Clear Sky Science · en
New indole-linked 1,2,4-triazole derivatives as dual FAK inhibitors and apoptosis inducers targeting survival and migration in triple-negative breast cancer in-vitro
Why this research matters for patients
Triple-negative breast cancer is one of the hardest forms of breast cancer to treat because it lacks the hormone receptors that many modern drugs target. Patients often face aggressive disease, limited options, and harsh chemotherapy. This study explores a different tactic: a new family of small molecules designed to shut down a key survival switch inside cancer cells and push them into self-destruction, while sparing healthy cells as much as possible.
A new way to hit an aggressive cancer
The researchers focused on triple-negative breast cancer, which tends to spread early and resist standard therapies. These tumors frequently rely on a protein called focal adhesion kinase, or FAK, which helps cancer cells cling, move, and survive stressful conditions. When FAK is overly active, tumors become more invasive and harder to kill. Because there are still no widely used targeted drugs for this cancer type, blocking FAK has become an attractive strategy to slow tumor growth and stop it from spreading.
Designing tailor‑made cancer blockers
To tackle FAK, the team synthesized a set of new molecules built from two building blocks that chemists know well: indoles and 1,2,4‑triazoles. By linking and arranging these pieces in different ways, they produced several related compounds and then tested them against two breast cancer cell lines and normal human fibroblast cells. Three candidates, labeled 3c, 4c, and 5c, stood out for their ability to slow cancer cell growth at relatively low doses while being less harmful to normal cells. Among them, compound 4c, which contains two indole rings connected through a triazole core, emerged as the most promising. 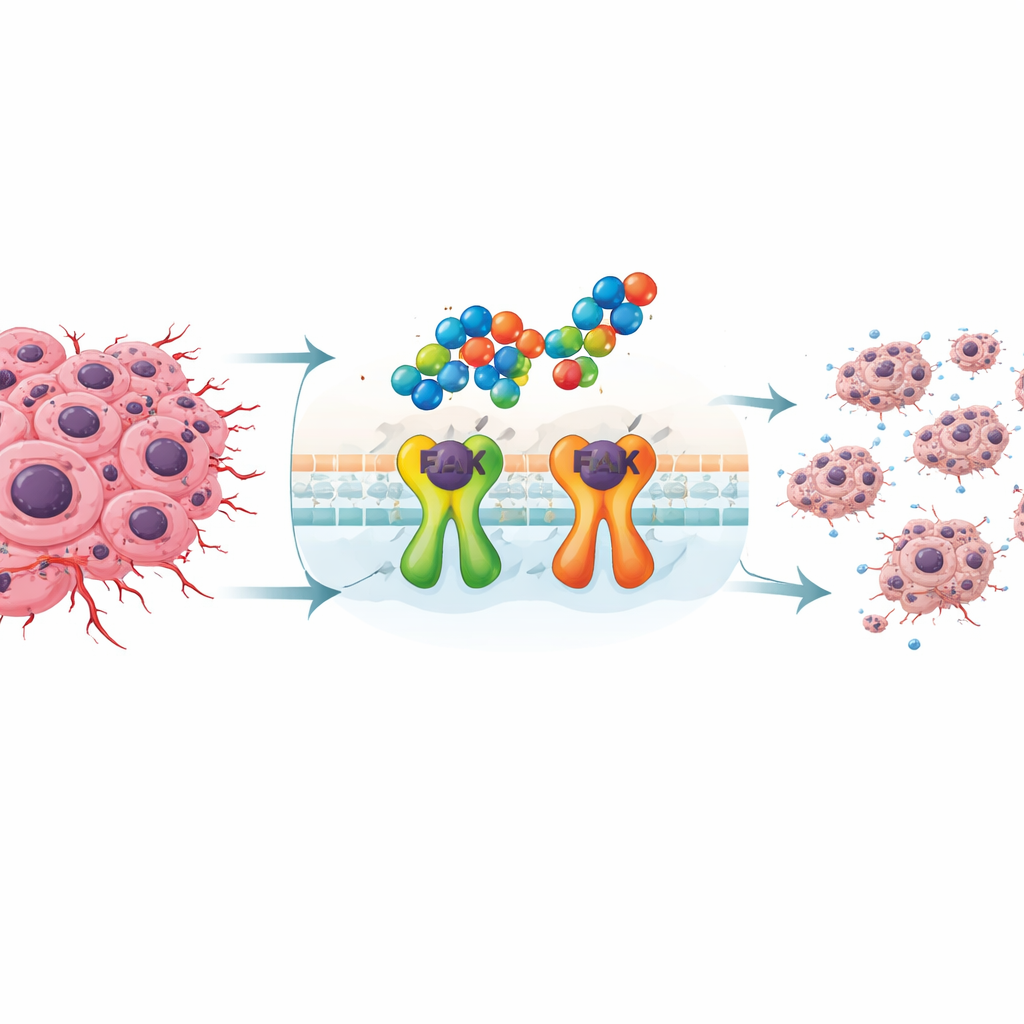
Slowing growth, blocking movement, and triggering cell suicide
Laboratory tests showed that these new compounds do more than just slow cancer cell division. In scratch “wound” experiments, where a gap is created in a sheet of cells and the researchers watch how quickly cells crawl to close it, compounds 3c, 4c, and 5c strongly delayed movement in both breast cancer lines. Compound 4c also forced cancer cells to pause at specific checkpoints in the cell cycle: it stalled one breast cancer line (MCF‑7) in the DNA‑copying phase and another triple‑negative line (MDA‑MB‑231) before division, limiting their ability to multiply. When the team looked at markers of programmed cell death, they found that 4c was especially potent in triple‑negative cells, driving nearly 91% of them into apoptosis with very little messy necrosis, a pattern that is generally more desirable for therapy.
Striking at the cancer’s survival switch
To understand how these molecules exert their effects, the researchers measured changes in key genes and proteins. In triple‑negative cells, 4c sharply reduced the activity of the gene that encodes FAK and lowered FAK protein levels by about 61%, almost matching a known FAK‑blocking drug used for comparison. At the same time, 4c decreased signals that protect cells from death, such as BCL2 and the chemokine CCL5, and boosted pro‑death signals like caspase‑3. Because FAK and CCL5 are also linked to how cancer cells migrate and evade the immune system, their suppression helps explain the strong anti‑migration and pro‑apoptotic effects seen in the lab. Computer‑based docking studies supported this picture, showing that 4c fits snugly into FAK’s active pocket and makes multiple stabilizing contacts, consistent with direct inhibition of the enzyme.
Early safety signs and next steps
Before any candidate drug can move toward clinical testing, its safety must be evaluated. The team gave single doses of 4c to mice and followed them for two days. At the lowest dose tested, 12.5 mg per kilogram of body weight, the animals showed no obvious signs of distress, and liver and kidney tissues remained largely intact under the microscope. Higher doses began to cause more noticeable tissue damage and biochemical signs of stress, indicating that careful dose selection will be important. Additional computer models of absorption, metabolism, and toxicity suggested that 4c has reasonably favorable properties compared with some existing cancer drugs, though potential liver effects and heart‑related risks will need to be monitored closely. 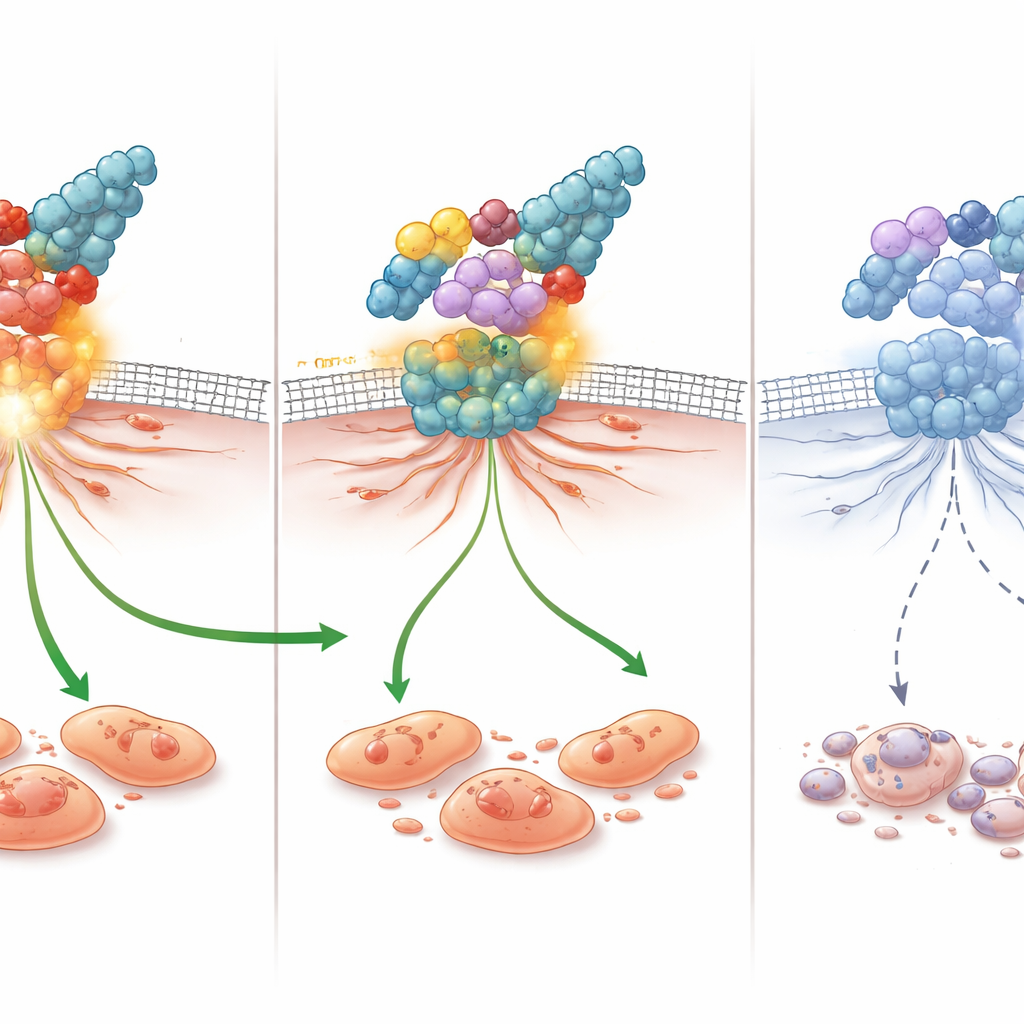
What this could mean for future treatments
In simple terms, this work identifies a new small molecule, 4c, that can both block a key survival pathway in triple‑negative breast cancer cells and actively push them into orderly self‑destruction, while slowing their ability to move and spread. Although the research is still at an early, pre‑clinical stage and much more testing is required in animal tumor models and long‑term safety studies, 4c provides a promising blueprint for a new class of targeted therapies against one of the most challenging forms of breast cancer.
Citation: Abd El Salam, H.A., Abu-Shahba, N., Ibrahim Fouad, G. et al. New indole-linked 1,2,4-triazole derivatives as dual FAK inhibitors and apoptosis inducers targeting survival and migration in triple-negative breast cancer in-vitro. Sci Rep 16, 13134 (2026). https://doi.org/10.1038/s41598-026-41032-1
Keywords: triple-negative breast cancer, focal adhesion kinase, targeted therapy, apoptosis, small-molecule inhibitor