Clear Sky Science · en
Fecal microbiota transplantation plus pembrolizumab and axitinib in metastatic renal cell carcinoma: the randomized phase 2 TACITO trial
Why Your Gut Might Matter in Kidney Cancer
Doctors have long known that our immune system can be coaxed to fight cancer, but these powerful drugs do not work for everyone. This study asks a surprising question with big implications: can changing the bacteria that live in a patient’s gut make immune-based cancer drugs work better for advanced kidney cancer? By moving selected gut microbes from patients who did exceptionally well on immunotherapy into newly diagnosed patients, the researchers tested whether the “right” microbes could tilt the odds in favor of longer control of the disease.
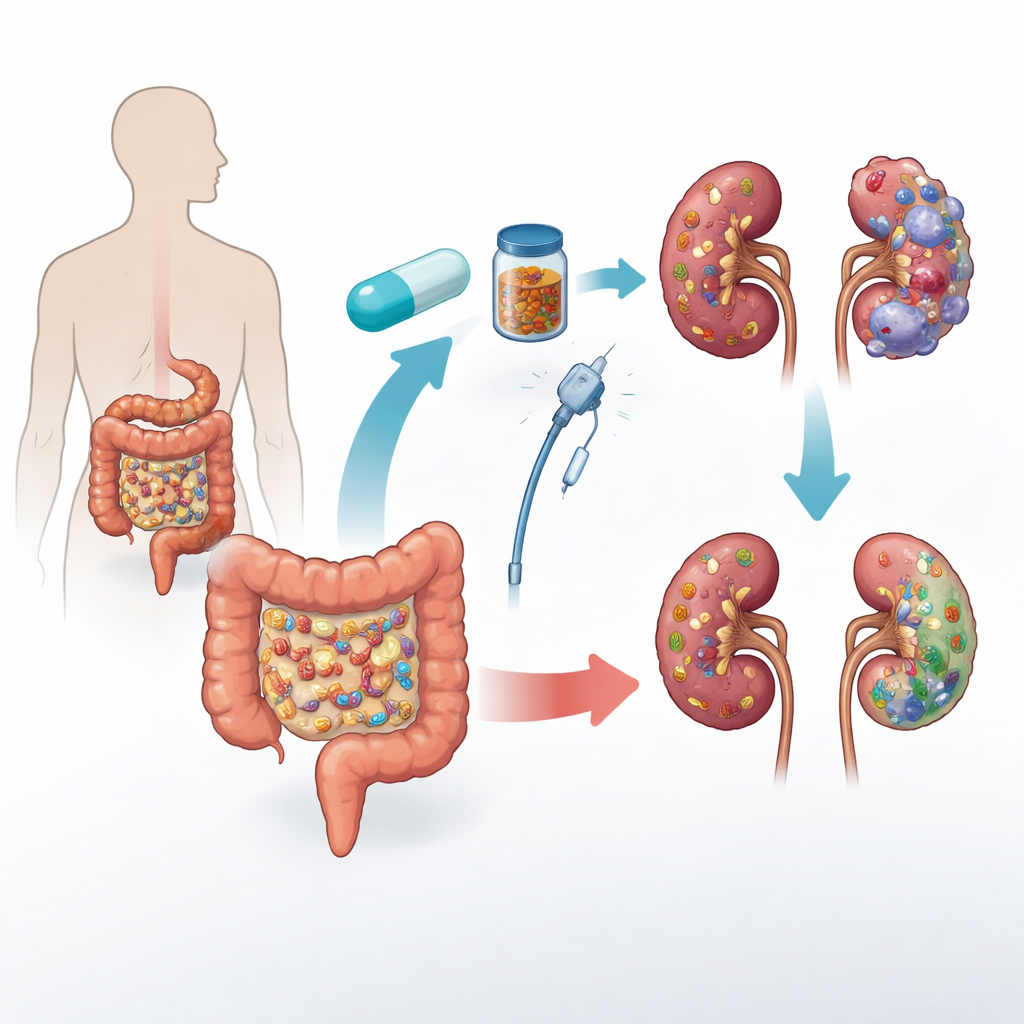
A New Partner for Cancer Immunotherapy
The trial focused on metastatic renal cell carcinoma, the most common form of advanced kidney cancer. Standard first-line treatment often combines two medicines: pembrolizumab, which helps immune cells recognize cancer, and axitinib, which cuts off blood supply to tumors. Even with this powerful duo, many patients see their cancer grow again within a year and a half. Earlier clues from other cancers suggested that people whose gut microbes were richer and more balanced tended to respond better to immune therapies. Building on this, the TACITO trial tested whether transplanting stool from two kidney cancer patients who had complete, long-lasting responses to immunotherapy could improve outcomes for newly treated patients.
How the Trial Was Run
In this randomized, double-blind study, 45 patients with untreated metastatic kidney cancer received standard pembrolizumab plus axitinib. Half were assigned to donor fecal microbiota transplantation (d-FMT) and half to placebo capsules and solution (p-FMT). Everyone had three procedures over six months: first a colonoscopy-based infusion, then two rounds of frozen capsules. Neither patients nor treating doctors knew who received real donor material or placebo. The main goal was to see how many patients were still free of cancer worsening one year after randomization; the team also tracked how long patients stayed progression free overall, how long they lived, how many tumors shrank, and how their gut microbes changed over time.
What Happened to Tumors and Survival
Although the trial just missed its primary statistical target at 12 months, the clinical signals were striking. A year after randomization, 70% of patients in the donor group and 41% in the placebo group had not seen their disease progress, a difference that was borderline by standard statistical rules. Looking over the full follow-up period, people who received donor microbes had a median progression-free survival of 24 months, compared with only 9 months in the placebo group. The share of patients whose tumors shrank (partial or complete response) was also higher with donor FMT: 52% versus 32%. Overall survival favored the donor group as well (41 versus 28.3 months at the time of analysis), although this difference was not yet statistically firm, likely because the number of deaths was still modest.
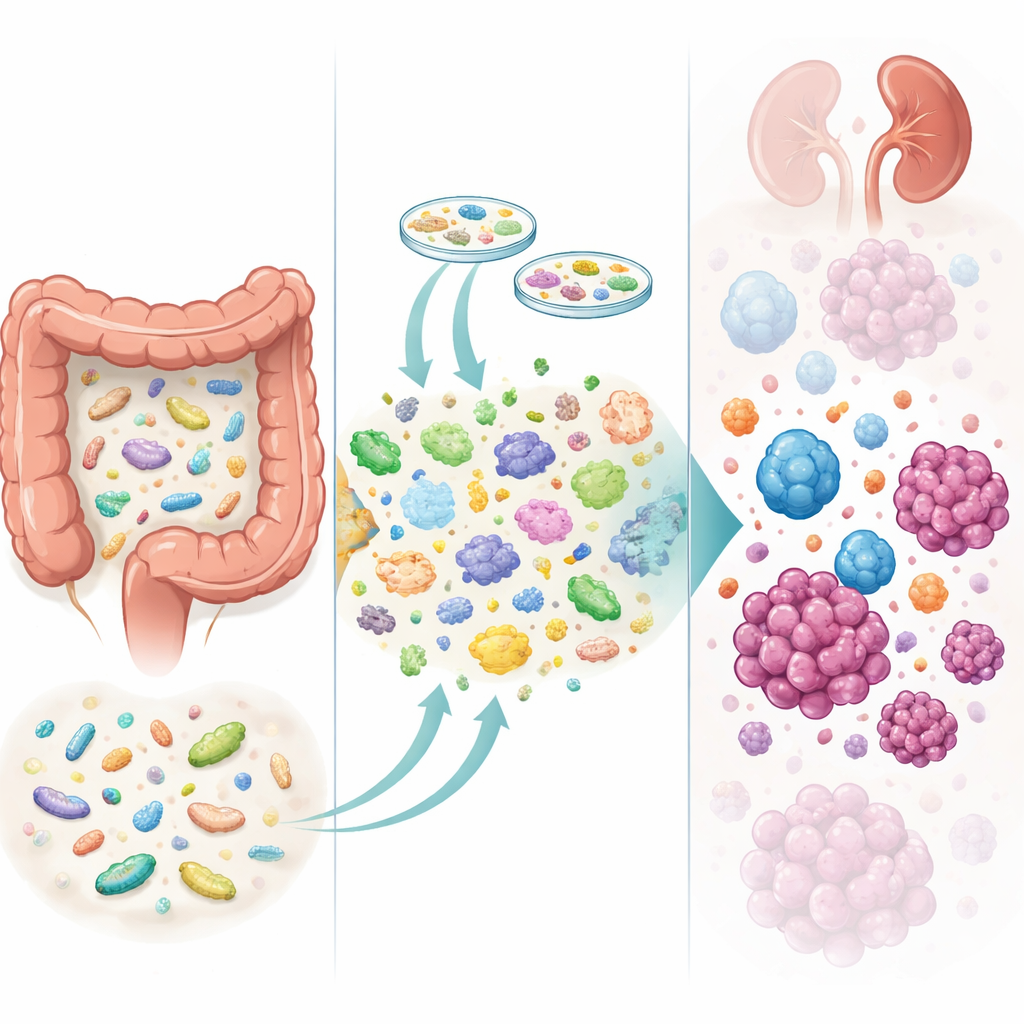
How the Microbes Changed
To see what was happening inside the gut, the team collected more than 240 stool samples before and after treatment. High-depth DNA sequencing showed that patients who received donor material gained more microbial species and had greater shifts in their gut composition than those on placebo. In other words, the transplant clearly “took.” However, simply having more donor strains did not guarantee better outcomes. Instead, the details mattered: acquiring certain helpful bacteria, such as a strain of Blautia wexlerae that produces beneficial short-chain fatty acids, was linked to longer control of the disease. Gaining other strains, including a recently described cousin of the well-known microbe Akkermansia muciniphila, was associated with shorter control. Likewise, losing some original gut residents, such as inflammatory Escherichia coli, appeared favorable, whereas losing a starch-breaking species called Ruminococcus bromii tended to be a bad sign.
Safety and Who Might Benefit Most
The procedures were generally well tolerated. Side effects directly tied to the transplant—whether via colonoscopy or capsules—were rare and mostly mild, with no deaths or detected infections from donor material. The usual side effects from pembrolizumab and axitinib, such as diarrhea and liver enzyme changes, occurred at similar rates to what is seen in standard practice. When researchers looked specifically at patients whose cancer carried a worse initial outlook, the advantages of donor FMT appeared even larger: these patients had substantially longer control of their disease and higher response rates if they received donor microbes rather than placebo. This suggests that microbiome-based strategies may be especially useful for people starting from a more fragile position.
What This Means for Patients
For someone facing advanced kidney cancer, these results hint at a future in which treatment is not just about drugs, but also about tuning the microbial community in the gut. In this relatively small trial, carefully screened stool transplants from exceptional responders appeared safe and prolonged the time before cancer worsened, and may ultimately improve survival, when added to standard immunotherapy. The findings also show that not all microbes are equal—specific bacterial gains and losses, rather than a blanket increase in donor strains, seem to matter most. Larger trials and more refined, pill-based microbial mixtures will be needed before such approaches become routine, but the message is clear: the microbes in our intestines may become powerful allies in the fight against cancer.
Citation: Porcari, S., Ciccarese, C., Heidrich, V. et al. Fecal microbiota transplantation plus pembrolizumab and axitinib in metastatic renal cell carcinoma: the randomized phase 2 TACITO trial. Nat Med 32, 1316–1324 (2026). https://doi.org/10.1038/s41591-025-04189-2
Keywords: gut microbiome, fecal microbiota transplantation, immune checkpoint therapy, metastatic renal cell carcinoma, cancer immunotherapy