Clear Sky Science · en
MHC class I on target cells regulates CD4+ T cell-mediated immunity
Why This Immune Story Matters
Our immune system is often described as an army with clearly assigned roles: some cells spot threats, some remember past battles and others do the actual attacking. This study overturns a long‑standing rule about who can attack which targets. The researchers show that a common marker on our cells, called MHC class I, does more than simply help one type of immune cell recognize danger. When this marker is missing, it unexpectedly changes how another type of immune cell, CD4 T cells, can kill both normal tissues and cancer cells. The results may help explain why some cancers that hide from the immune system still respond well to modern immunotherapies, and they point to new ways of treating cancer and complications from bone marrow transplants.
A Security Badge With a Hidden Job
Every nucleated cell in our body normally carries MHC class I molecules on its surface, like security badges displaying small protein fragments. Textbooks say these badges are mainly for CD8 T cells, the classic “killer” cells, while a different badge type, MHC class II, guides CD4 T cells, the “helper” cells. Many viruses and tumors dodge CD8 T cells by reducing their MHC I badges, a well‑known escape trick. The authors asked a question that had been largely overlooked: beyond its role as a badge for CD8 T cells and natural killer cells, does MHC I on the target cell itself change how vulnerable that cell is to being destroyed by CD4 T cells?
When Missing Badges Make Tissues Fragile
To test this, the team used mouse models of a serious complication of bone marrow transplantation called graft‑versus‑host disease, where donor immune cells attack the recipient’s gut and other organs. They engineered mice whose tissues lacked MHC I but whose incoming donor cells still saw the same foreign MHC II signals. This setup isolates CD4 T cell–driven damage. Surprisingly, mice whose gut cells lacked MHC I suffered far worse disease and died more often than normal mice, even though the donor CD4 T cells were activated to a similar degree in both groups. The extra damage did not show up when the gut was injured by chemicals or radiation alone, and it did not depend on natural killer cells. This pointed to a specific weakness of MHC I–deficient tissues to CD4 T cell attack, not a general frailty.
Cancers That Hide Become Easier Targets
The researchers then turned to melanoma, a common skin cancer. They used gene editing to remove MHC I from mouse melanoma cells. In dishes, these MHC I–deficient tumor cells were killed more efficiently by CD4 T cells that recognized a melanoma antigen. In mice, tumors lacking MHC I shrank more after transfer of these CD4 T cells than tumors with normal MHC I. These results suggest that when cancers lose MHC I to avoid CD8 T cells, they may inadvertently make themselves more vulnerable to a different kind of immune attack driven by CD4 T cells, especially under the right therapeutic conditions.
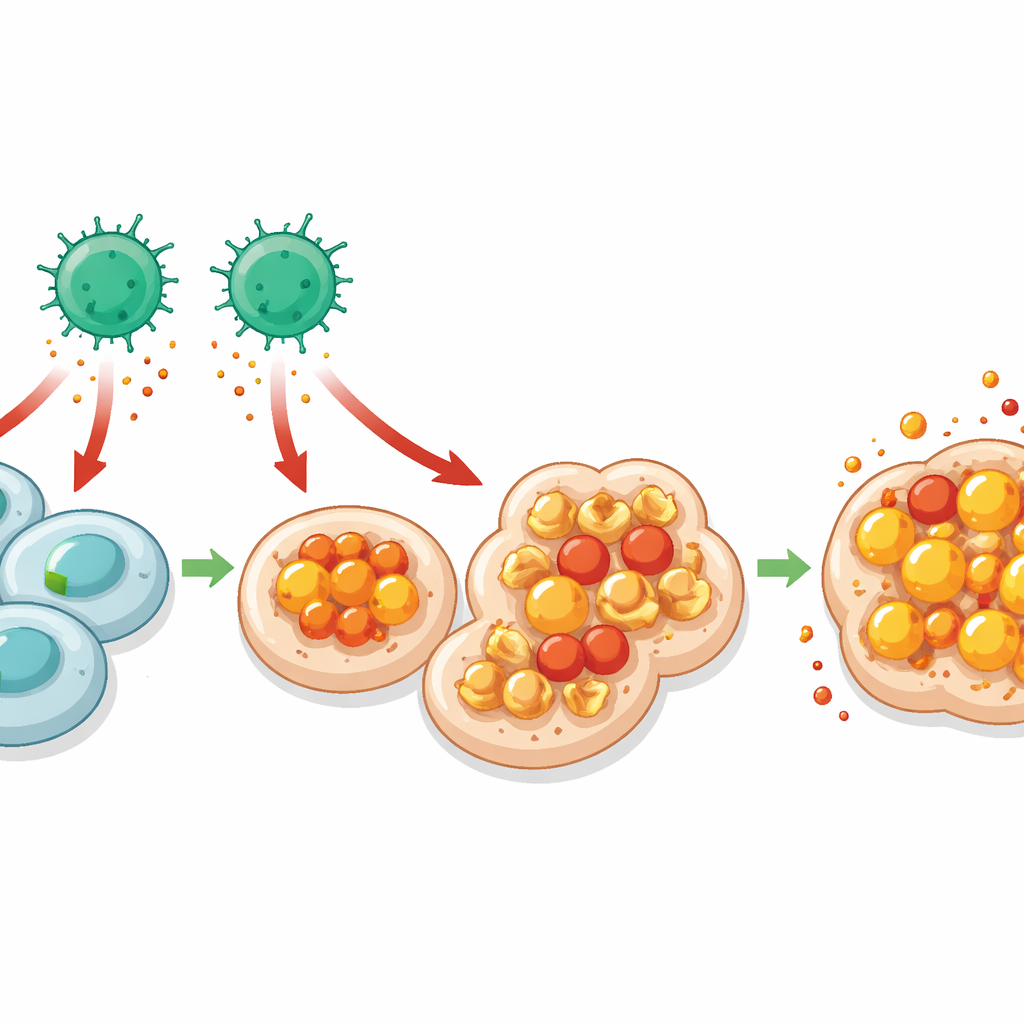
Iron, Oxidized Fats and a Fiery Cell Death
Diving deeper, the team asked why MHC I–deficient cells are easier for CD4 T cells to kill. Single‑cell RNA sequencing of gut cells revealed stronger signs of response to interferon‑gamma, a potent alarm signal often released by T cells, specifically in cells lacking MHC I. These same cells showed signatures of disrupted iron handling and increased lipid peroxidation—chemical damage to fats in cell membranes. Together, these features point to ferroptosis, a form of iron‑dependent cell death marked by runaway fat oxidation. The authors confirmed that gut and tumor cells without MHC I accumulated more lipid damage and were protected when iron was chelated or when ferroptosis was chemically blocked. Neutralizing interferon‑gamma reduced this damage, tying CD4 T cell–derived signals to ferroptosis in the vulnerable target cells.
Clues From Patients on Immunotherapy
To see whether this mechanism might matter in people, the researchers analyzed large datasets from patients with melanoma and colon cancer treated with immune checkpoint blockers. Across several studies, tumors with low MHC I expression tended to contain more CD4 T cells but not more CD8 T cells, and in melanoma, patients whose MHC I–low tumors had abundant CD4 T cells lived longer. MHC I–low tumors also showed molecular signs of weaker anti‑ferroptosis defenses and stronger interferon‑gamma responses, echoing the patterns seen in mice. These findings suggest that, in some human cancers, downregulating MHC I does not simply shut off immune attack; instead, it may shift the balance toward CD4 T cell–driven, ferroptosis‑like killing.
What This Means for Future Treatments
Overall, the study reveals that MHC class I on target cells acts as more than a recognition tag for CD8 T cells; it also helps shield normal tissues and tumors from CD4 T cell–induced, iron‑dependent cell death. When this shield is lost, CD4 T cells that recognize the target through MHC II can trigger intense oxidative damage and ferroptosis, worsening transplant‑related gut injury but potentially improving control of certain cancers. For patients, this new understanding could guide strategies that intentionally harness CD4 T cells and ferroptosis to treat tumors that have lost MHC I, while also pointing to iron‑modulating drugs as a way to protect vulnerable tissues during bone marrow transplantation.
Citation: Lauder, E., Gondal, M., Wu, MC. et al. MHC class I on target cells regulates CD4+ T cell-mediated immunity. Nat Immunol 27, 1000–1012 (2026). https://doi.org/10.1038/s41590-026-02480-z
Keywords: CD4 T cells, MHC class I, ferroptosis, cancer immunotherapy, graft-versus-host disease