Clear Sky Science · en
Decarboxylative alkylation of alkenes
A New Way to Tweak Everyday Molecules
Many of the products we rely on—from plastics and soaps to medicines and crop sprays—are built from simple chemical units called alkenes. Chemists would love an easy way to "plug" new carbon pieces onto these units late in the manufacturing process, letting them fine‑tune a drug or material without rebuilding it from scratch. This paper introduces a new, broadly useful method to snap alkyl building blocks onto a wide range of alkenes, opening shortcuts to molecules that were previously slow, tricky or impossible to make.
Why Changing Alkenes Has Been So Hard
Alkenes are carbon–carbon double bonds that act like tiny magnets for other chemical groups. When they encounter reactive partners, they usually add new atoms across the double bond, changing its character entirely instead of simply swapping one piece for another. In contrast, aromatic rings in dyes and pharmaceuticals can readily undergo substitution, where one hydrogen is replaced by a new group. Because alkenes lack an equivalent, general "swap‑in" reaction, chemists must often resort to longer, multi‑step routes that build the double bond only after the rest of the molecule is assembled.
Turning Simple Ingredients into Versatile Connectors
The authors tackle this problem by pairing two extremely common ingredients: alkenes and carboxylic acids. Carboxylic acids are abundant, stable and structurally diverse; they show up in natural products, drugs and bulk chemicals. The team converts these acids into special "redox‑active esters," which can lose carbon dioxide and reveal a hidden carbon fragment ready to be attached elsewhere. At the same time, they transform ordinary alkenes into "electrophilic" partners using a reagent based on thianthrene, allowing those alkenes to behave as if one of their double‑bond hydrogens were a handle for substitution rather than simple addition.
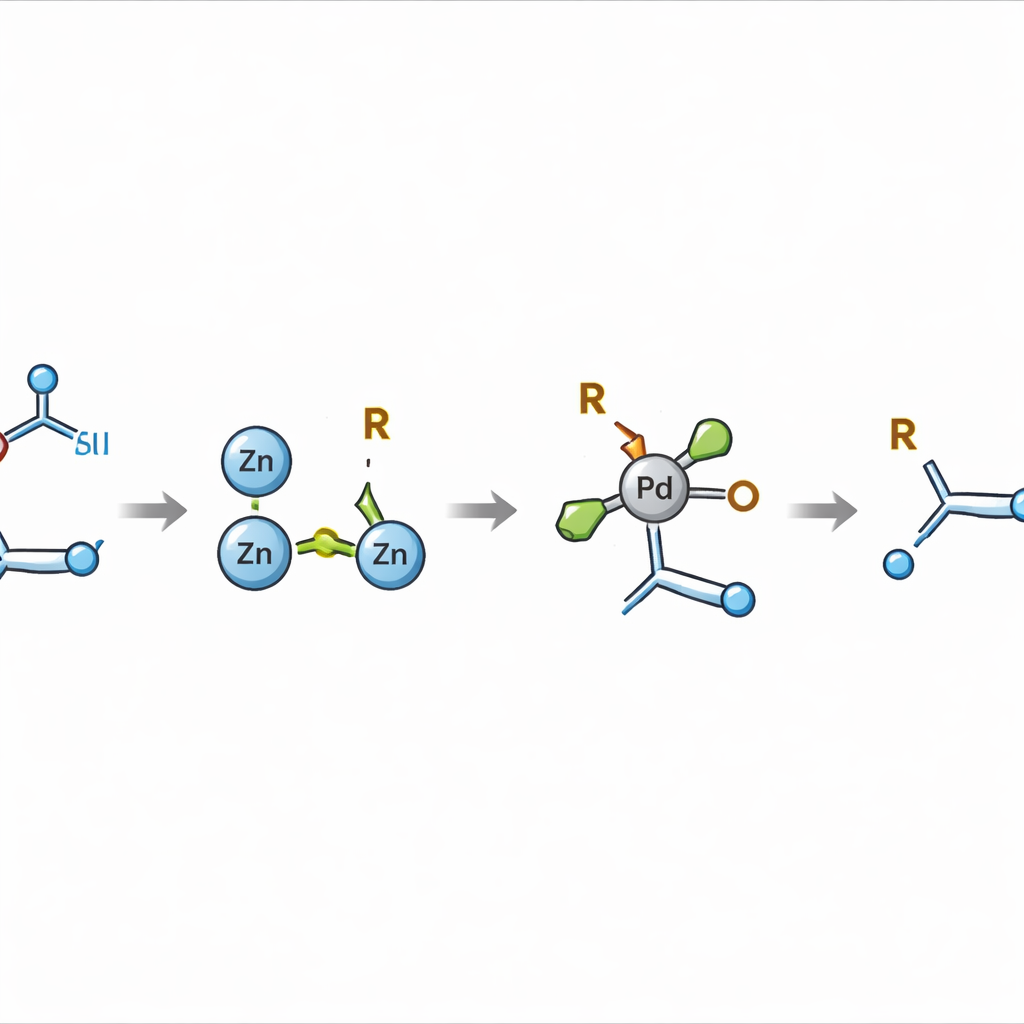
From Fleeting Radicals to Tame Metal Partners
Earlier attempts to combine these two types of components mostly relied on highly reactive radical species—short‑lived fragments that are difficult to steer and tend to produce side products instead of clean, selective bonds. The key innovation here is to divert the process away from direct radical coupling and instead create a more controlled organometallic partner: an alkyl–zinc compound. By carefully choosing the solvent and using zinc metal, the authors show that the redox‑active esters can be converted into relatively persistent alkyl–zinc intermediates. These species survive long enough to be channeled, under palladium catalysis, into a precise union with the activated alkene, forging a new bond between a carbon in the double bond and a carbon from the acid.
Reaching Difficult Molecular Targets
Because both steps—the conversion of the alkene into a thianthrenium salt and the coupling with the alkyl–zinc partner—are highly selective, the method works on many challenging structures. It can modify simple feedstock alkenes, internal and cyclic double bonds, and even tri‑substituted alkenes that are notoriously hard to alter. Sensitive groups such as aldehydes, ketones and terminal alkynes can remain untouched, making the approach suitable for late‑stage diversification of complex molecules, including terpenes and drug‑like compounds. The reaction also accepts a broad range of primary and secondary alkyl fragments, including strained rings and heterocycles, greatly expanding the menu of shapes chemists can plug onto a given alkene.
A New Logic for Building Carbon Frameworks
Beyond the immediate synthetic convenience, this work introduces a fresh design principle for making carbon–carbon bonds. Instead of planning routes around unstable radicals, chemists can think in terms of a "polar" path that turns carboxylic acids into stable metal‑bound partners, then couples them with specially activated alkenes. This polarity‑based strategy provides a new way to disconnect target molecules on paper and then assemble them in the lab. For non‑specialists, the bottom line is that the study delivers a powerful new tool for attaching carbon fragments to one of the most common motifs in organic chemistry, paving the way for faster discovery and optimization of materials, agrochemicals and medicines.
Citation: Roy, T.K., Tamborini, F.M., Petzold, R. et al. Decarboxylative alkylation of alkenes. Nature 653, 104–109 (2026). https://doi.org/10.1038/s41586-026-10463-1
Keywords: alkene functionalization, decarboxylative coupling, palladium catalysis, organometallic chemistry, carboxylic acids