Clear Sky Science · en
Tumor-specific lncRNA IGF1R-AS1 trans-regulates chromatin interactions associated with oncogenic MYC signaling
How a Hidden RNA Switch May Fuel Certain Cancers
Why do some prostate and lung cancers grow so aggressively, and can we find switches that exist only in tumor cells to control them? This study uncovers a previously unrecognized piece of RNA that is almost exclusive to tumors and appears to help flip on MYC, one of the most powerful growth drivers in cancer. Understanding how this RNA works could open new ways to detect and possibly disarm dangerous cancers without harming healthy tissue. 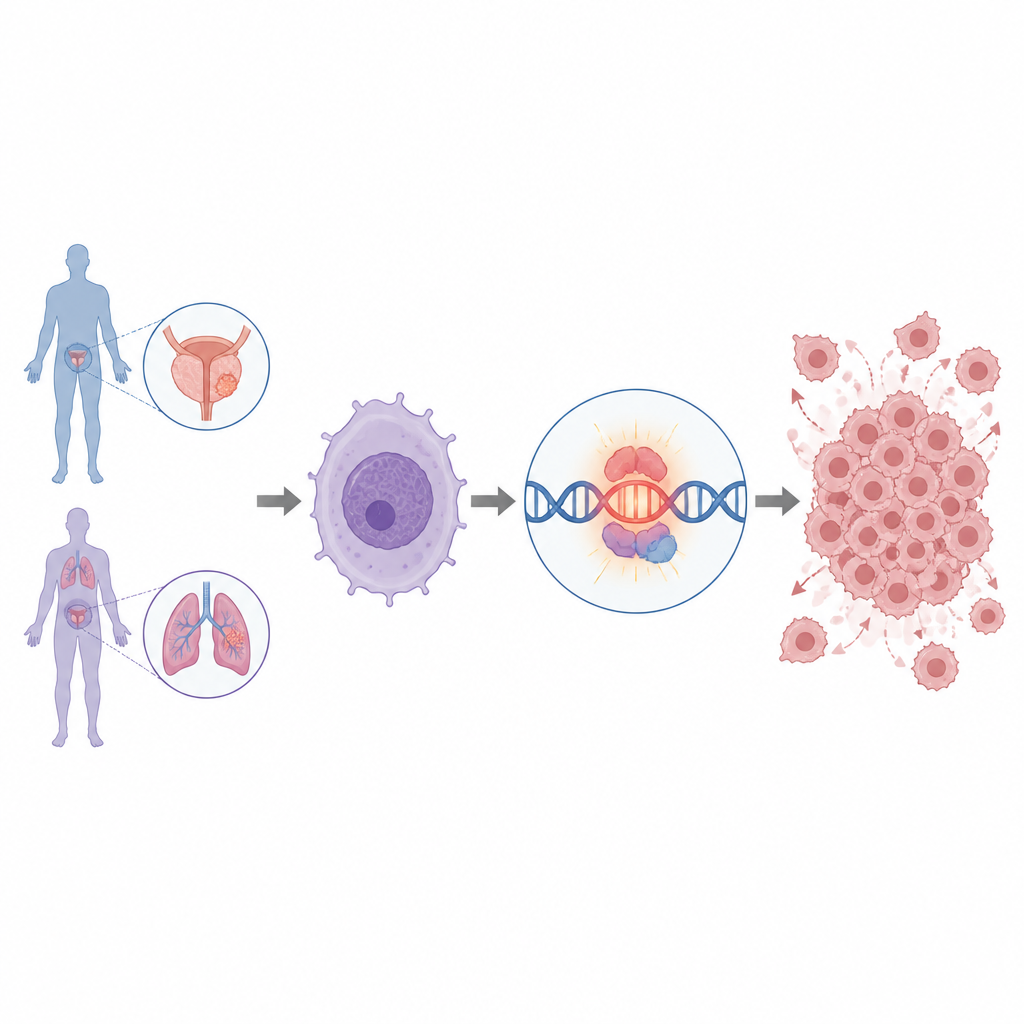
A Tumor-Only Signal in Prostate and Lung Cancer
The researchers began by sifting through RNA data from men with advanced, treatment-resistant prostate cancer. They focused on long non-coding RNAs, a class of RNA molecules that do not make proteins but can still strongly influence how genes behave. By combining patient data with maps of DNA control regions called super-enhancers, they identified more than a thousand long non-coding RNAs. One of these, named IGF1R-AS1, stood out because it was tightly linked to a powerful super-enhancer and was turned on almost only in tumors, especially in advanced prostate cancer and in some lung cancers, while being nearly silent in normal tissues.
From RNA Discovery to Cancer Behavior
Finding a tumor-only RNA is intriguing, but does it actually matter for cancer growth? To test this, the team used molecular tools to reduce IGF1R-AS1 levels in prostate and lung cancer cells grown in the lab. When this RNA was knocked down, cells grew more slowly, moved less, and were less able to invade through a barrier, all hallmarks of reduced cancer aggressiveness. In mice implanted with human prostate cancer cells, tumors lacking IGF1R-AS1 grew more slowly and animals survived longer. These experiments showed that IGF1R-AS1 is not just a bystander but plays an active, cancer-promoting role.
A New Route to Turn On the MYC Oncogene
Digging deeper, the scientists looked at which genes changed when IGF1R-AS1 was removed. A clear pattern emerged: many genes controlled by MYC were dialed down, and MYC itself dropped at both the RNA and protein level. Yet the nearby IGF1R gene, sitting next to IGF1R-AS1 in the genome, hardly changed. This meant IGF1R-AS1 was not simply fine-tuning its neighbor, as many similar RNAs do. Instead, it was acting at a distance to boost MYC, a master regulator of cell division. Across patient tumors, high activity of the IGF1R-AS1-linked gene network and high MYC activity tracked with poorer survival, especially in advanced prostate cancer. 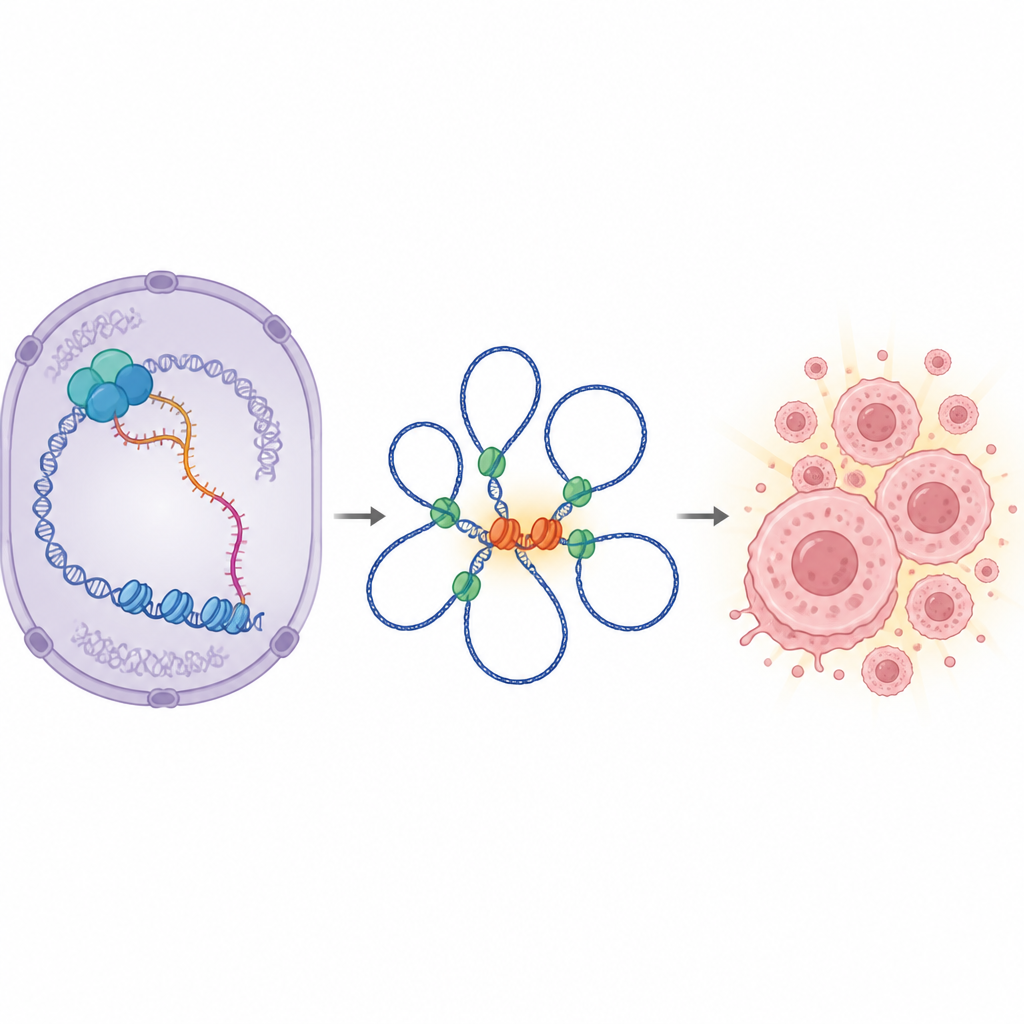
Shaping the 3D Genome to Favor Cancer Growth
How can an RNA molecule far from MYC crank up its activity? The answer lies in the three-dimensional folding of DNA inside the nucleus. The team showed that IGF1R-AS1 sits mainly in the nucleus and physically binds to chromatin remodeling proteins (such as SMARCA4 and SMARCA1) and to the architectural protein CTCF, which helps form DNA loops. These loops bring distant enhancer regions into contact with the MYC gene. When IGF1R-AS1 was reduced, many enhancer sites became less accessible, and long-range DNA loops that connect key enhancer hubs to the MYC promoter weakened or disappeared. In particular, a well-known enhancer region near another RNA called CCAT1 lost contact with MYC in prostate cancer cells that depend on IGF1R-AS1. The data support a model in which IGF1R-AS1 acts like a scaffold, helping chromatin remodelers and CTCF cooperate to maintain loops that keep MYC switched on.
Why This Hidden RNA Matters for Patients
For lay readers, the takeaway is that this study reveals a tumor-specific RNA that helps reshape the DNA architecture inside cancer cells to keep a powerful growth gene, MYC, in an “on” position. Because IGF1R-AS1 is largely absent from normal tissues but important for tumor growth and spread, it may serve as a marker to identify high-risk cancers and a potential handle for therapies that selectively target cancer cells. More broadly, the work shows how non-coding RNAs can organize the 3D structure of the genome to influence which genes are active, adding an important new layer to our understanding of how cancers arise and progress.
Citation: Yang, Y., Wang, TY., Fry, J. et al. Tumor-specific lncRNA IGF1R-AS1 trans-regulates chromatin interactions associated with oncogenic MYC signaling. Nat Commun 17, 4171 (2026). https://doi.org/10.1038/s41467-026-70814-4
Keywords: prostate cancer, long noncoding RNA, MYC signaling, chromatin looping, tumor biomarkers