Clear Sky Science · en
Immune-induced TCR-like antibodies regulate specific T cell response in mice
Why some immune responses quietly turn themselves down
Our immune system must strike a delicate balance: it has to attack invaders like viruses and bacteria, yet avoid damaging the body’s own tissues. This paper reveals a previously overlooked self‑braking feature inside that system—special antibodies that recognize the same molecular targets as certain T cells and can selectively calm them down. Understanding this built‑in brake may open new, highly targeted ways to treat autoimmune diseases without broadly weakening immunity.
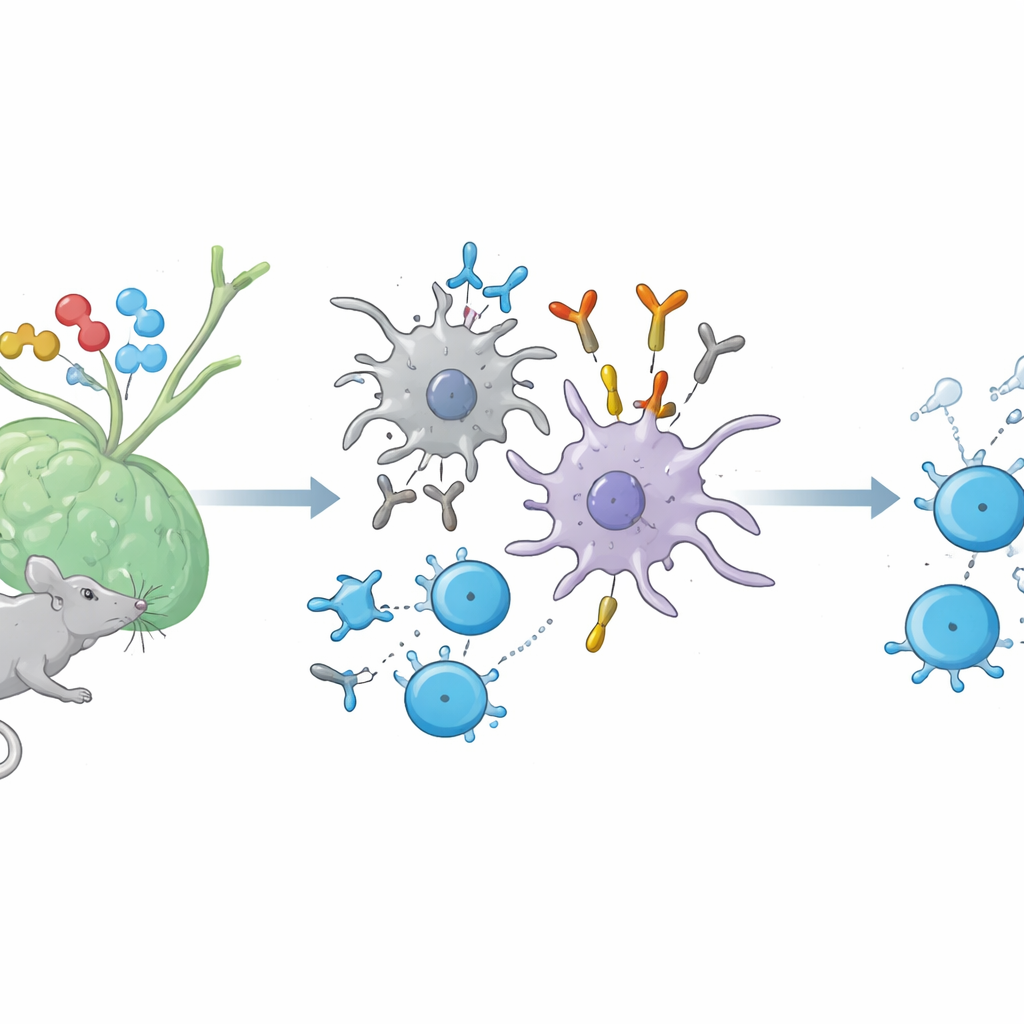
A hidden conversation between antibodies and helper T cells
When we are immunized with a protein, two major forces are set in motion. B cells make antibodies against the foreign protein, while helper T cells recognize tiny fragments of that protein displayed on the surface of immune cells by molecules called MHC class II. Until now, scientists believed that antibodies which recognize this combined "fragment plus MHC" structure—much like a T cell receptor does—only appeared in artificial lab setups. In mice, the authors show that such TCR‑like antibodies are in fact generated naturally during ordinary immune responses to several proteins, including hen egg lysozyme, ovalbumin, and a nerve‑sheath protein related to multiple sclerosis.
How peptide “tails” switch on these special antibodies
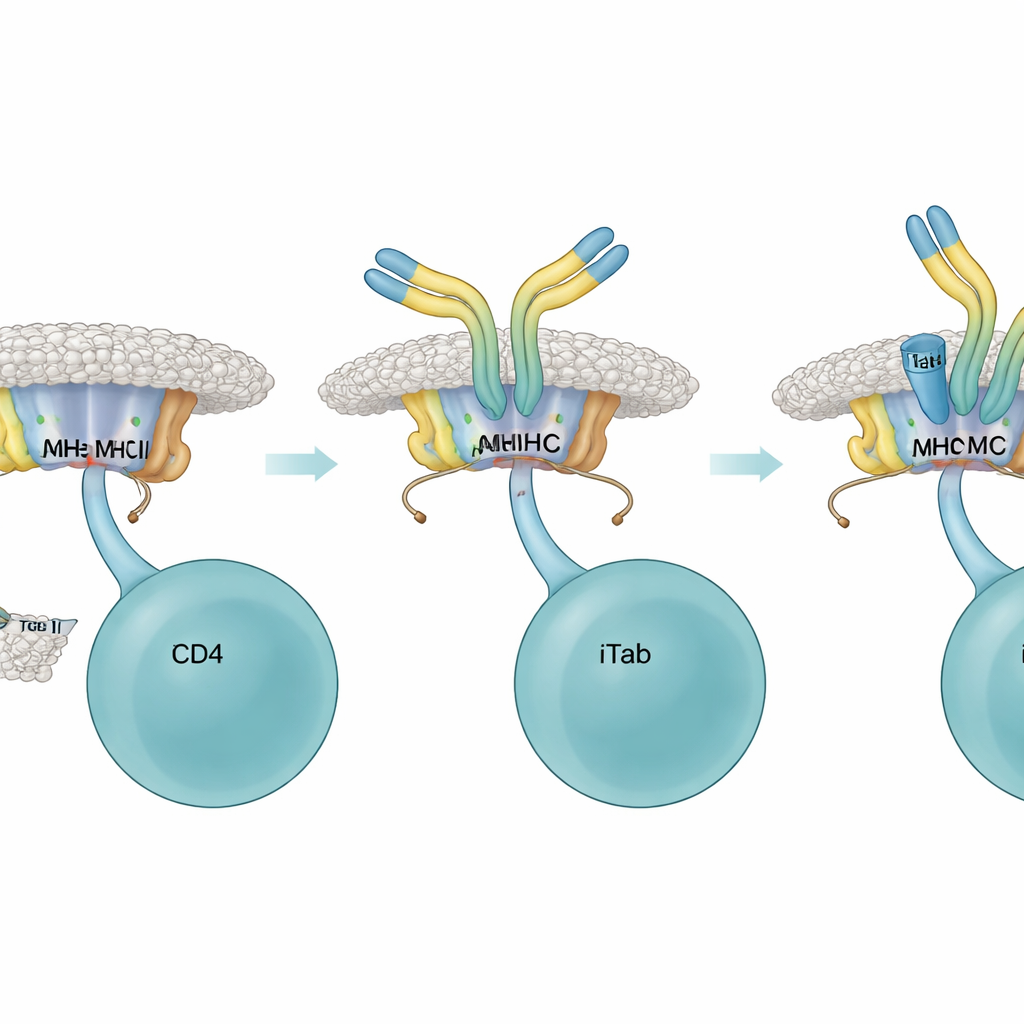
To understand when these unusual antibodies arise, the researchers compared immune responses to short peptide fragments versus slightly longer versions that carry extra amino‑acid "tails" at one end, known as flanking residues. Both versions can be shown by MHC class II and seen by T cells, but they differ in how much extra sequence hangs off the ends. The team found that only peptides with such flanking residues reliably triggered the appearance of the new antibodies, which they named immune‑induced TCR‑like antibodies, or iTabs. These iTabs did not simply recognize the peptide or the MHC molecule alone; they recognized the precise three‑dimensional combination of a specific peptide, its flanking stretch, and a particular MHC type—mirroring the fine specificity of a T cell receptor.
Antibody brakes on overactive helper T cells
Once present, iTabs acted as highly selective brakes on helper T cells. In cell‑culture assays, iTabs bound to peptide–MHC complexes and blocked T cell receptor fusion proteins and reporter T cells from engaging their targets. They also reduced production of IL‑2, a key T cell growth signal, and dampened a classic T‑cell–driven skin swelling response in living mice. Structural work using high‑resolution cryo‑electron microscopy revealed why: an iTab’s binding region clamps onto both the MHC surface and the peptide’s tail, creating a snug fit that physically interferes with T cell access. Some iTabs could also mark antigen‑presenting B cells for destruction through standard antibody “tail” functions, adding another route to quiet an ongoing response.
Turning iTabs into tools against autoimmunity
Armed with this knowledge, the researchers asked whether iTabs could be harnessed to tame harmful self‑reactive T cells in a mouse model of multiple sclerosis called experimental autoimmune encephalomyelitis. They designed peptides from a myelin protein that included flanking residues and, in some versions, subtle changes that prevent them from activating disease‑causing T cells while still allowing iTab induction. Immunizing mice with these tailored peptides generated iTabs that recognized disease‑relevant peptide–MHC complexes and blocked most of the pathogenic T cell receptors tested. Treating mice with such iTabs, or pre‑immunizing them with iTab‑inducing peptides before disease was triggered, led to milder symptoms and delayed or reduced disease progression, even though overall antibody levels against the peptide were similar.
What this means for future precision therapies
Overall, the study uncovers iTabs as a natural, transient layer of immune regulation: antibodies that mimic T cell receptors, latch onto the exact peptide–MHC complexes recognized by certain helper T cells, and selectively tone down those responses. By carefully choosing peptides with the right flanking sequences, it may be possible to deliberately induce iTabs that target only harmful autoreactive T cells, leaving protective immunity intact. While these findings are in mice and human translation will be complex, they sketch a path toward “designer brakes” for autoimmune and allergic diseases—treatments that work not by broadly suppressing the immune system, but by quietly blocking the very molecular conversations that drive unwanted attacks.
Citation: Kishida, K., Kawakami, K., Tanabe, H. et al. Immune-induced TCR-like antibodies regulate specific T cell response in mice. Nat Commun 17, 3227 (2026). https://doi.org/10.1038/s41467-026-71384-1
Keywords: autoimmunity, helper T cells, MHC class II, regulatory antibodies, peptide flanking residues