Clear Sky Science · en
Molecular and functional dissection using CaMPARI-seq reveals the neuronal organization for dissociating optic flow-dependent behaviors
How the Brain Tells Turning from Straight Swimming
When animals move, the whole visual world seems to stream past their eyes. From this constantly shifting scene, the brain must figure out a simple but vital question: are we turning, or moving straight ahead? This paper uses tiny zebrafish and a new molecular "highlighter" technique to reveal how specific brain cells separate these two kinds of motion to control either eye movements or whole-body swimming.
Seeing Motion as a Flowing Landscape
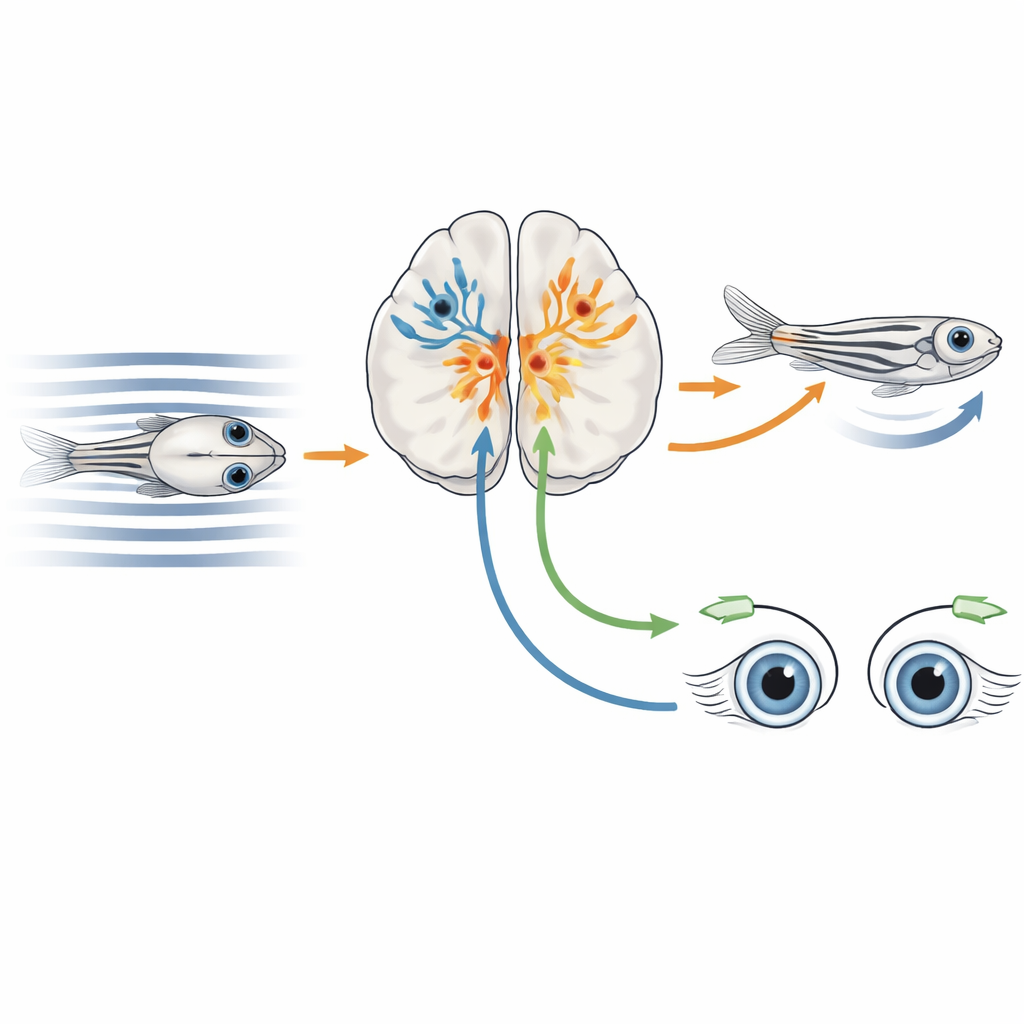
As an animal swims or walks, patterns of light sweep across the eyes, a phenomenon called optic flow. Rotational flow, as when an animal turns its head, should mainly drive eye movements that stabilize the gaze. Translational flow, as when the animal swims forward, should instead drive the body to keep moving with the surrounding water or ground. In many vertebrates, including zebrafish, a midbrain region called the pretectum plays a central role in interpreting these motion patterns and routing signals to motor centers. Previous work had mapped many pretectal neurons that respond to optic flow, but it remained unclear which precise cell types, defined by their gene expression and wiring, were responsible for different motion-driven behaviors.
Lighting Up Active Neurons and Reading Their Genes
The researchers developed a hybrid approach they call CaMPARI-seq, combining optical tagging of active neurons with single-cell gene expression profiling. They engineered zebrafish larvae so that most neurons produced a special fluorescent protein, CaMPARI2, targeted to the cell nucleus. When neurons are active and illuminated with ultraviolet light, this protein permanently switches from green to red. The team exposed the fish to moving stripe patterns that create a range of binocular optic flow conditions, while shining ultraviolet light on the pretectum. Active, motion-responsive neurons turned red, were later isolated one by one, and their RNA sequences were read to determine which genes each cell expressed.
Building a Cellular Atlas of Optic-Flow Neurons
By clustering thousands of tagged cells based on their gene expression patterns, the authors identified a major pretectal group marked by the gene tcf7l2, most of whose cells also carried genes typical of inhibitory neurons. Using genetically engineered fish in which tcf7l2-positive cells drove a calcium indicator, they verified that this broad population included nearly all the previously described optic-flow response types, from neurons tuned to motion seen by just one eye to others that responded only when both eyes sensed coherent forward or backward motion. Further dividing the tcf7l2 group revealed seven molecularly distinct subtypes, each with its own combination of marker genes and a largely non-overlapping spatial position within the pretectum, suggesting a patchwork of specialized inhibitory circuits layered around the motion-processing hub.
Two Key Subtypes with Very Different Jobs
Among these subtypes, two stood out. Cells expressing the gene mafaa sat in a lateral migrated region that overlaps with incoming direction-selective retinal fibers. Imaging and anatomical tracing showed that these neurons make local connections and respond strongly when stripes move in a particular direction in just one eye, matching a role in encoding simple, eye-specific motion that can support turning of the eyes. In contrast, neurons expressing nkx1.2lb occupied a more medial zone and sent long, crossed projections through a dorsal brain commissure to the opposite side, forming a bridge between left and right pretectal circuits. These nkx1.2lb-positive cells were also mostly inhibitory, and together they covered a wide variety of optic-flow response types, including both monocular and binocular patterns, hinting that they help compare signals from the two eyes.
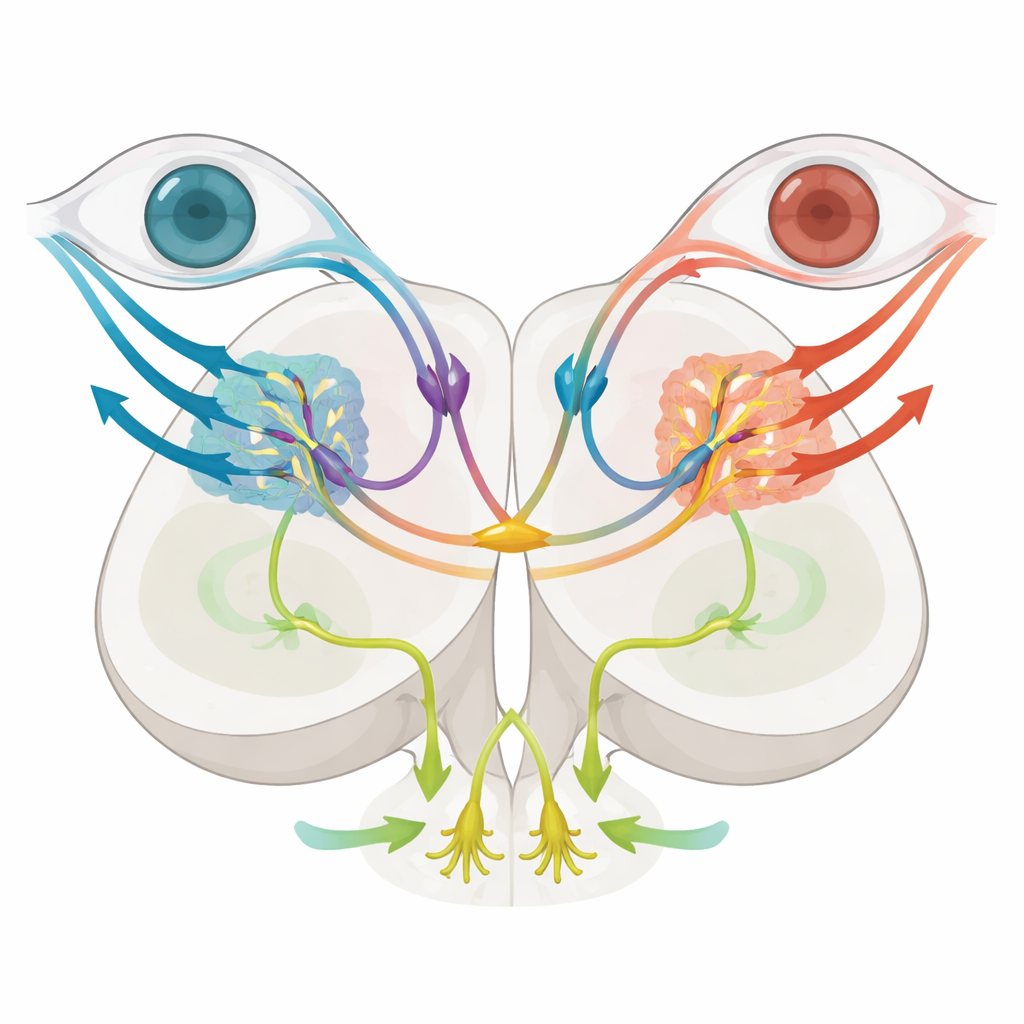
Disentangling Eye and Body Responses
To test function, the team selectively eliminated nkx1.2lb-positive pretectal neurons using a genetic "suicide" enzyme that becomes toxic only in those cells when the fish are given a harmless drug. After ablation, larvae still showed normal eye-tracking movements when the world rotated around them, indicating that rotational optic flow and the resulting optokinetic response remained intact. However, when the ground pattern moved to mimic forward swimming, the optomotor response was severely weakened: fish traveled shorter distances and their tail beats became less coordinated, showing disrupted computation of swim direction. Brain imaging revealed that cells tuned specifically to translational optic flow were reduced, while basic monocular motion detectors were largely preserved. Together, these experiments suggest that the commissural nkx1.2lb neurons are essential for integrating signals from both eyes to estimate straight-ahead motion and drive forward swimming, but are not required for stabilizing gaze during turning.
Why This Matters for Understanding Motion Sensing
To a non-specialist, the key message is that the brain separates "am I turning?" from "am I moving forward?" by assigning these questions to different sets of cells, even within a tiny fish. The authors show that a specific inhibitory bridge between the left and right sides of the pretectum is crucial for recognizing translational motion and steering the body, while other circuits can handle rotational motion and eye movements without this bridge. Their CaMPARI-seq method—using light to tag active cells and then reading their molecular identity—offers a powerful way to link what neurons do, how they are built, and which behaviors they control. Insights from this compact zebrafish system could help clarify how larger vertebrate brains, including our own, keep vision stable and navigation accurate in a moving world.
Citation: Matsuda, K., Wang, CH., Kakinuma, H. et al. Molecular and functional dissection using CaMPARI-seq reveals the neuronal organization for dissociating optic flow-dependent behaviors. Nat Commun 17, 3411 (2026). https://doi.org/10.1038/s41467-026-71371-6
Keywords: optic flow, zebrafish, pretectum, single-cell RNA sequencing, visual motion processing