Clear Sky Science · en
DC-CD4 bispecific tolerogenic nanovesicles induce antigen-specific regulatory T cells and ameliorate collagen-induced arthritis in mice
Teaching the Immune System When Not to Attack
Autoimmune diseases occur when the body’s defense system mistakenly turns on its own tissues, leading to chronic pain and damage. Instead of broadly shutting down immunity, scientists are searching for ways to retrain these defenses so they ignore specific harmless targets while still fighting real threats. This study in mice describes a tiny, engineered package that aims to do just that, hinting at more precise treatments for conditions like rheumatoid arthritis in the future. 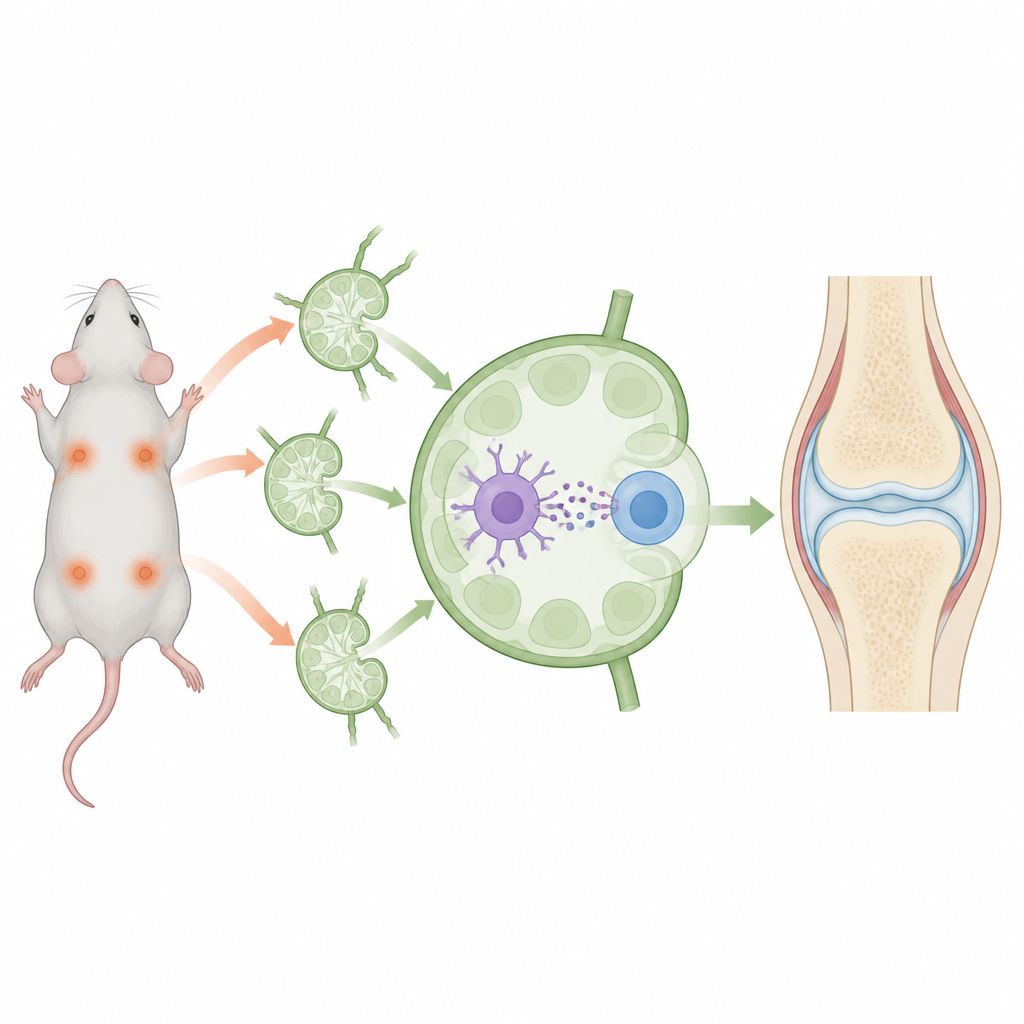
Why Turning Down the Right Cells Matters
A healthy immune system can tell the difference between “self” and “invader,” a balance known as immune tolerance. Special white blood cells called regulatory T cells act as peacekeepers, calming harmful reactions before they spiral into disease. Doctors have tried to boost these cells either by growing them outside the body and infusing them back, or by using drugs that favor their growth inside the body. Both paths have drawbacks, including complex cell handling and the risk of blunting immunity too broadly. The goal is to increase only those regulatory cells that recognize the troublesome self-molecules linked to a particular disease.
Designing Tiny Peacekeeping Packages
The researchers built nanosized bubbles, or nanovesicles, from the outer membranes of immune cells that already have calming features. These vesicles were loaded with two key ingredients: a short fragment of collagen, a joint protein targeted in arthritis, and the drug rapamycin, which encourages a more tolerant immune state. The nanovesicles were then “decorated” so they could latch onto two different partners in lymph nodes: dendritic cells, which present antigens, and CD4 T cells, which can become either aggressive fighters or regulatory peacemakers. By physically bringing these two cell types together around the same collagen fragment and drug, the vesicles were designed to nudge the conversation toward tolerance rather than attack.
Guiding Immune Traffic to the Right Place
When injected into the skin of mice, the nanovesicles drained into nearby lymph nodes, the hubs where immune decisions are made. There, they attached strongly to both dendritic cells and CD4 T cells, effectively serving as bridges between them. Compared with free drug and collagen, the vesicles delivered much more material into dendritic cells and greatly boosted how well these cells displayed the collagen fragment. At the same time, they shifted both dendritic cells and CD4 T cells toward a quieter, more regulatory profile, with fewer inflammatory signals and more calming ones such as TGF-beta and IL-10. This setting favored the growth of collagen-specific regulatory T cells that would later recognize and restrain immune attacks focused on joints.
Protecting Joints in an Arthritis Model
To test whether this immune re-education mattered in disease, the team used a standard mouse model of collagen-induced arthritis, which mimics many features of human rheumatoid arthritis. Mice treated with collagen-loaded nanovesicles developed far milder joint swelling, retained more normal movement, and lost less weight than untreated animals. Detailed imaging and tissue slices showed less joint erosion, fewer signs of active inflammation, and reduced scarring in treated mice. Importantly, regulatory T cells increased not only in lymph nodes and spleen but also within the joint lining itself, while damaging inflammatory molecules dropped. Versions of the nanovesicles that lacked the correct antigen, or that could not bridge both cell types effectively, provided weaker protection, underscoring the importance of both antigen choice and cell-to-cell linking. 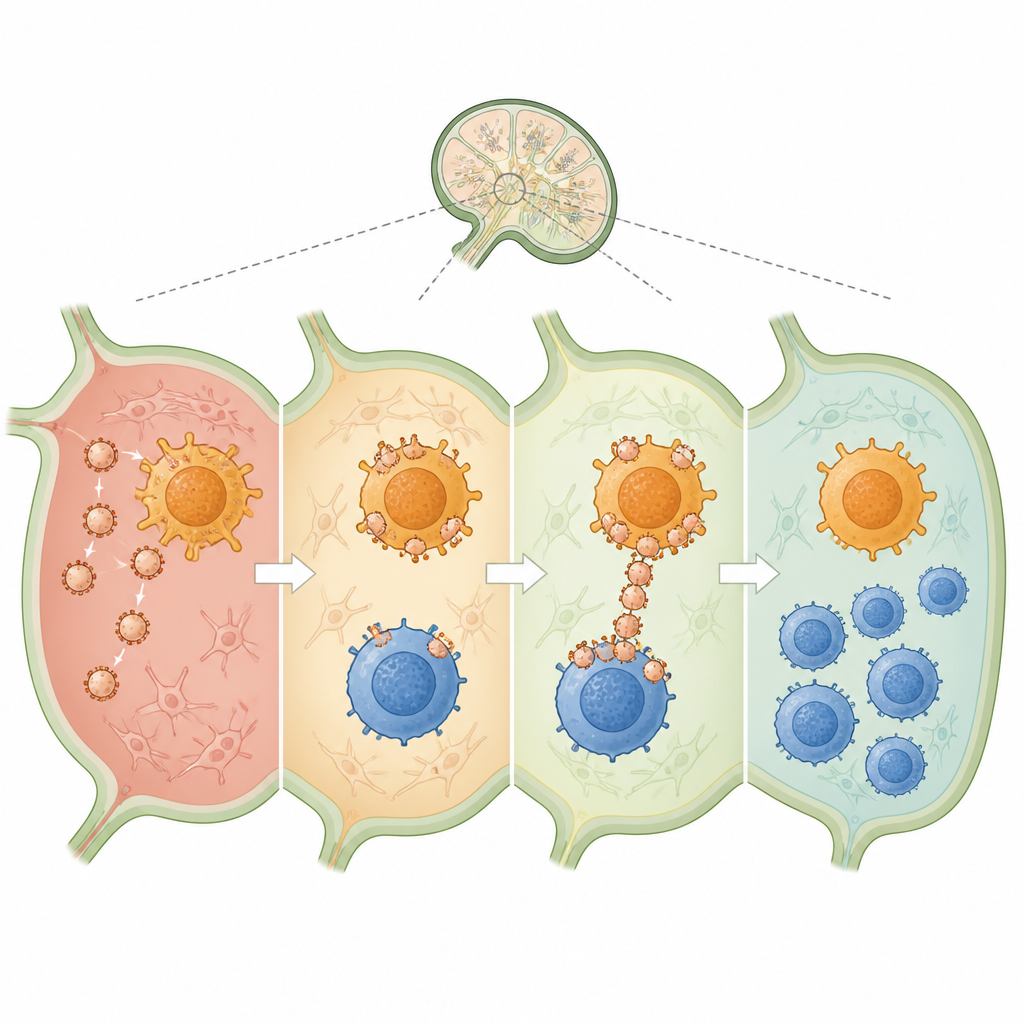
Lasting Calm and What Comes Next
The researchers further showed that the regulatory T cells sparked by these nanovesicles could transfer protection to other mice and help resist a later flare of arthritis, suggesting that the immune system “remembers” the lesson. While the work is still limited to mice and one arthritis model, it points toward a flexible platform in which disease-specific self-fragments could be swapped in to retrain immunity in different autoimmune conditions. For patients, this approach aims not to silence the immune system entirely, but to teach it to stand down only where it is doing harm.
Citation: Zhao, L., Gao, Z., Yuan, Z. et al. DC-CD4 bispecific tolerogenic nanovesicles induce antigen-specific regulatory T cells and ameliorate collagen-induced arthritis in mice. Nat Commun 17, 4170 (2026). https://doi.org/10.1038/s41467-026-70898-y
Keywords: autoimmune tolerance, regulatory T cells, nanovesicles, rheumatoid arthritis, immune therapy