Clear Sky Science · en
Neoadjuvant palbociclib and endocrine therapy versus chemotherapy in ER + /HER2- breast cancer: a randomized phase II trial
Why this breast cancer study matters
For many people with hormone-sensitive breast cancer, the first major treatment decision is whether to receive traditional chemotherapy or newer targeted pills before surgery. Chemotherapy can be effective but often comes with taxing side effects, while pill-based treatments aimed at blocking tumor growth may be gentler but are not yet routine in this early setting. This study asks a simple yet crucial question: can we safely use targeted pills instead of, or in a different order than, chemotherapy—and can modern genetic tests on the tumor tell us who really needs which treatment?
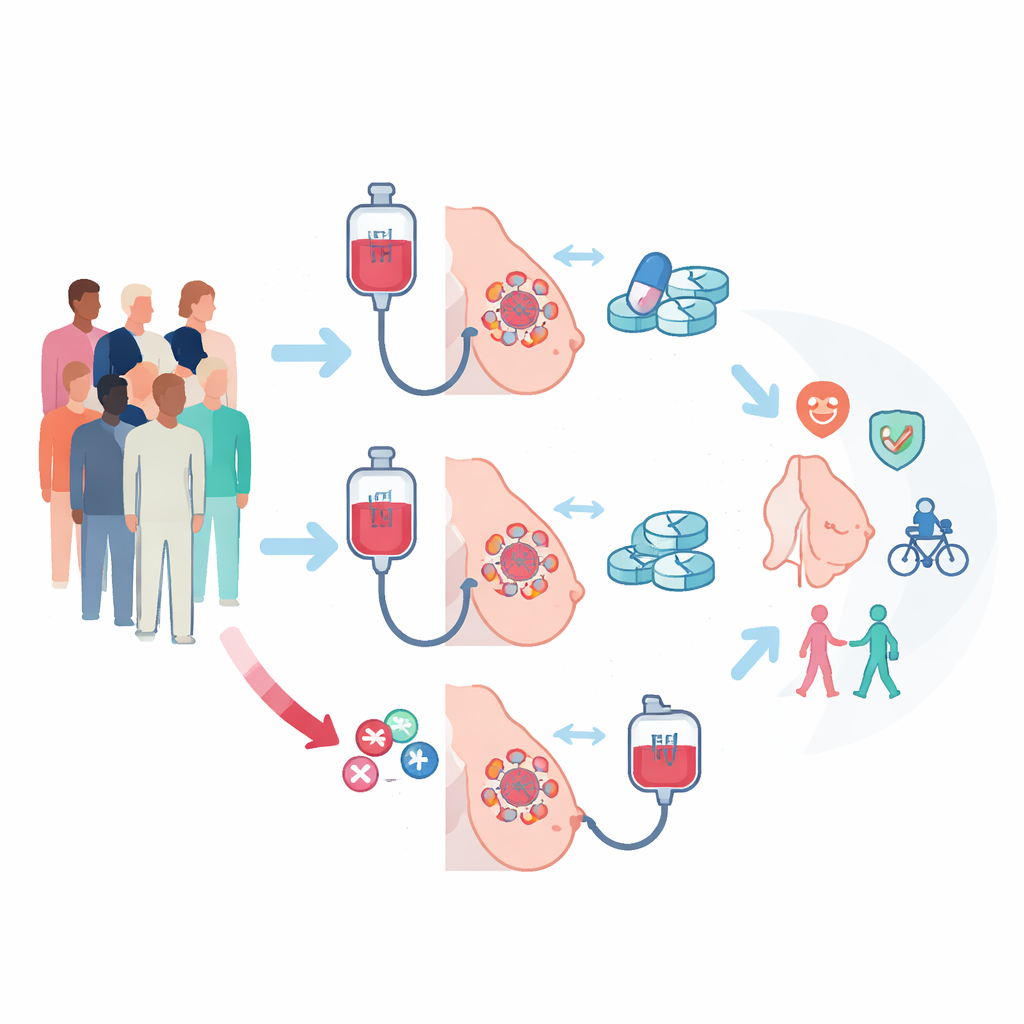
Two different roads before surgery
The PREDIX LumB trial enrolled 179 people with a common form of breast cancer that is sensitive to estrogen but does not overproduce the HER2 protein (ER-positive/HER2-negative). All tumors were fairly large and/or had spread to nearby lymph nodes, so shrinking them before surgery was medically important. Participants were randomly assigned to one of two paths. In one arm, they first received standard chemotherapy with weekly paclitaxel for 12 weeks, followed by 12 weeks of a targeted pill called palbociclib combined with hormone-blocking therapy. In the other arm, the order was reversed: palbociclib plus hormone therapy first, then paclitaxel. After these 24 weeks of pre-surgery treatment, everyone went on to standard surgery and additional therapy as recommended.
Similar tumor shrinkage and survival, different day‑to‑day impact
The main yardstick was how many patients had their tumors clearly shrink on scans after the first 12 weeks. Chemotherapy first led to responses in 59% of patients, while starting with the pill plus hormone therapy led to responses in 45%; this difference did not reach the level researchers consider statistically firm. By 24 weeks—after everyone had received both types of treatment in opposite order—response rates were high and similar in both groups. Longer-term outcomes also lined up: event-free survival, relapses after surgery, and overall survival over about four and a half years were essentially the same regardless of which treatment came first. Side effects, however, differed in character. Chemotherapy more often caused nerve damage in the hands and feet, rash, and fatigue, while palbociclib commonly led to drops in white blood cells. Patients reported that their quality of life was better preserved during the pill-based phase and dipped more during chemotherapy, although most measures returned to baseline about a year after surgery.
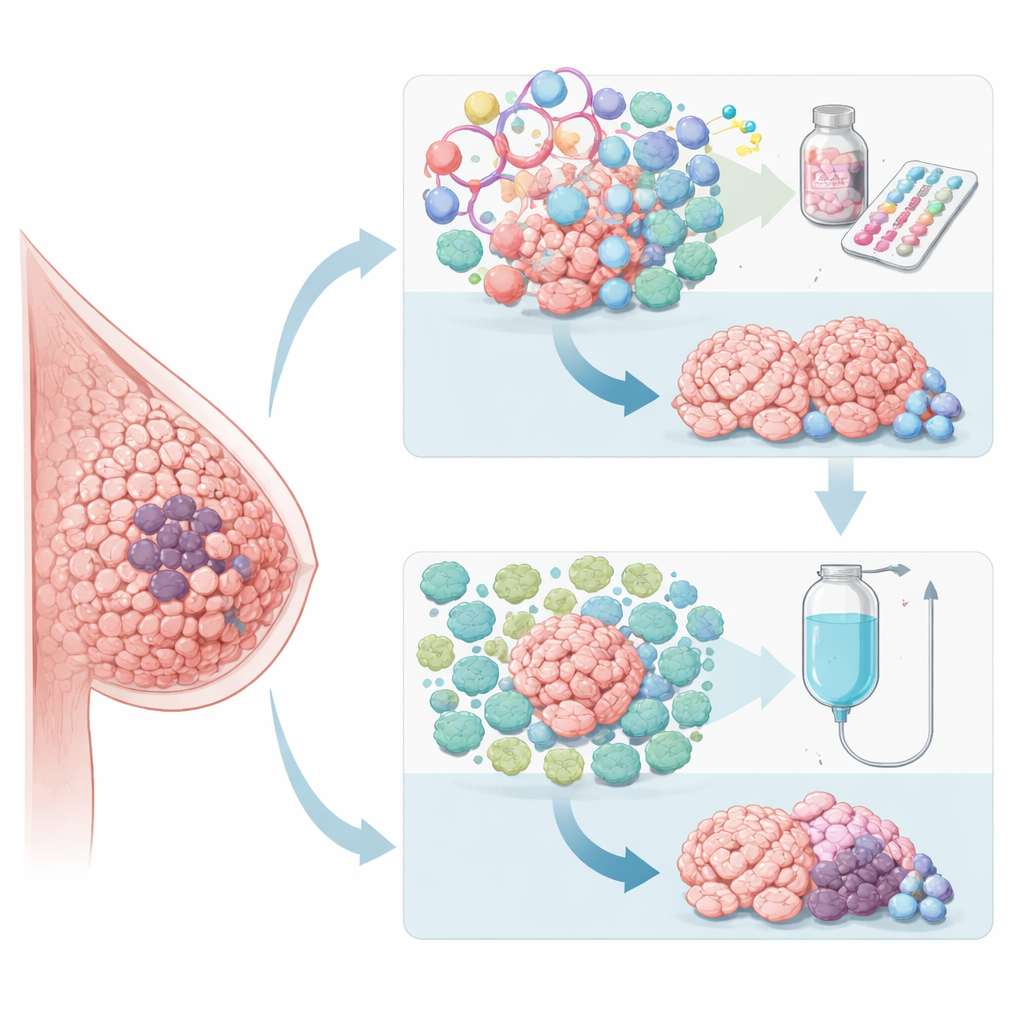
Looking inside tumors for hidden clues
Because standard measures like tumor grade and routine lab tests were not enough to guide who should get which drug first, the researchers turned to deep analyses of tumor tissue. They used whole-exome and RNA sequencing to profile thousands of genes before treatment and linked these patterns to who responded to which therapy. Tumors that did well with paclitaxel chemotherapy tended to show stronger signs of immune activity and weaker estrogen-related signals. In contrast, tumors that responded better to palbociclib plus hormone therapy showed high activity in genes tied to cell division and estrogen signaling, but relatively muted immune signatures. These observations suggested that the biological “personality” of the cancer—not just its size and stage—shapes how it reacts to different treatments.
A new genetic guide for treatment choice
Based on these patterns, the team built a new genetic tool called CDKPredX. It combines 31 genes into three simple themes: how fast the tumor cells are dividing, how strongly they depend on estrogen, and how active the local immune response is. Tumors flagged as CDKPredX-positive were highly proliferative, strongly hormone-driven, and had low immune activity. In the PREDIX LumB trial, this group was less likely to benefit from chemotherapy but still responded to palbociclib plus hormone therapy. The researchers then tested CDKPredX in other clinical studies, including the CORALLEEN trial and large external datasets. Across these independent groups, CDKPredX repeatedly identified patients who gained little from chemotherapy yet remained sensitive to CDK4/6 inhibitors like palbociclib. At the same time, the signature did not simply forecast who would do better overall; instead, it specifically highlighted which treatment type was more likely to work.
What this means for people with breast cancer
This study shows that, on average, giving chemotherapy or targeted palbociclib plus hormone therapy in different sequences before surgery leads to similar tumor shrinkage and long-term outcomes for this kind of breast cancer. However, it also reveals that the cancers themselves are far from uniform. By reading the genetic “fingerprint” of each tumor, tools like CDKPredX may soon help doctors identify patients who could safely avoid the harshest effects of chemotherapy without sacrificing effectiveness, and others who truly need chemotherapy’s punch. While CDKPredX still needs prospective testing before it can guide routine care, this work marks an important step toward more personalized, less one-size-fits-all treatment for people with hormone-sensitive breast cancer.
Citation: Matikas, A., Tzoras, E., Sarafidis, M. et al. Neoadjuvant palbociclib and endocrine therapy versus chemotherapy in ER + /HER2- breast cancer: a randomized phase II trial. Nat Commun 17, 3403 (2026). https://doi.org/10.1038/s41467-026-71452-6
Keywords: ER-positive breast cancer, CDK4/6 inhibitors, neoadjuvant therapy, chemotherapy resistance, tumor gene profiling