Clear Sky Science · en
Fecal microbiota transplantation plus immunotherapy in non-small cell lung cancer and melanoma: the phase 2 FMT-LUMINate trial
Why Your Gut Might Matter in Cancer Treatment
Cancer drugs that “wake up” the immune system have transformed the outlook for many people with advanced lung cancer and melanoma. Yet, for roughly half of patients, these drugs do not work well enough. This study asks a surprising question with real-world consequences: can changing the community of microbes living in the gut—using fecal material from healthy donors—make immune-based cancer drugs more effective and reveal why they sometimes cause serious side effects?
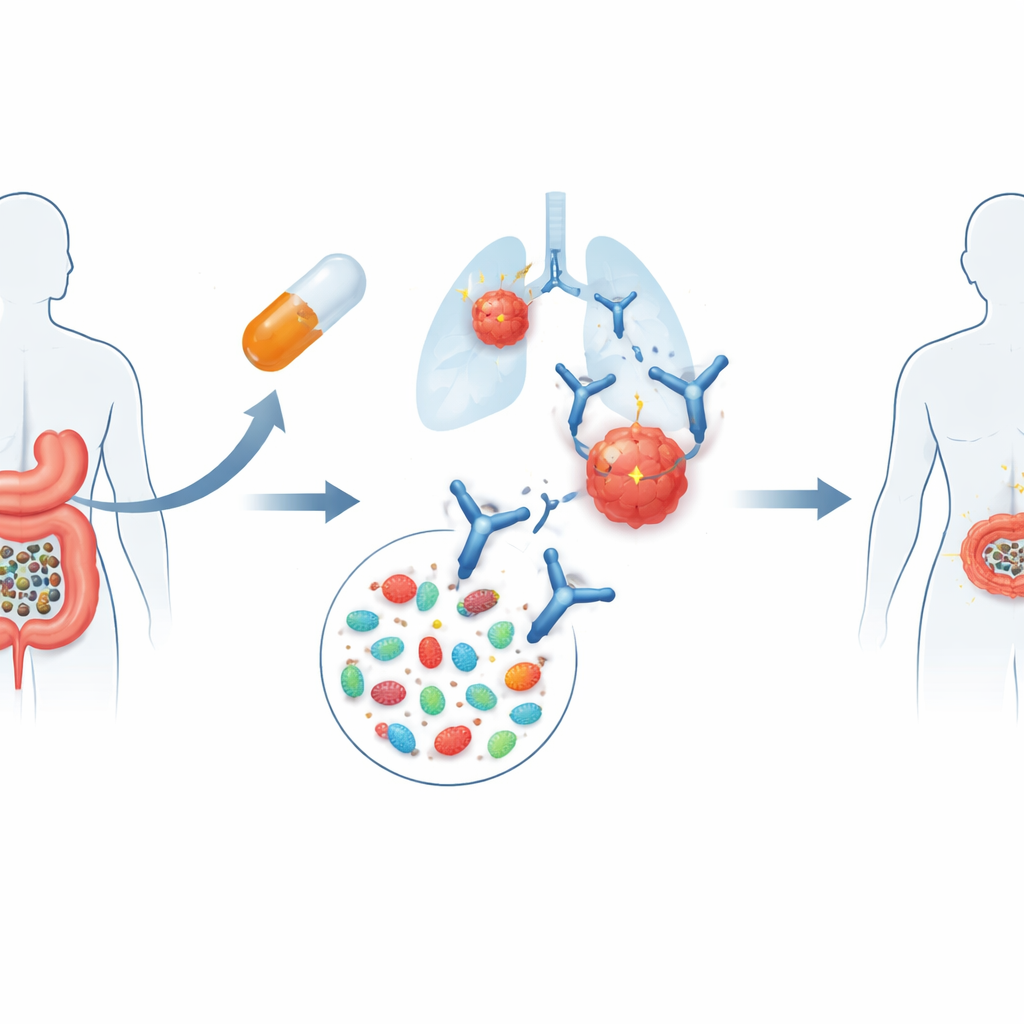
Using Donor Stool as a Helper Therapy
The FMT-LUMINate trial tested a simple but bold idea. Patients with advanced non-small cell lung cancer or melanoma, who were just about to start standard immune checkpoint inhibitor therapy, first swallowed capsules containing processed stool from rigorously screened healthy volunteers. This procedure, known as fecal microbiota transplantation (FMT), aims to reset the gut ecosystem. Within a week, patients began their usual cancer immunotherapy: lung cancer patients received a single drug targeting PD-1, while melanoma patients received a combination targeting both PD-1 and CTLA-4, a more intensive regimen known to be powerful but also more toxic.
Stronger Responses Than Expected
In the lung cancer group, 16 of 20 patients saw their tumors shrink, an objective response rate of 80%—notably higher than the roughly 40–45% success rates typically reported for the same drug alone. Most of the remaining patients at least kept their disease stable for six months or more, and all were still alive one year after starting treatment. In the melanoma group treated with combination immunotherapy, 15 of 20 patients responded, including four complete responses, again above historical expectations. These results suggest that a single course of healthy-donor FMT, given before immunotherapy, can substantially boost the chances that these powerful drugs will work.
Safety: When Gut Microbes and Strong Drugs Interact
Safety looked different depending on the cancer and treatment backbone. In lung cancer patients receiving only PD-1–blocking therapy, no severe (grade 3 or higher) side effects were attributed to the combination of FMT plus immunotherapy. In melanoma patients on the dual-drug regimen, however, serious immune-related problems such as severe diarrhea, colitis, and heart inflammation (myocarditis) were more common and appeared earlier than usually seen. Careful genetic analysis of stool samples from donors and patients pointed to one donor whose gut was rich in a group of bacteria called Prevotella. All recipients of this donor’s stool in the dual-therapy group developed severe side effects, while similar donors caused no such problems when paired with single-drug therapy. This highlights that the safety of FMT may depend strongly on both donor microbiology and the specific cancer drug combination.
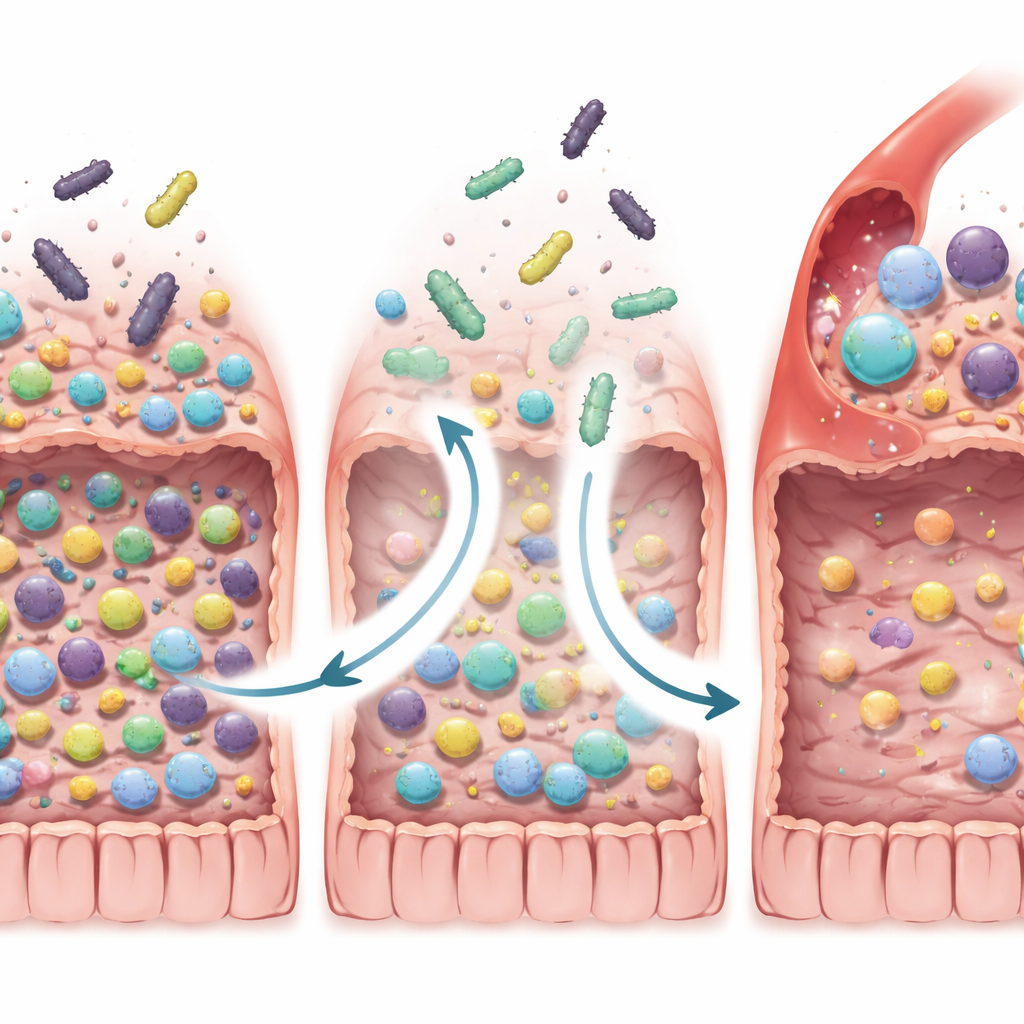
Good Outcomes Linked to Losing the Wrong Microbes
The scientists then dug deeper into how FMT reshaped the gut ecosystem. They expected that patients who did well might simply acquire more of their donor’s “good” bacteria. Instead, they found something more subtle: responders tended to lose a larger fraction of the bacterial species they had at baseline, especially certain microbes previously tied to resistance to immunotherapy and to chronic inflammation, including specific Enterocloster, Clostridium, Streptococcus, and Dialister species. This pattern was seen not only in FMT-LUMINate but also when the team re-analyzed data from three earlier FMT cancer trials. In lab mice, reintroducing these “lost” bacteria into animals that had received responder stool weakened the anti-tumor effects of immunotherapy, supporting the idea that getting rid of harmful residents is crucial.
Ripples Through Metabolism and the Immune System
Removing troublesome microbes did not just change the gut map on paper—it altered chemistry and immunity throughout the body. Patients who lost more of their baseline bacteria showed different blood metabolite profiles, particularly in compounds derived from the amino acid tryptophan. Non-responders and those who retained more of their original microbes had higher levels of metabolites such as quinolinic acid and kynurenine, which have been linked to immune suppression and poor responses to cancer immunotherapy. At the same time, patients with greater bacterial loss displayed more activated cancer-fighting CD8 T cells and fewer regulatory T cells, which normally act as brakes on immune responses. Together, these shifts point to a more hostile environment for tumors and a friendlier one for anti-cancer immune cells.
What This Means for Future Cancer Care
For non-specialists, the central message is that the microbes already living in a patient’s gut can either help or hinder cutting-edge cancer drugs. This phase 2 study shows that a single FMT from carefully screened healthy donors can safely increase the success rates of immunotherapy for lung cancer and melanoma, while underlining the need for rigorous donor selection to avoid boosting dangerous side effects. Crucially, the benefits seem to come less from “adding good bugs” than from “evicting bad ones.” That insight offers a roadmap for designing next-generation microbiome-based treatments—whether FMT or more refined microbial cocktails—aimed at selectively removing harmful species, rebalancing metabolism, and giving the immune system a clearer shot at eradicating cancer.
Citation: Duttagupta, S., Messaoudene, M., Hunter, S. et al. Fecal microbiota transplantation plus immunotherapy in non-small cell lung cancer and melanoma: the phase 2 FMT-LUMINate trial. Nat Med 32, 1337–1350 (2026). https://doi.org/10.1038/s41591-025-04186-5
Keywords: fecal microbiota transplantation, cancer immunotherapy, gut microbiome, non-small cell lung cancer, melanoma