Clear Sky Science · en
Biallelic variants in RNU2-2 cause the most prevalent known recessive neurodevelopmental disorder
Hidden Clues in Family DNA
Some of the most serious childhood brain conditions have long remained unexplained, leaving families without clear answers or guidance. This study uncovers one of the most common inherited causes of such conditions, tracing it not to a protein-coding gene but to a tiny piece of RNA that helps cells process genetic messages. Understanding this new disorder not only offers long‑awaited diagnoses for many families, it also opens practical options for carrier testing, family planning, and earlier care for affected children. 
A Tiny RNA with a Big Job
Our cells need to edit raw genetic messages before they can be used to make proteins. This editing, called splicing, is carried out by a large machine known as the spliceosome. One of its key components is U2 small nuclear RNA, a short RNA molecule that helps recognize where pieces of genetic code should be cut and joined. The gene RNU2-2 encodes one version of this U2 RNA. Until recently, changes in this and related RNA genes were known to cause brain disorders in a dominant way—where a single faulty copy is enough to cause disease. The new work reveals that when both copies of RNU2-2 are damaged, a different, recessive form of neurodevelopmental disorder emerges, and it is surprisingly common.
Discovering a Common Inherited Brain Disorder
The researchers combed through genetic data from tens of thousands of people with rare conditions enrolled in the United Kingdom’s 100,000 Genomes Project and National Health Service genomic programs. Using a statistical tool designed to find rare, disease‑causing variants, they compared non‑protein‑coding genes in over 14,000 individuals with neurodevelopmental problems against more than 50,000 without such diagnoses. Only two genes stood out, RNU4-2 and RNU2-2, but when they looked specifically for changes that act in a recessive way—where a person must inherit one altered copy from each parent—RNU2-2 showed overwhelming evidence. They identified 18 high‑confidence families in which affected children carried two damaging RNU2-2 variants on opposite copies of the gene, plus additional candidate families and nine more cases from independent cohorts in the United States, Italy, and the Netherlands. 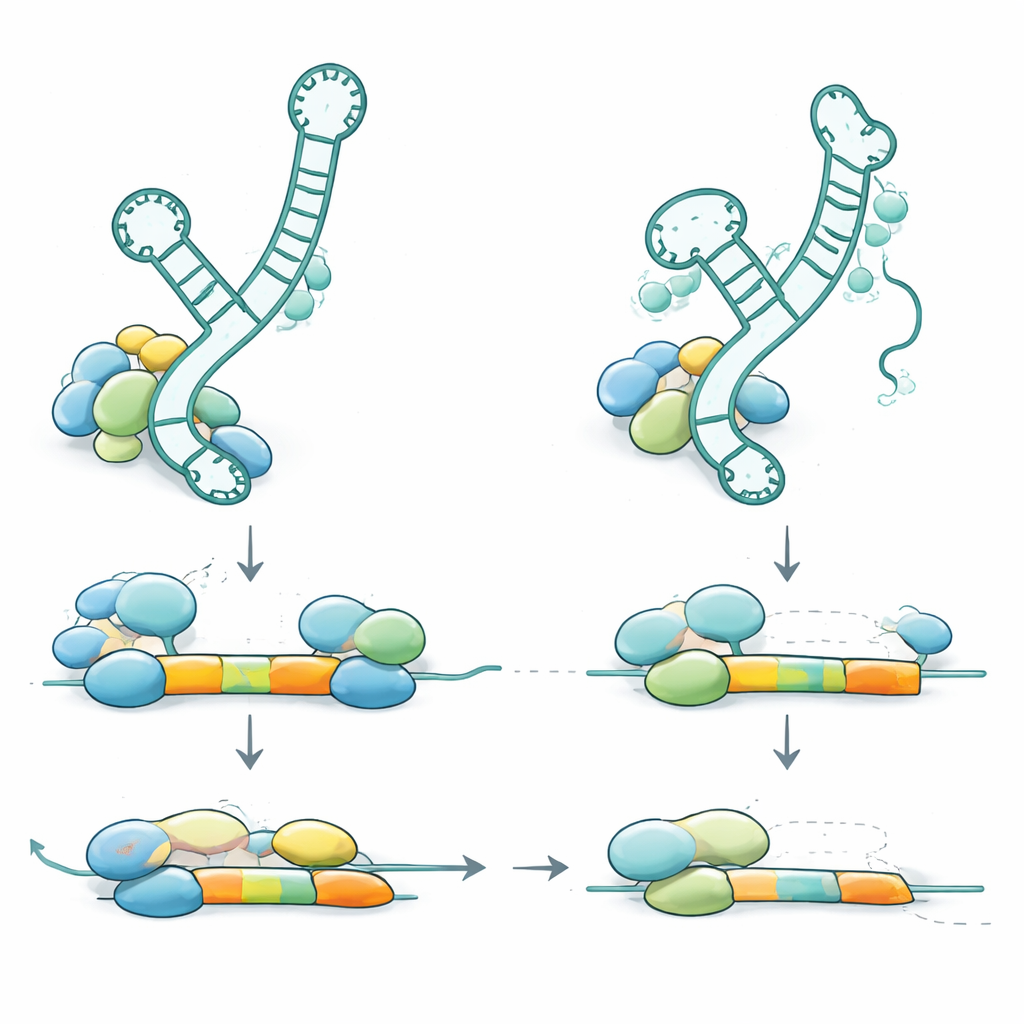
What the Condition Looks Like in Real Life
Children with this recessive RNU2-2 syndrome typically come to medical attention in infancy or early childhood. Most have delays in reaching developmental milestones, such as sitting, walking, or speaking, and many have moderate to severe intellectual disability. Seizures are very common and can begin early; in some children they evolve into difficult‑to‑treat epileptic syndromes. Muscle tone and movement are often affected, ranging from low tone in infancy to stiffness, abnormal postures, or involuntary movements later on. Brain scans may initially appear normal but can later show loss of brain tissue or changes in the white matter that connects different regions. Some individuals are mildly affected and remain relatively stable, while others develop severe complications, including problems with breathing, feeding, and, in rare cases, early death.
How Changes in RNU2-2 Disrupt Cellular Editing
To understand how these genetic changes cause disease, the team examined where the variants fall within the U2-2 RNA and how they affect its structure and behavior. Many of the recessive variants are predicted to weaken stem‑loop structures—tiny hairpins in the RNA that help it bind partner proteins and other RNAs. Others lie directly in the region that recognizes splice sites or in the docking site for a ring of helper proteins. When the researchers studied blood samples, they found that faulty RNU2-2 copies were expressed at less than 10% of normal levels in affected individuals, indicating that the altered RNA is unstable and largely destroyed. The body partly compensates by boosting a related U2 gene (RNU2-1), so total U2 levels stay roughly normal, but this compensation is not enough to prevent disease. In healthy carriers who have only one faulty copy, the damaged RNA is also strongly reduced, yet the remaining normal copy ramps up its output, keeping overall function above the threshold for symptoms.
Why This Discovery Matters for Families
Because this syndrome follows recessive inheritance, it tends to recur among siblings when both parents are healthy carriers. In the UK genome project, recessive RNU2-2 syndrome accounts for about one in ten families with a known recessive neurodevelopmental diagnosis, making it the single most frequent cause in that category and almost as common as a previously described dominant disorder caused by a different spliceosomal RNA gene. Crucially, the authors show that a simple blood RNA test can help distinguish truly harmful variants from harmless ones by measuring how much RNU2-2 is lost and how much the backup U2-1 is upregulated. For families, this means clearer diagnoses, better recurrence risk estimates, and the possibility of preconception or prenatal genetic counseling—turning an obscure piece of RNA biology into actionable information for real‑world medical decisions.
Citation: Greene, D., Mendez, R., Lees, J. et al. Biallelic variants in RNU2-2 cause the most prevalent known recessive neurodevelopmental disorder. Nat Genet 58, 774–781 (2026). https://doi.org/10.1038/s41588-026-02539-5
Keywords: neurodevelopmental disorder, recessive inheritance, spliceosome, small nuclear RNA, genome sequencing