Clear Sky Science · en
TBL1X/TBL1XR1 govern β-cell identity through a PAX6-containing gene regulatory network
Why Diabetes May Be a Problem of Cell Identity
Most people think of diabetes as a disease of blood sugar, but deep inside the pancreas the story is really about identity. Specialized beta cells normally sense rising sugar and release insulin. In type 2 diabetes, many of these cells lose their mature character and stop doing their job properly. This study uncovers a previously missing layer of control that helps beta cells stay true to their identity, and shows how its failure can tip the body toward diabetes.
The Guardians Inside Beta Cells
Beta cells live in tiny clusters in the pancreas called islets and are responsible for making and secreting insulin. Their identity is enforced by “master switches” in the DNA called transcription factors, which turn the right genes on and the wrong ones off. But these master switches do not act alone; they rely on helper proteins that fine‑tune when genes are activated or silenced. The authors focus on two such helpers, TBL1X and TBL1XR1 (together called TBL/R1), asking whether they are essential for keeping beta cells functional and whether they matter for human diabetes.
What Happens When the Guardians Are Removed
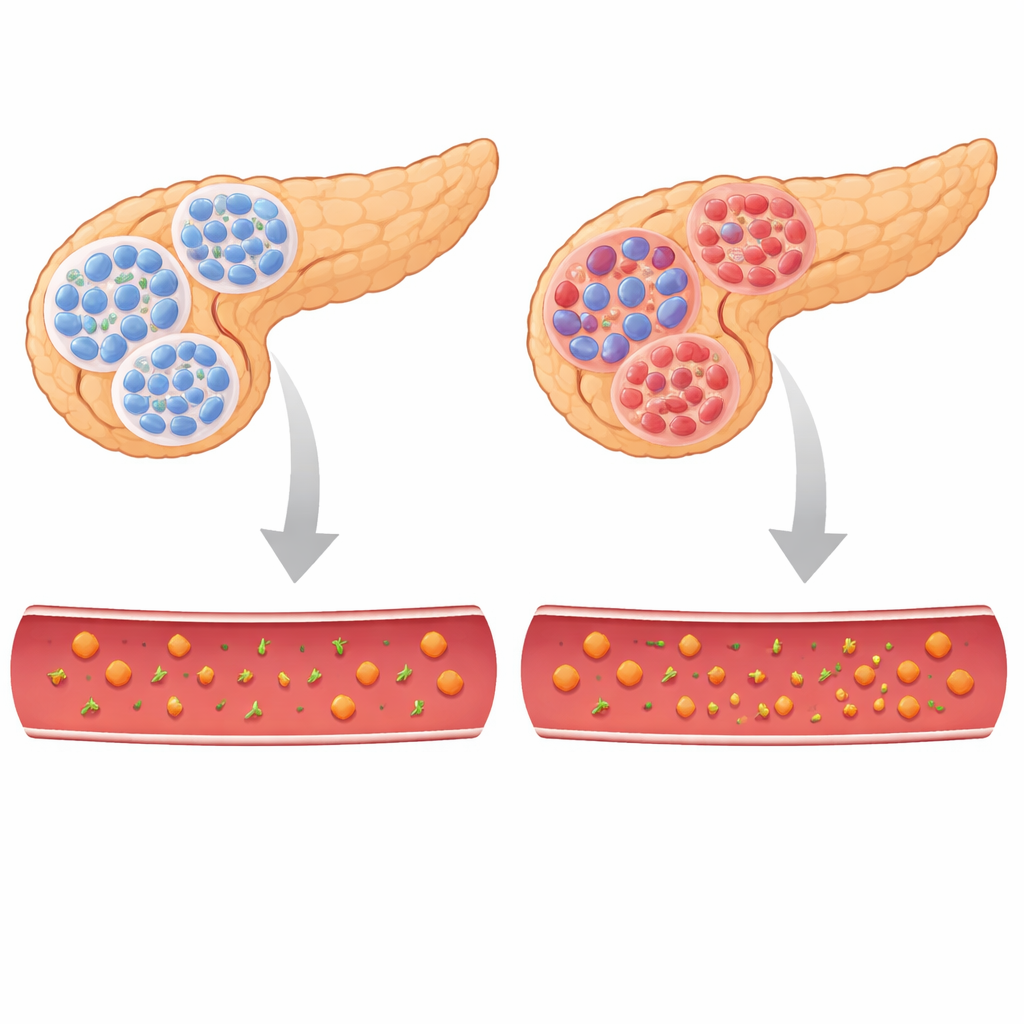
To test TBL/R1’s role, the researchers engineered mice in which these helpers were deleted only in beta cells. At first, young animals looked normal, but soon they developed very high blood sugar and lost weight, a classic diabetes‑like picture. Their bodies were not resistant to insulin; rather, their pancreases simply did not make enough. Measurements showed low insulin in the blood and reduced insulin stores in the pancreas, while levels of the hormone glucagon, which raises blood sugar, were increased. Even before blood sugar rose, the structure of islets was already disturbed: beta cells were fewer, alpha cells were scattered throughout the islets, and the balance between these cell types resembled that seen in diabetic animals.
Cells That Forget Who They Are
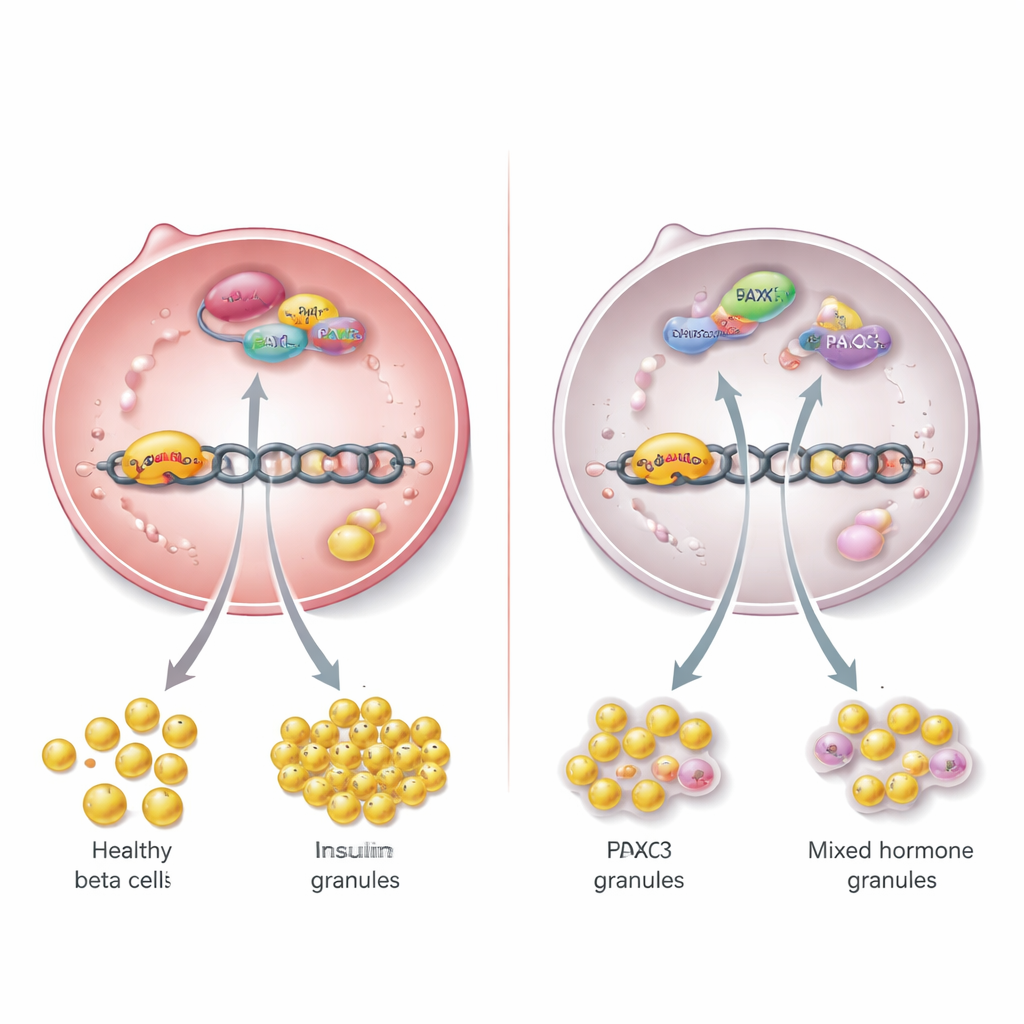
Looking more closely at gene activity in the islets, the team found sweeping changes. Thousands of genes shifted their behavior when TBL/R1 were missing. Key markers of mature beta cells, including the genes that encode insulin and proteins needed for sensing and handling glucose, were dialed down. At the same time, genes usually “disallowed” in healthy beta cells, as well as markers of immature or alternative islet cell types, were turned up. Single‑cell RNA sequencing, which profiles individual cells, confirmed that genuine beta cells were being lost and replaced by less mature cells and rare “polyhormonal” cells that produced mixtures of hormones. These patterns indicate that without TBL/R1, beta cells do not necessarily die; instead, they drift away from their specialized identity toward a more primitive or mixed state that cannot sustain normal insulin output.
A Molecular Teamwork Between PAX6 and TBL/R1
To understand the underlying mechanism, the researchers mapped which nuclear proteins physically interact with TBL/R1 in beta‑cell lines. Among many partners, they identified PAX6, a well‑known regulator of islet development and function. They showed that PAX6 and TBL/R1 both sit on the insulin gene’s control region together with another protein, HDAC3, forming a regulatory team directly on the DNA. When TBL/R1 were experimentally reduced, PAX6 could no longer boost insulin gene activity, and insulin production and release dropped. Inhibiting HDAC3 also weakened insulin gene activation in a way that depended on TBL/R1, suggesting that this trio works as a switch that promotes insulin gene expression while helping to keep other, inappropriate genes in check.
From Mouse Mechanism to Human Risk
Importantly, this regulatory system is not restricted to rodents. In a human beta‑cell line, TBL1X, TBL1XR1, and PAX6 were all found at the human insulin gene and formed similar complexes with HDAC3. Reducing TBL/R1 in these cells lowered insulin gene activity and insulin secretion. The team then looked at human donor islets and found that lower TBL1X expression was associated with higher long‑term blood sugar levels, measured by HbA1c. Large‑scale genetic studies in tens of thousands of people further showed that common DNA variants near the TBL1X and TBL1XR1 genes are linked to higher HbA1c and random blood glucose, tying this regulatory layer to diabetes risk in the population.
Why These Findings Matter for Future Therapies
Taken together, the study reveals that TBL1X and TBL1XR1 act as crucial guardians of beta‑cell identity by partnering with PAX6 and associated complexes to keep insulin genes active and inappropriate genes silent. When this control layer falters, beta cells progressively forget who they are, leading to too little insulin and rising blood sugar. Because loss of beta‑cell identity can be reversible, targeting this TBL/R1‑centered network—either to protect existing beta cells or to improve the maturation of lab‑grown cells for transplantation—may open new, more precise treatment strategies for type 2 diabetes.
Citation: Walth-Hummel, A.A., Jouffe, C., Weber, P. et al. TBL1X/TBL1XR1 govern β-cell identity through a PAX6-containing gene regulatory network. Nat Commun 17, 3736 (2026). https://doi.org/10.1038/s41467-026-72077-5
Keywords: beta cell identity, insulin regulation, transcriptional cofactors, type 2 diabetes, PAX6 network