Clear Sky Science · en
Integrated transcriptomic and metabolomic analyses reveal distinct energy metabolic signatures and functional properties of RPE cells under two culture conditions
Why eye support cells and lab recipes matter
Behind the light‑sensing cells of our eyes sits a thin, crucial layer called the retinal pigment epithelium (RPE). When these support cells fail, sight can fade in diseases such as age‑related macular degeneration. Scientists can now grow RPE cells from stem cells in the lab for research and potential transplants, but they use different “recipes” of nutrients to keep them alive. This study asks a deceptively simple question with big implications: how much do those recipes change what the cells actually are and how they behave?
Two ways to grow the same eye cells
The researchers started with human induced pluripotent stem cells, which can be coaxed to become almost any cell type, and guided them into RPE‑like cells. They then kept these RPE cells in two of the most commonly used lab media: one based on an additive called B27, and another using a component called KSR. At first glance, both sets of cells looked and acted like RPE: they formed a single layer, made pigment, and could swallow worn‑out pieces of photoreceptor cells, one of the RPE’s key jobs in the eye. But closer inspection revealed clear differences. Cells in B27 became more deeply pigmented and formed a tighter barrier, while cells in KSR showed stronger signals for proteins involved in the visual cycle, the chemical steps that allow us to turn light into vision.
Different fuel choices inside similar cells
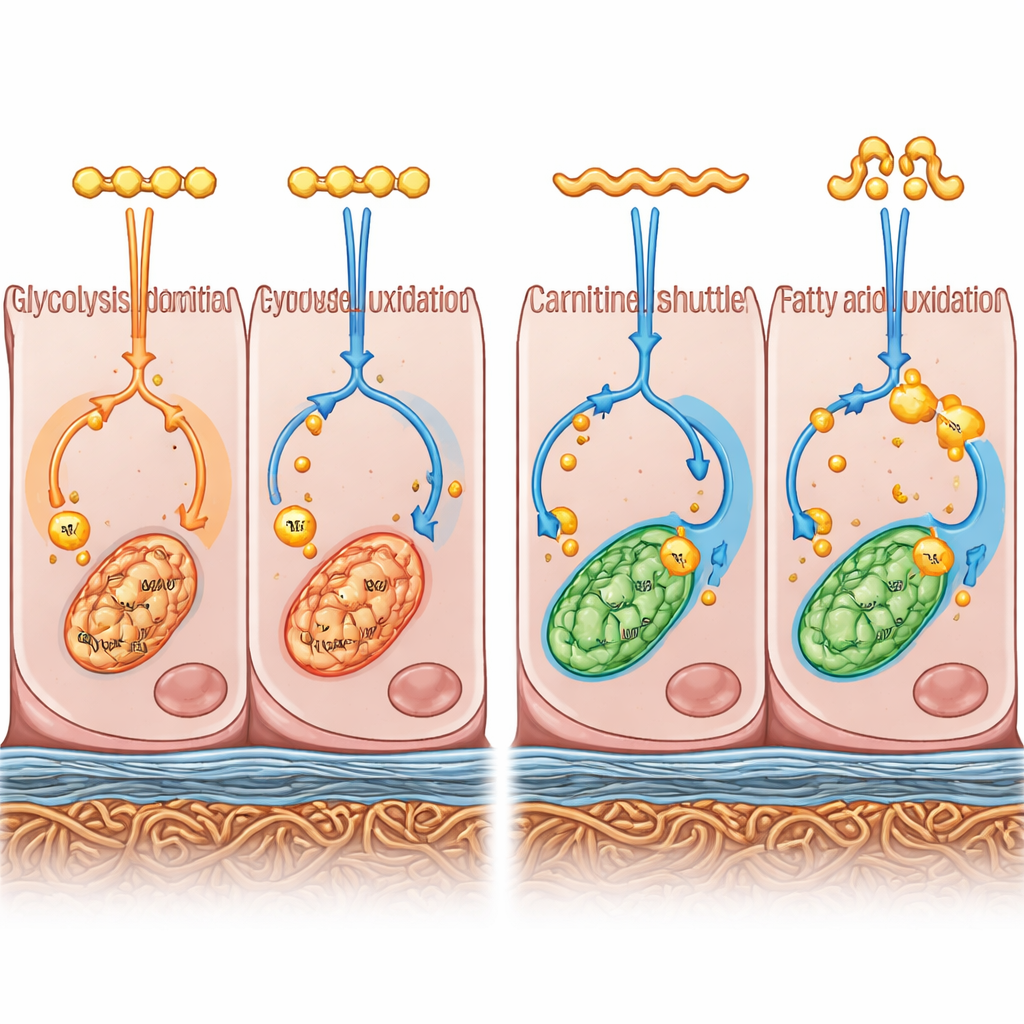
To move beyond appearance, the team combined two powerful approaches. They measured which genes were active across the entire genome (transcriptomics) and which small molecules related to cell metabolism were present (metabolomics). Together, these “multi‑omics” layers painted a picture of how the cells were making and using energy. RPE cells in B27 medium leaned toward breaking down glucose quickly in the cell fluid, a pathway known as glycolysis, and toward building fats for storage. By contrast, RPE cells in KSR medium favored burning fatty acids more completely in their mitochondria, the cell’s energy factories, and channeled this into oxidative energy production. In other words, the two culture conditions nudged the same starting cells into distinct energy‑use preferences.
How energy use shapes structure and barriers
The way the cells chose their fuel was tightly linked to how they organized themselves as a tissue. B27‑grown cells, with their glycolysis‑heavy metabolism, turned on more genes that control the extracellular matrix—the meshwork of proteins and fibers that supports and glues cells together. These cells also showed higher electrical resistance across the layer, a readout of how well they form a barrier similar to that in the eye. When the scientists chemically slowed glycolysis in B27 cells, expression of these matrix genes dropped and the barrier became weaker, suggesting that the faster glucose‑driven state helps build and maintain a robust seal between cells.
A more oxygen‑hungry mode in the other medium
In KSR‑grown cells, the story looked different. Their internal chemistry showed higher levels of free fatty acids and key components needed to shuttle these fats into mitochondria. They accumulated fewer fatty acid–linked intermediates that can build up when this process stalls, hinting at more efficient fat burning. The cells also had higher amounts of the molecules that feed into the final energy‑generating machinery and turned on more genes for oxidative phosphorylation, the oxygen‑using process that produces most of the cell’s ATP, its universal energy currency. Interestingly, both groups of cells had broadly similar activity in the central hub of metabolism, the TCA cycle, suggesting that it is the balance of input fuels and downstream energy conversion that differs most.
What this means for disease models and future therapies
For non‑specialists, the key message is that lab conditions do not just keep RPE cells alive—they help decide what kind of RPE cells they become. One medium pushes the cells toward a glucose‑driven, barrier‑focused state with stronger structural features, while the other encourages a fat‑burning, mitochondria‑driven state that may better mirror the RPE’s role as an energy partner for photoreceptors. Neither culture fully reproduces the environment inside the human eye, but each captures a different slice of normal RPE behavior. The authors argue that recognizing and deliberately choosing between these metabolic “personalities” will be vital when using stem cell–derived RPE to study blinding diseases or to prepare cells for transplantation, where the right balance of barrier strength and energy support could help protect vision.
Citation: Zhang, F., Wang, C., Tang, Q. et al. Integrated transcriptomic and metabolomic analyses reveal distinct energy metabolic signatures and functional properties of RPE cells under two culture conditions. Sci Rep 16, 11992 (2026). https://doi.org/10.1038/s41598-026-39689-9
Keywords: retinal pigment epithelium, stem cell-derived RPE, cell metabolism, age-related macular degeneration, cell culture conditions