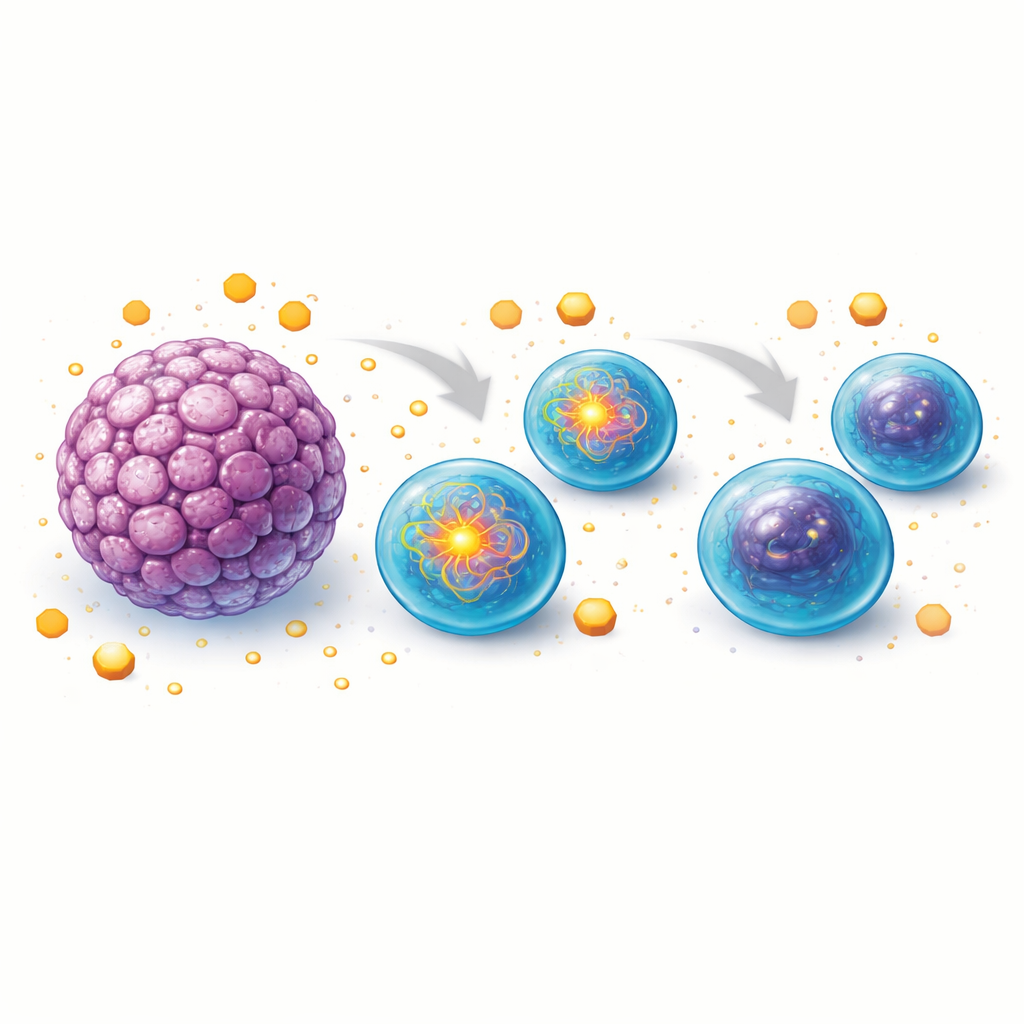
Broken fuel lines and tired immune soldiers
Further analysis showed that B cells co-cultured with tumor organoids also took up fewer fatty acids and produced lower levels of enzymes needed to use fats as fuel. This meant that two major energy sources—sugars and fats—were both compromised. As a result, while the B cells appeared increasingly dependent on mitochondria, they lacked the necessary fuel and functional components to keep mitochondrial energy production running at high speed. The picture that emerges is of B cells initially trying to ramp up their activity in the tumor environment, only to stall as nutrient competition and tumor-derived factors erode their metabolic capacity.
What this means for future cancer treatments
Put simply, this study shows that many B cells inside colorectal tumors are pushed into an exhausted, low-energy mode. They change the types of antibodies they favor, lose their ability to fully mature, and have their fuel systems progressively sabotaged by the tumor surroundings. For non-specialists, the key takeaway is that cancer can disarm parts of the immune system not only through direct suppression, but also by cutting off or misdirecting their energy supply. By mapping these changes, the work opens the door to potential therapies that restore or rewire B cell metabolism, with the goal of turning these tired cells back into active allies in the body’s fight against colorectal cancer.
Citation: Martinis, E., Tonon, S., Valeri, V. et al. Immunophenotypic skewing of B cells toward IgD⁻CD27⁻IgG⁺ subtype and metabolic attenuation in colorectal cancer. Sci Rep 16, 11403 (2026). https://doi.org/10.1038/s41598-026-41446-x
Keywords: colorectal cancer, B cells, tumor microenvironment, immune metabolism, cancer immunotherapy