Clear Sky Science · en
Macrophages recruited by implanted fibrin gels promote regeneration of injured lymphatic vessels
Why damaged drainage vessels matter
When the body’s lymphatic vessels are damaged during cancer surgery, trauma, or infection, fluid can no longer drain properly from tissues. The result is lymphedema: chronic swelling, discomfort, and a higher risk of infection that can last a lifetime. Today there is no approved drug that reliably repairs these vessels. This study explores a simple idea with big implications: can a tiny plug of the body’s own clotting material, fibrin, guide damaged lymphatic vessels to grow back and restore flow?
Building a temporary bridge for broken pipes
The lymphatic system is a network of vessels that collect excess fluid, fats, and immune cells from tissues and return them to the bloodstream. Larger “collecting” vessels actively pump lymph forward using rhythmic contractions and one-way valves. When these vessels are cut or removed, as often happens when lymph nodes are taken out during cancer treatment, the remaining segments do not always reconnect on their own. The authors reasoned that fibrin, the natural scaffold formed during blood clotting, might act as a temporary bridge across a gap in a severed collecting vessel, giving cells something to grow through as they rebuild the connection. 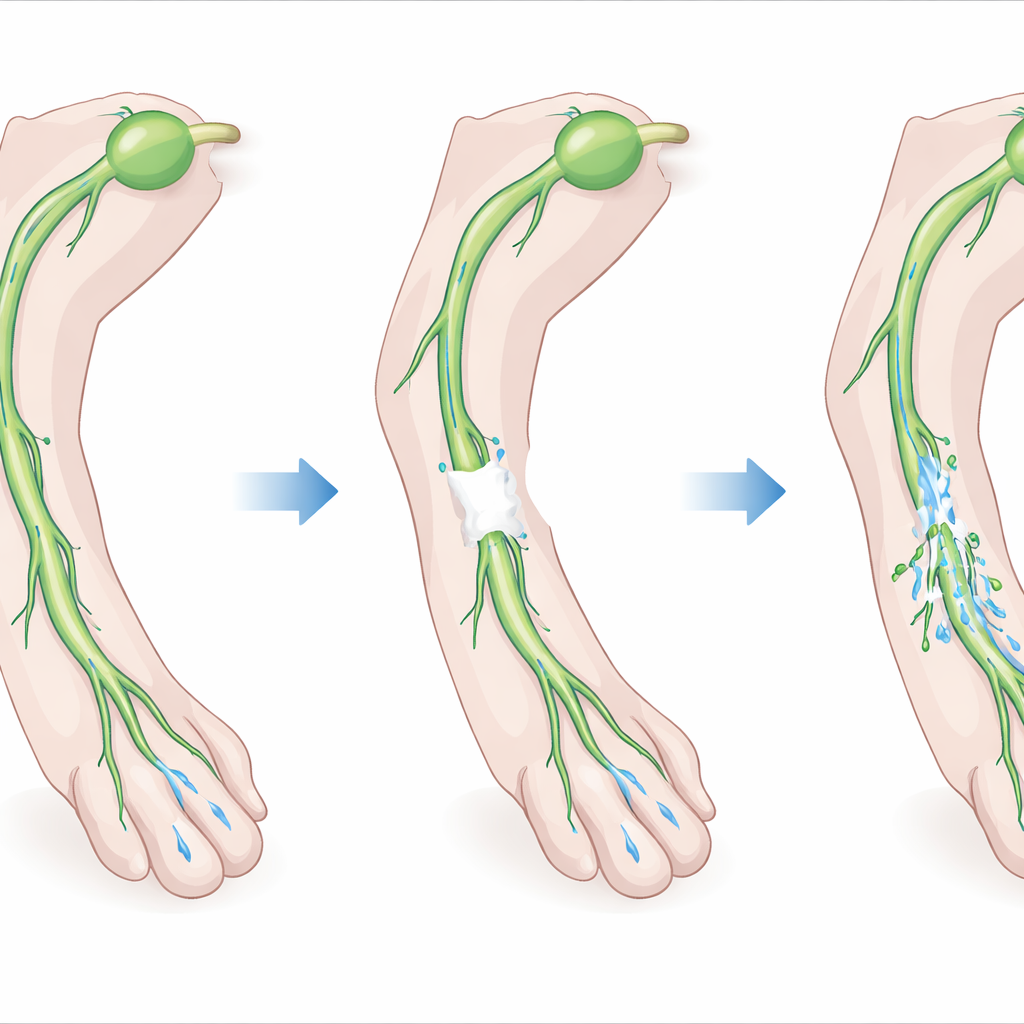
Testing a gel guide in living tissue
To test this idea, the researchers first cultured short segments of mouse collecting lymphatic vessels inside three-dimensional fibrin gels. They found that the vessels sent out many thin sprouts into the gel, made up of both the lining cells of lymphatic vessels and their surrounding muscle cells. The amount and pattern of sprouting depended strongly on how concentrated, and therefore how stiff, the fibrin was: an intermediate concentration produced the longest and most numerous sprouts, while gels that were too soft or too stiff limited growth. This helped them choose an optimal gel formulation to implant into animals.
Restoring flow in injured legs
The team then created a controlled injury in the hindlimbs of mice by removing a 1–2 millimeter segment of a key collecting lymphatic vessel while leaving nearby blood vessels intact. In some animals they filled the gap with the fibrin gel; in others they did not. Using near-infrared imaging to track a fluorescent dye injected into the paw, they showed that, without gel, lymph flow to the main draining lymph node largely failed to recover. With gel, new lymphatic sprouts grew from the cut ends of the vessel into and through the gel, reconnecting the broken segments within a few weeks. Pumping activity upstream of the injury returned, and dye once again reached the original draining lymph node, although some flow still took alternate routes—evidence of both repair and rerouting.
Immune helpers and a key signaling molecule
The fibrin plugs did more than provide structure: they also drew in immune cells. Flow cytometry and microscopy revealed that macrophages, and to a lesser extent neutrophils, accumulated in the gel around regenerating vessels. When the researchers locally depleted macrophages and functionally disabled neutrophils using clodronate-loaded particles, lymphatic sprouting and the return of pumping were largely abolished, underscoring the importance of these cells. A survey of tissue signaling proteins showed that one molecule, the chemokine CCL5, rose sharply after injury plus gel implantation. Mice lacking CCL5 were significantly less likely to regain proper filling of the vessel upstream of the injury, and their early pumping activity was reduced, even though immune cells could still enter the area. This suggests that CCL5 helps control how these cells support repair, not simply whether they arrive. 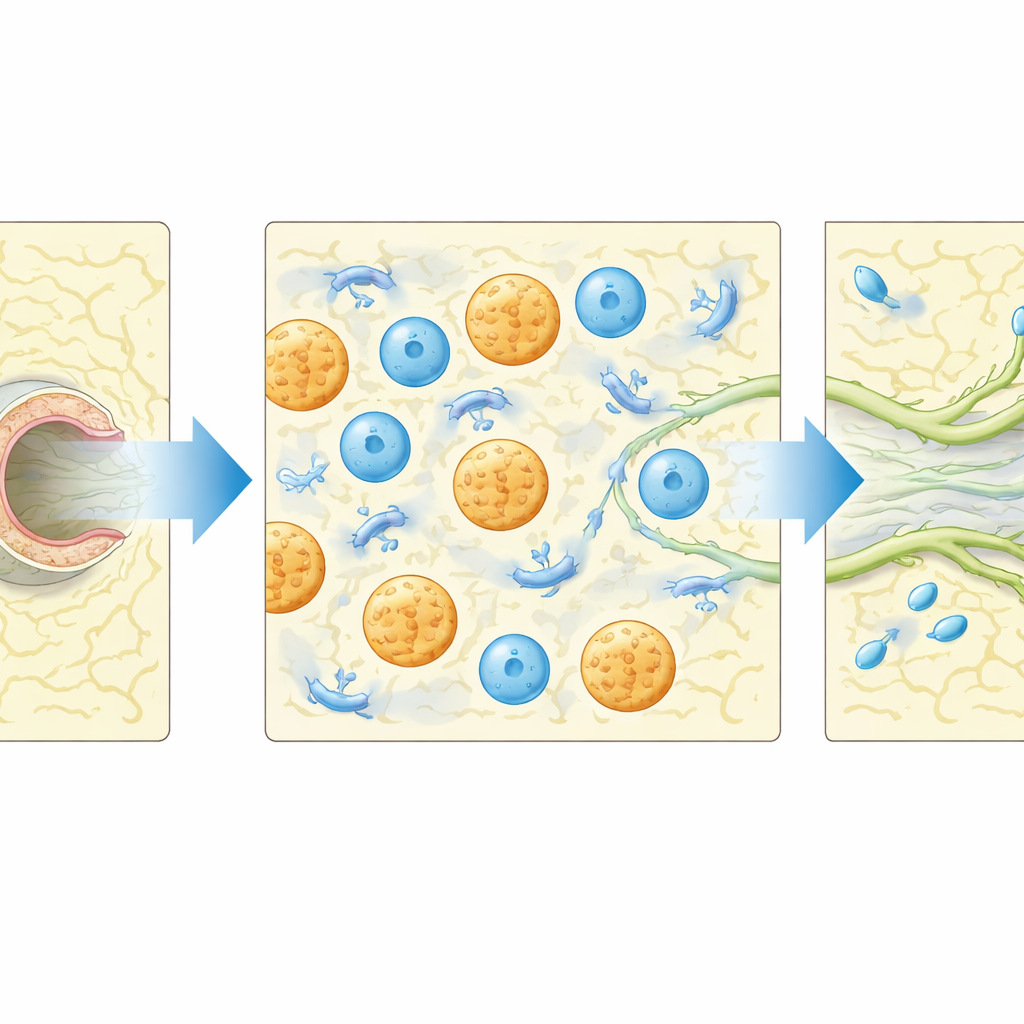
What this could mean for people with swelling
Overall, the study shows that a carefully tuned fibrin gel can act as a guided repair kit for damaged lymphatic vessels, allowing them to sprout, reconnect, and resume pumping fluid to the correct lymph node. The work also highlights that the body’s own cleanup crew—macrophages and neutrophils—and the signaling molecule CCL5 are central to this regrowth process. While the mouse model does not fully mimic human lymphedema, these results point toward future treatments that combine biomaterial scaffolds with targeted control of immune signals to encourage durable lymphatic repair after surgery or injury.
Citation: Razavi, M.S., Lei, PJ., Amoozgar, Z. et al. Macrophages recruited by implanted fibrin gels promote regeneration of injured lymphatic vessels. Sci Rep 16, 14337 (2026). https://doi.org/10.1038/s41598-026-39167-2
Keywords: lymphedema, lymphatic regeneration, fibrin hydrogel, macrophages, tissue repair