Clear Sky Science · en
In silico design of a multiepitope vaccine against antibiotic drug-resistant Acinetobacter baumannii
Why this matters for everyday health
Hospitals are meant to heal us, yet some of the most dangerous infections now arise inside them. One culprit, a hardy bacterium called Acinetobacter baumannii, has learned to shrug off many of our strongest antibiotics, turning routine care into a life‑threatening risk for vulnerable patients. This study explores a new way to stay ahead of such superbugs: using computers to design a next‑generation vaccine made from carefully chosen tiny protein fragments, with the goal of preventing infections before they start.
A hospital germ that won’t go away
Acinetobacter baumannii thrives on hospital surfaces and equipment, where it can infect people with weakened immune systems, chronic lung disease, or long stays in intensive care. It belongs to a notorious group of hospital microbes known for resisting multiple drugs. Once this germ becomes resistant to carbapenems, a class of last‑resort antibiotics, doctors are left with few safe options. Because no approved vaccine exists, the World Health Organization has labeled this bacterium a top priority for vaccine development, spurring researchers to search for smarter ways to target it.
Letting the bacterium reveal its weak spots
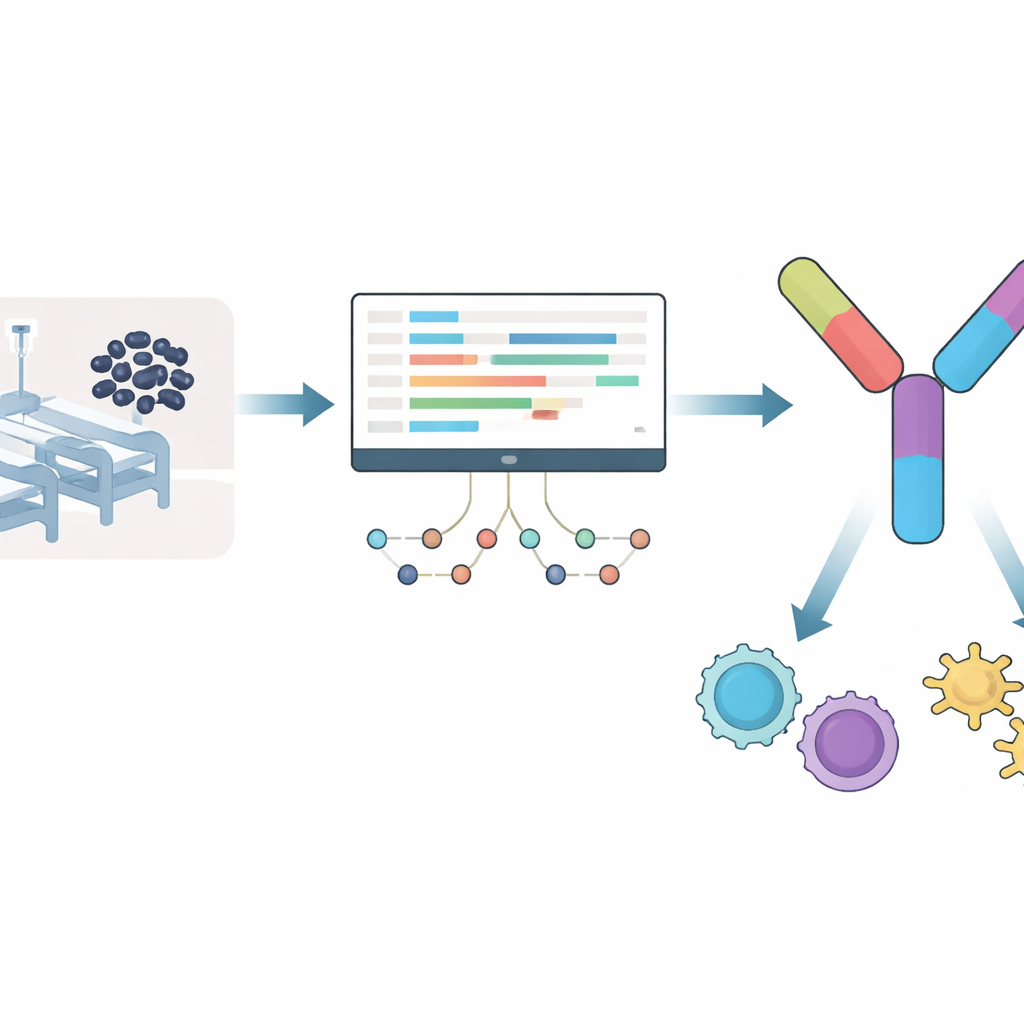
Instead of starting from guesswork about which bacterial parts might make good vaccine targets, the team watched how Acinetobacter baumannii responds when it is attacked by a key antibiotic, meropenem. They analyzed RNA sequencing data, which shows which genes are switched on or off, after nine hours of drug exposure. Over a thousand genes were turned up and a similar number turned down, especially in pathways linked to metabolism, secretion systems, and drug‑pumping machinery the bacterium may use to escape treatment. From the proteins encoded by these activated genes, the researchers eliminated any that resembled human proteins or those from common gut bacteria, to reduce the risk of side effects. This filtering left a shortlist of bacterial proteins that are both important under drug attack and unlikely to confuse the immune system.
Building a many‑piece vaccine from tiny snippets
From this refined set, the scientists looked for short stretches of protein, called epitopes, that human immune cells are especially good at recognizing. They focused on two types: pieces that stimulate “killer” T cells, which destroy infected cells, and those that activate “helper” T cells, which coordinate broader immune defenses. Using established prediction tools, they selected 12 killer‑cell epitopes and 7 helper‑cell epitopes that were non‑toxic, unlikely to trigger allergies, and strongly predicted to bind common human immune markers. Crucially, these epitopes came from proteins that were fully conserved across several highly virulent strains of Acinetobacter, increasing the chance that one vaccine could protect against many versions of the germ.
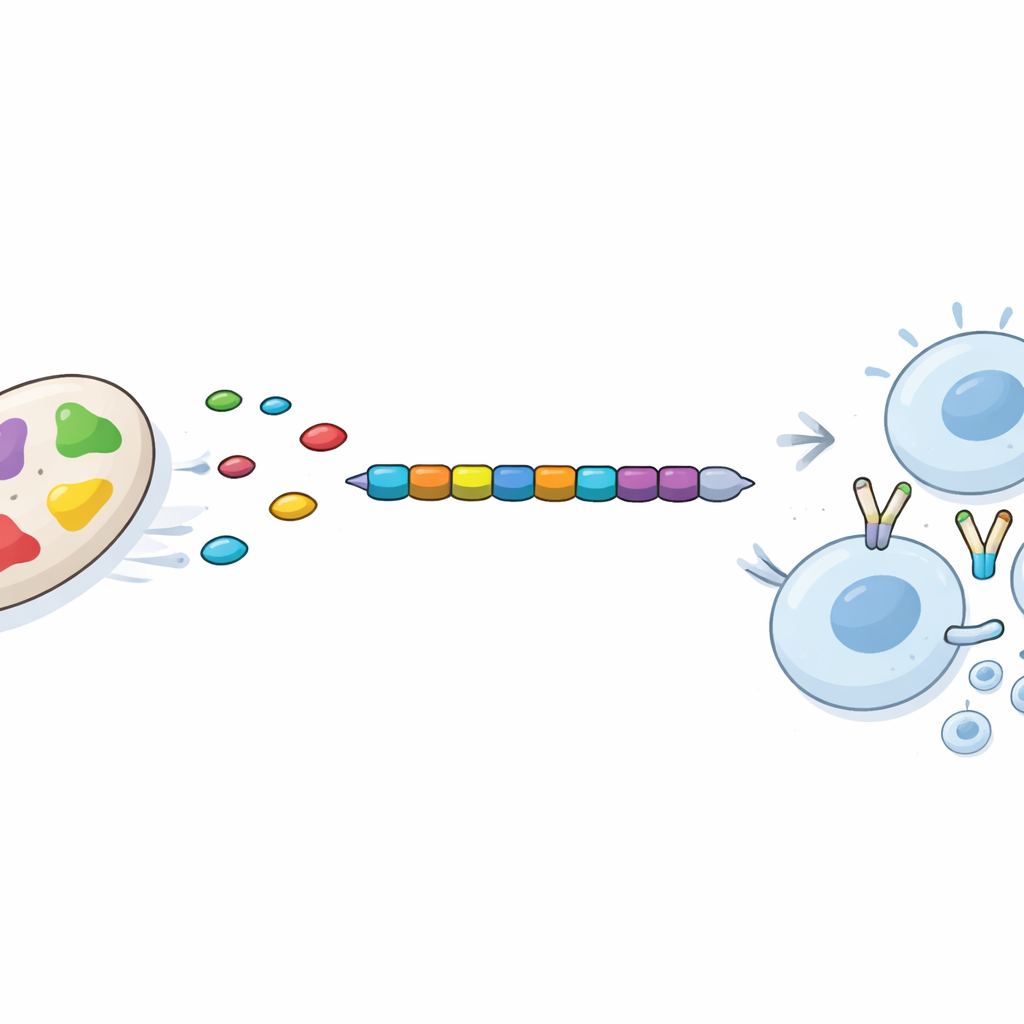
Testing the virtual vaccine for stability and response
The chosen epitopes were stitched together in a defined order to create a single 335‑amino‑acid vaccine protein, with a human‑derived immune‑boosting component added at one end. The team then used state‑of‑the‑art computer modeling to predict how this multi‑piece protein would fold in three dimensions and how stable it would be in a watery environment like the body. Detailed simulations suggested the structure quickly settled into a compact, stable shape and maintained it over time. Separate docking studies indicated that each epitope could snugly fit into human immune receptors. Finally, an in silico immune system simulator predicted that a dose of this vaccine would rapidly clear the introduced antigen, spur early antibody production, strongly activate killer and helper T cells, and build lasting immune memory, all without signs of an exaggerated or unbalanced response.
What this means and what comes next
This work does not yet produce an injection ready for patients, but it shows how powerful computer‑based methods can be for designing vaccines that target the very features that make a hospital germ drug‑resistant. By focusing on bacterial components that are switched on during antibiotic stress and shared across many dangerous strains, and by assembling multiple such pieces into one construct, the study outlines a promising blueprint for a broad‑coverage vaccine against Acinetobacter baumannii. The next crucial steps will be laboratory and animal studies to confirm that this virtual vaccine is safe, stable, and protective in the real world—an essential bridge from clever design on a screen to lifesaving protection at the bedside.
Citation: Khadka, S., Khan, M.A., Iqbal, S. et al. In silico design of a multiepitope vaccine against antibiotic drug-resistant Acinetobacter baumannii. Sci Rep 16, 14151 (2026). https://doi.org/10.1038/s41598-025-30795-8
Keywords: antibiotic resistance, hospital infections, Acinetobacter baumannii, vaccine design, computational immunology