Clear Sky Science · en
Pseudolysogeny-mediated evolutionary trade-offs favor phage therapy by limiting antibiotic resistance and virulence in Cutibacterium acnes
Why this matters for people with acne
Acne is one of the most common skin problems, and many people struggle with side effects and fading benefits from long-term antibiotic use. This study explores a different approach: harnessing naturally occurring viruses that prey on acne-causing bacteria. The authors show that a particular type of virus, using an unusual "half-asleep" strategy inside bacteria, can both calm acne and make the bacteria weaker and less drug-resistant—without disturbing the rest of the skin’s microbial community.
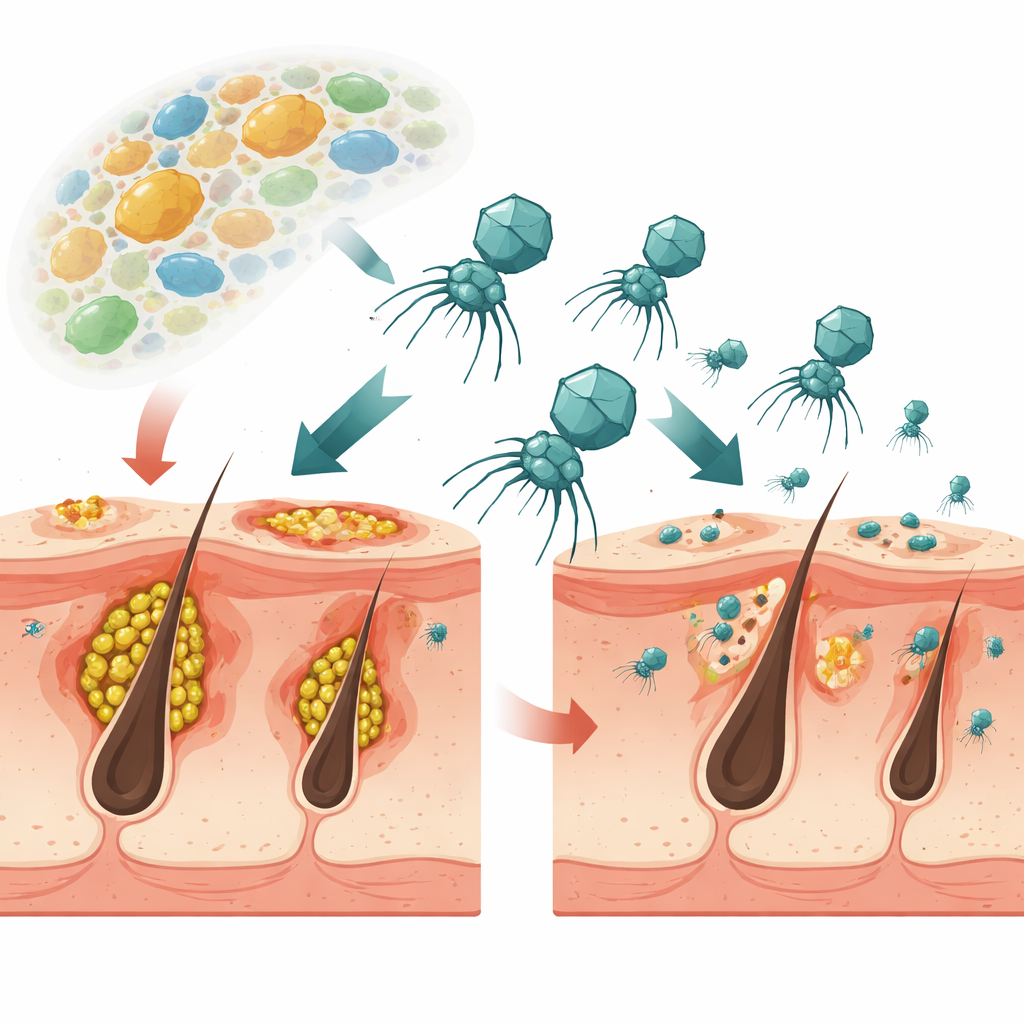
A new kind of virus-based treatment for acne
The research focuses on Cutibacterium acnes, the main bacterium linked to acne, and on viruses that infect it, called phages. Unlike standard antibiotics that hit many bacteria at once, these phages are highly targeted. The team isolated several phages from the skin of healthy people and chose one, named ΦCaCom2, because it could kill many different C. acnes strains, remained stable at room temperature, and was very good at breaking down sticky bacterial layers known as biofilms. In lab tests, this phage suppressed C. acnes growth and destroyed biofilms more effectively than the commonly used antibiotic clindamycin, even when the bacteria were already resistant to that drug.
How a half-asleep virus weakens its bacterial host
ΦCaCom2 uses a strategy called pseudolysogeny: instead of permanently merging with the bacterial DNA or immediately bursting the cell, it can persist inside the bacterium as a circular piece of genetic material. Bacteria that carry this viral circle become temporarily protected against further attack by the same phage, but they pay a price. The study shows that these “pseudolysogen” bacteria grow more slowly, form thinner, weaker biofilms, and lose their competitive edge when grown together with other common skin microbes. In other words, if C. acnes tries to hide from the phage by entering this state, it becomes a poorer competitor in the crowded world of the skin.
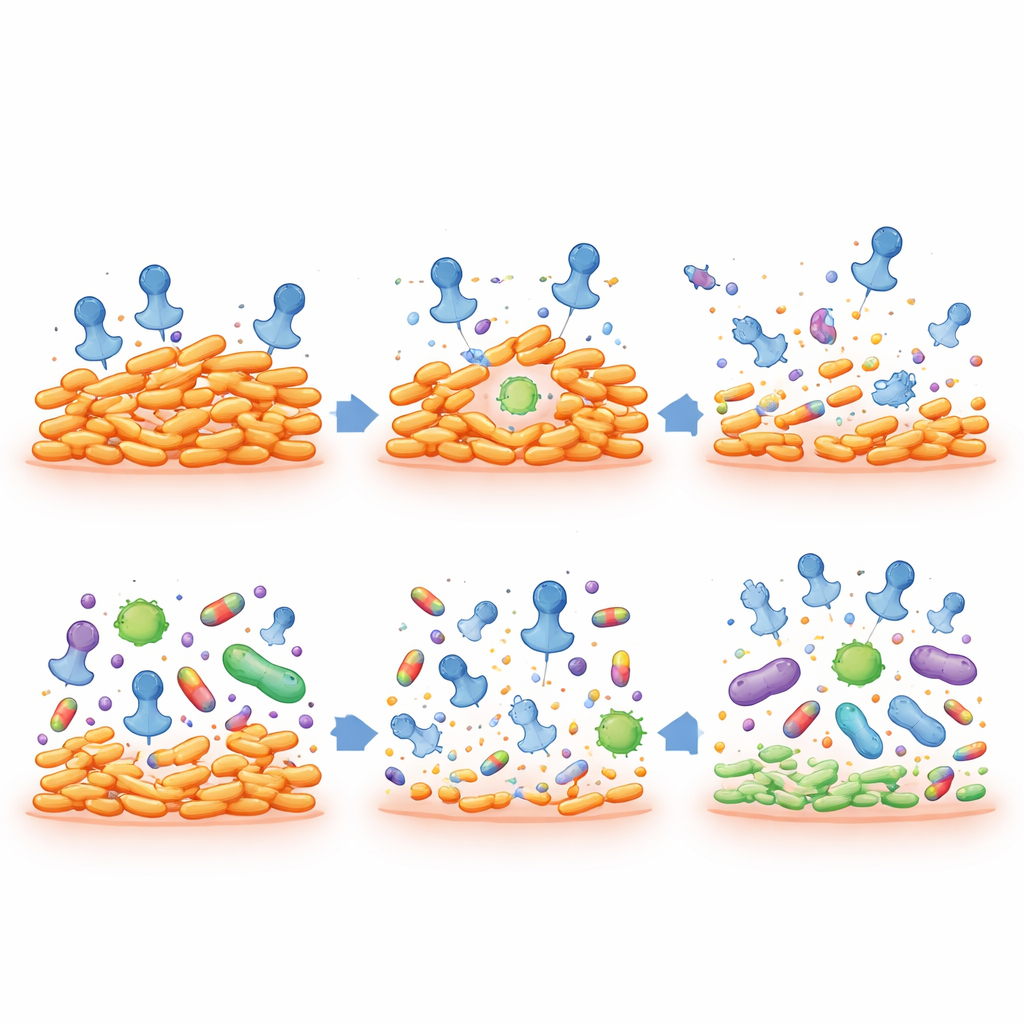
Turning back antibiotic resistance while sparing the rest of the microbiome
The most striking finding is that when drug-resistant C. acnes strains become pseudolysogens, they lose their resistance to key acne antibiotics, including clindamycin and erythromycin. These weakened bacteria become easier to kill, even at low drug doses. At the same time, the phage itself does not appear to spread antibiotic-resistance genes: its genome lacks the machinery needed for this, and direct experiments found no transfer of resistance between strains. In cell culture tests with human skin cells, the phage preparation showed no meaningful toxicity, even at very high doses.
Results from a small human study
To see how this works on actual skin, the researchers conducted a three-month, proof-of-concept trial in 18 adults with mild to moderate acne who had not responded well to standard treatments. Participants sprayed a solution containing the ΦCaCom2 phage on their faces once a day. Over 12 weeks, both red, inflamed lesions and blocked pores were reduced by about two-thirds on average. Bacterial sampling showed that the amount of C. acnes on the skin dropped by roughly one-third and stayed lower even two weeks after stopping treatment. Importantly, the levels of Staphylococcus species—other important skin bacteria—did not rise, suggesting that the treatment did not unbalance the broader skin microbiome. No clear phage-resistant C. acnes clones were detected in the volunteers, and phages themselves persisted on the skin, indicating ongoing gentle pressure on the target bacteria.
What this could mean for future acne care
Taken together, the work suggests that pseudolysogenic phages like ΦCaCom2 can do more than simply kill acne bacteria. By forcing C. acnes into an evolutionary trade-off—temporary viral protection in exchange for weaker growth, thinner biofilms, and lost antibiotic resistance—they help keep the bacterium in check and make it easier to treat without harming the rest of the skin’s microbes. While larger clinical trials and deeper microbiome studies are still needed, this strategy points toward virus-based acne treatments that reduce inflammation, limit the rise of drug resistance, and work in step with the skin’s natural ecosystem rather than against it.
Citation: Trejo-Hernández, A., Checa, A., Quijada-Ibarra, R. et al. Pseudolysogeny-mediated evolutionary trade-offs favor phage therapy by limiting antibiotic resistance and virulence in Cutibacterium acnes. Sci Rep 16, 10184 (2026). https://doi.org/10.1038/s41598-026-40701-5
Keywords: acne phage therapy, Cutibacterium acnes, antibiotic resistance, skin microbiome, pseudolysogeny