Clear Sky Science · en
MALDI-TOF mass spectrometry imaging of sulfatide lipid expression in the CNS of mice with experimental autoimmune encephalomyelitis
Why This Matters for Brain Health
Multiple sclerosis (MS) is a disease in which the body’s own immune system attacks the insulation around nerve fibers in the brain and spinal cord. That insulation, called myelin, is unusually rich in fats known as lipids. This study asks a deceptively simple question with big implications: do subtle changes in these myelin lipids show up before obvious symptoms and visible damage appear? By using a specialized form of mass spectrometry to “map” lipids directly in mouse brain tissue, the researchers show that specific myelin fats shift early and broadly during experimental MS, hinting at new ways to detect and perhaps treat the disease.
A Closer Look at Myelin’s Fatty Armor
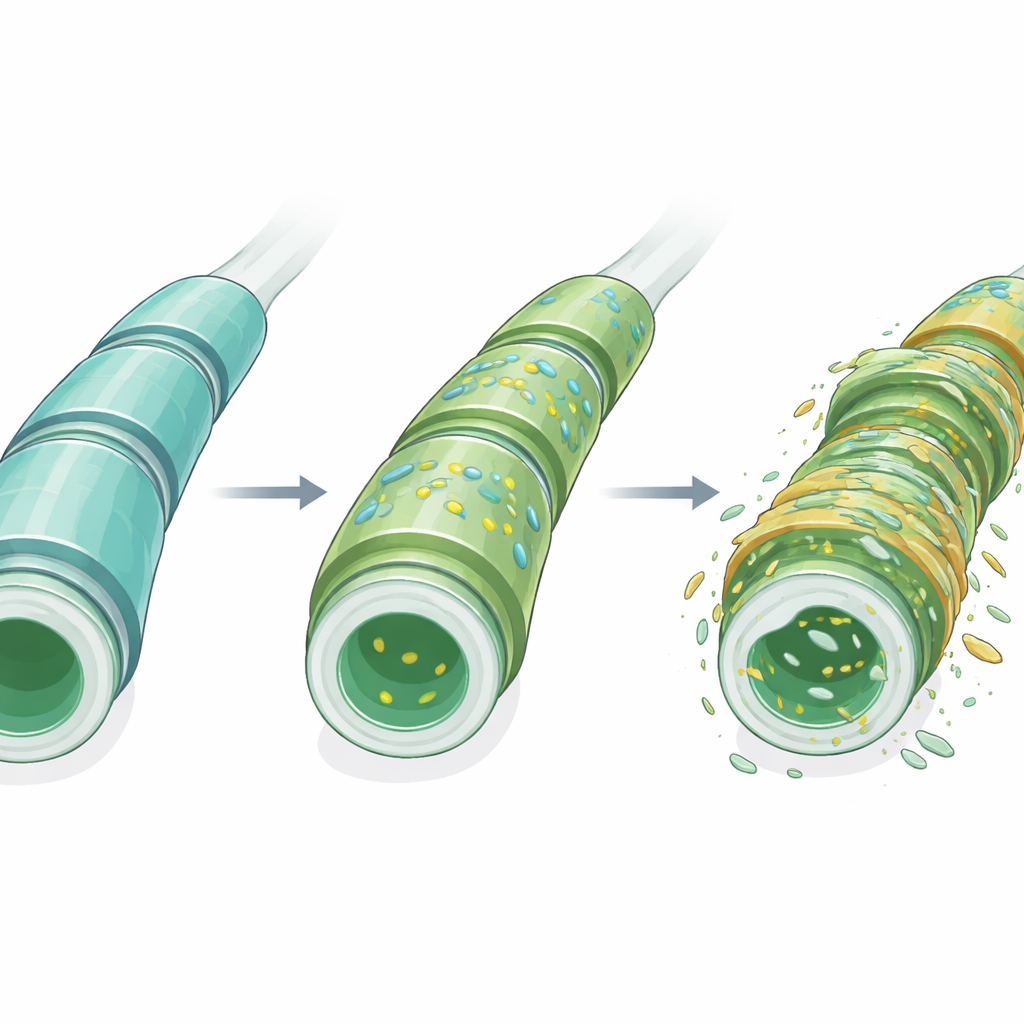
Myelin is a multilayered wrapping that surrounds many nerve fibers and is essential for fast electrical signaling and long-term nerve health. Unlike most tissues, myelin is dominated by lipids rather than proteins. Among its most distinctive components are sulfatides, a family of specialized fats found at particularly high levels in myelin compared with other parts of the body. These molecules help organize the myelin layers, support the cells that make myelin, and participate in communication between nerve fibers and their glial support cells. Disruptions in sulfatide production or breakdown have been linked to several brain disorders, including Alzheimer’s disease and rare inherited myelin diseases, making them a prime suspect in MS as well.
Seeing Molecules in Place, Not in a Test Tube
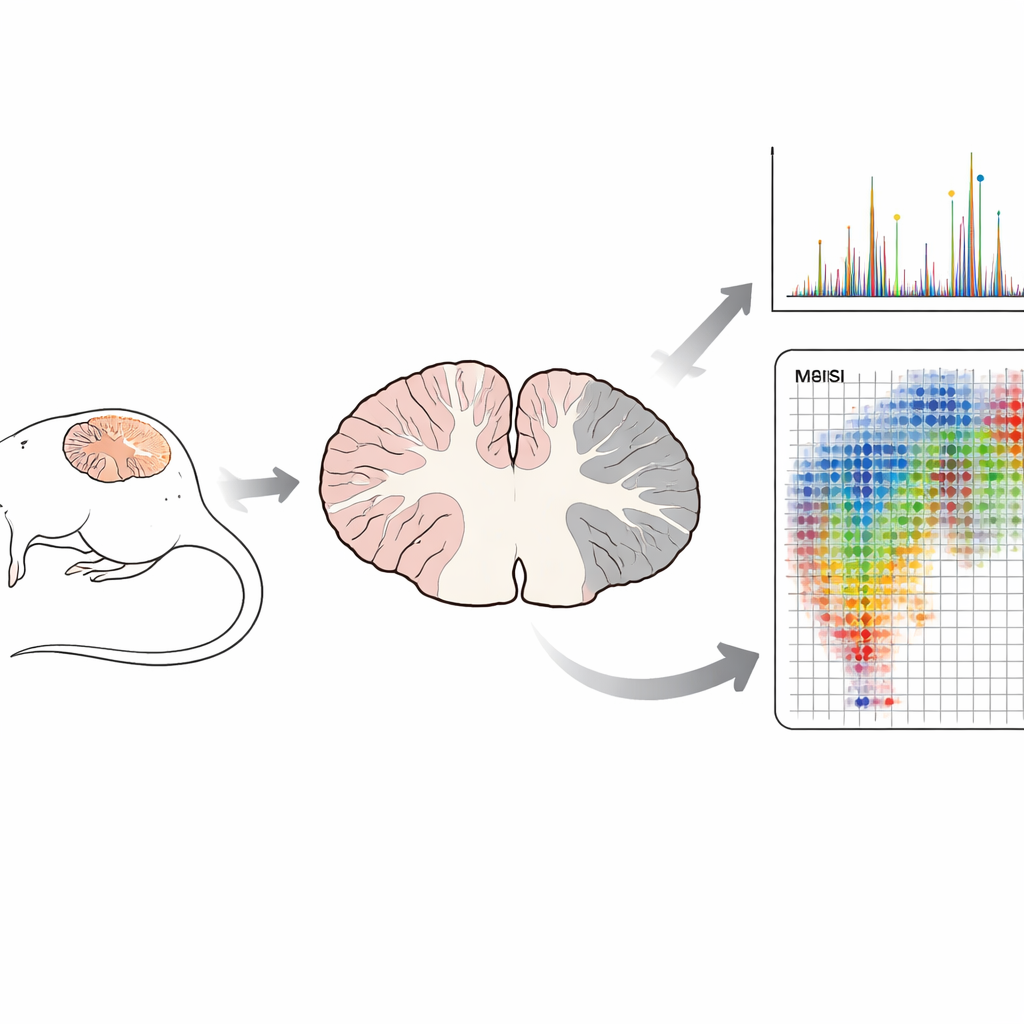
Traditional biochemical methods often grind tissue into a uniform mixture before measuring its contents. That approach can reveal what is present, but it destroys the spatial context of where each molecule lives. In this study, the team used matrix-assisted laser desorption/ionization time-of-flight mass spectrometry imaging (MALDI-TOF MSI), a technique that scans thin brain slices point by point. At each point, a laser releases molecules, and the instrument records their masses, building “molecular maps” of hundreds of compounds at once. Crucially, this is done without labels or dyes and preserves the fine structure of the tissue, which can later be compared directly with standard microscope stains.
How Myelin Lipids Shift During Experimental MS
The researchers turned to a well-established mouse model of MS called experimental autoimmune encephalomyelitis (EAE), which reproduces inflammation, myelin loss, and nerve damage similar to that seen in patients. They examined the cerebellum, a brain region where EAE damage often appears, at four stages: healthy, an early “pre-onset” phase without obvious symptoms, disease onset, and peak paralysis. Focusing on a set of known myelin sulfatides, they first confirmed that, in healthy mice, these lipids are concentrated in the white matter tracts, the myelin-rich highways of nerve fibers. Using mass spectrometry fragmentation patterns, they distinguished sulfatides from other lipids with nearly identical masses and confirmed the identity of nine out of ten target molecules.
Early and Widespread Changes Before Symptoms
When they compared lipid maps across disease stages, a clear pattern emerged. Several sulfatide species increased in abundance in the white matter before any outward signs of EAE appeared, while at least one important sulfatide decreased. Principal component analysis—a way to summarize complex data into a few key patterns—showed that the overall sulfatide profile shifted markedly, especially around disease onset, and then partially returned toward the earlier pattern at peak disease. Importantly, these changes were not confined to the focal inflammatory spots typically highlighted in MS pathology; instead, they extended across the broader white matter. In addition, the untargeted nature of MSI revealed dozens of other lipids in both white and gray matter whose distributions changed with disease stage, suggesting that a wider network of lipid metabolism is involved.
What This Could Mean for Diagnosis and Treatment
By showing that myelin sulfatides and other lipids change their levels and locations early and broadly during experimental MS, this work highlights lipid profiles as a potential early warning system for damage to nerve insulation. The ability to visualize these molecules in place, rather than after tissue is homogenized, offers a more precise picture of how neuroinflammation unfolds. In the future, such molecular patterns might guide the development of imaging agents for use with clinical MRI or point to new drug targets aimed at stabilizing myelin lipids. While more work is needed to translate these findings from mice to humans, the study demonstrates that mass spectrometry imaging can capture a “molecular snapshot” of the diseased brain that may help predict, monitor, and ultimately modify the course of MS.
Citation: Berlin, K.A., Huizar, C.C., Garza, C. et al. MALDI-TOF mass spectrometry imaging of sulfatide lipid expression in the CNS of mice with experimental autoimmune encephalomyelitis. Sci Rep 16, 11462 (2026). https://doi.org/10.1038/s41598-026-41147-5
Keywords: multiple sclerosis, myelin lipids, mass spectrometry imaging, neuroinflammation, sulfatides