Clear Sky Science · en
Structure of a pH-sensitive pentameric ligand-gated ion channel from the Sarcoptes scabies mite
Why tiny mites and their hidden switches matter
Scabies is a common but often overlooked skin disease that causes intense itching and can lead to serious complications if untreated. It is caused by the microscopic Sarcoptes scabiei mite, and doctors rely heavily on the drug ivermectin to control it. However, signs of growing drug resistance are emerging. This study reveals the detailed 3D structure and behavior of a key protein in the mite’s nervous system—a pH-sensitive chloride channel called SsCl—and shows exactly how ivermectin interacts with it. By understanding this molecular switch, scientists hope to design next-generation treatments that remain effective even as resistance rises.
A special ion channel in a troublesome parasite
SsCl belongs to a family of proteins that form pores, or channels, across cell membranes to let charged particles pass in and out. These movements of ions underlie electrical signaling in nerves and muscles. Unlike classic receptors that respond to specific chemical messengers, SsCl responds to changes in acidity or alkalinity (pH) around the cell. When the environment becomes more alkaline, SsCl normally opens to let chloride ions flow, helping the mite’s cells maintain balance and communicate. Because pH-sensitive chloride channels are found mainly in invertebrates, they are attractive targets for drugs that kill parasites without harming humans. Yet, until now, scientists did not know how SsCl senses pH or how ivermectin alters its activity.
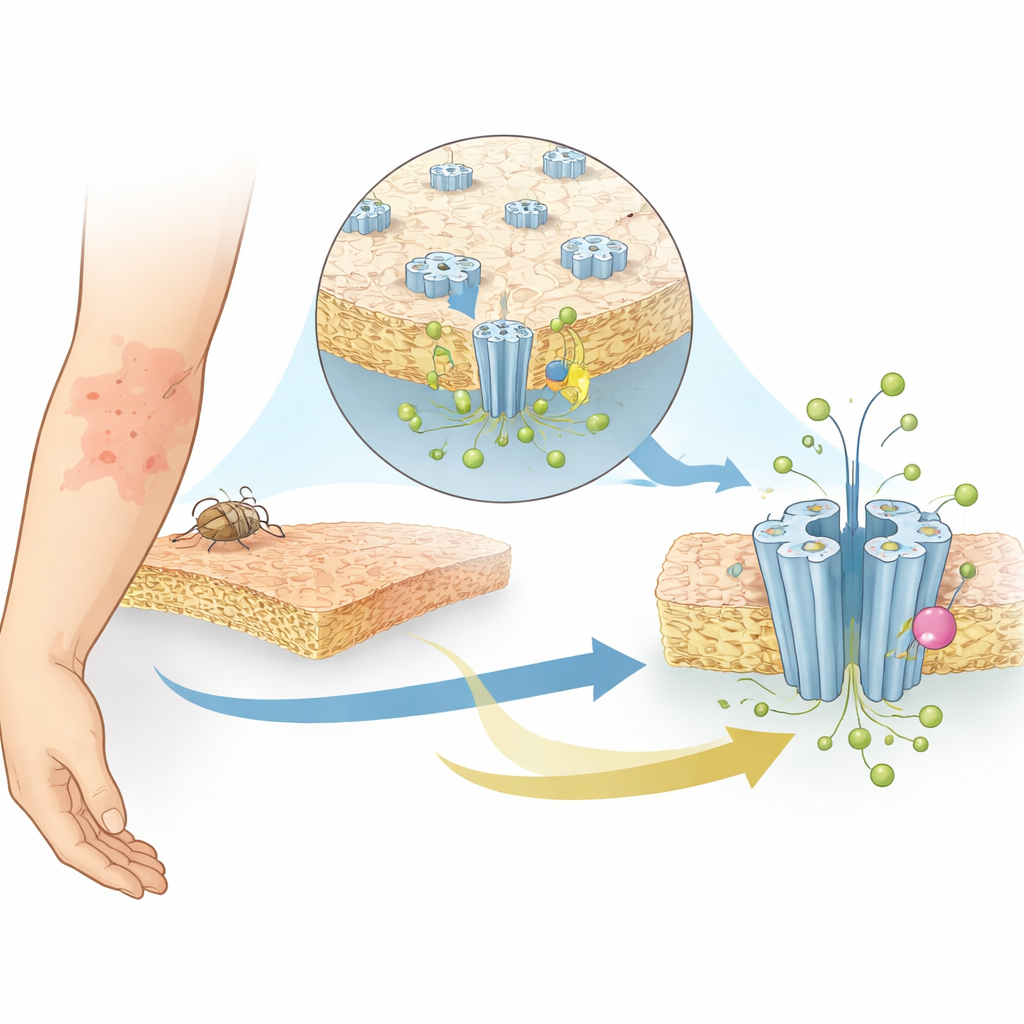
Capturing the channel in action with cryo-EM
The researchers used single-particle cryo-electron microscopy, a technique that flash-freezes proteins and images them at near-atomic resolution, to capture SsCl in four different shapes, or states. They studied the channel at a mildly acidic pH (6.5), where it is closed, and at a more alkaline pH (9), where it should be active but instead adopts a “desensitized” form that no longer conducts ions after prolonged stimulation. They also determined structures of SsCl at both pH levels in the presence of ivermectin. In all cases, the protein assembles as a five-part ring forming a central pore. Surprisingly, the pore in the closed and desensitized states takes on an hourglass-like shape, with the narrowest point in the middle of the membrane—more reminiscent of channels that conduct positively charged ions than of other known chloride channels.
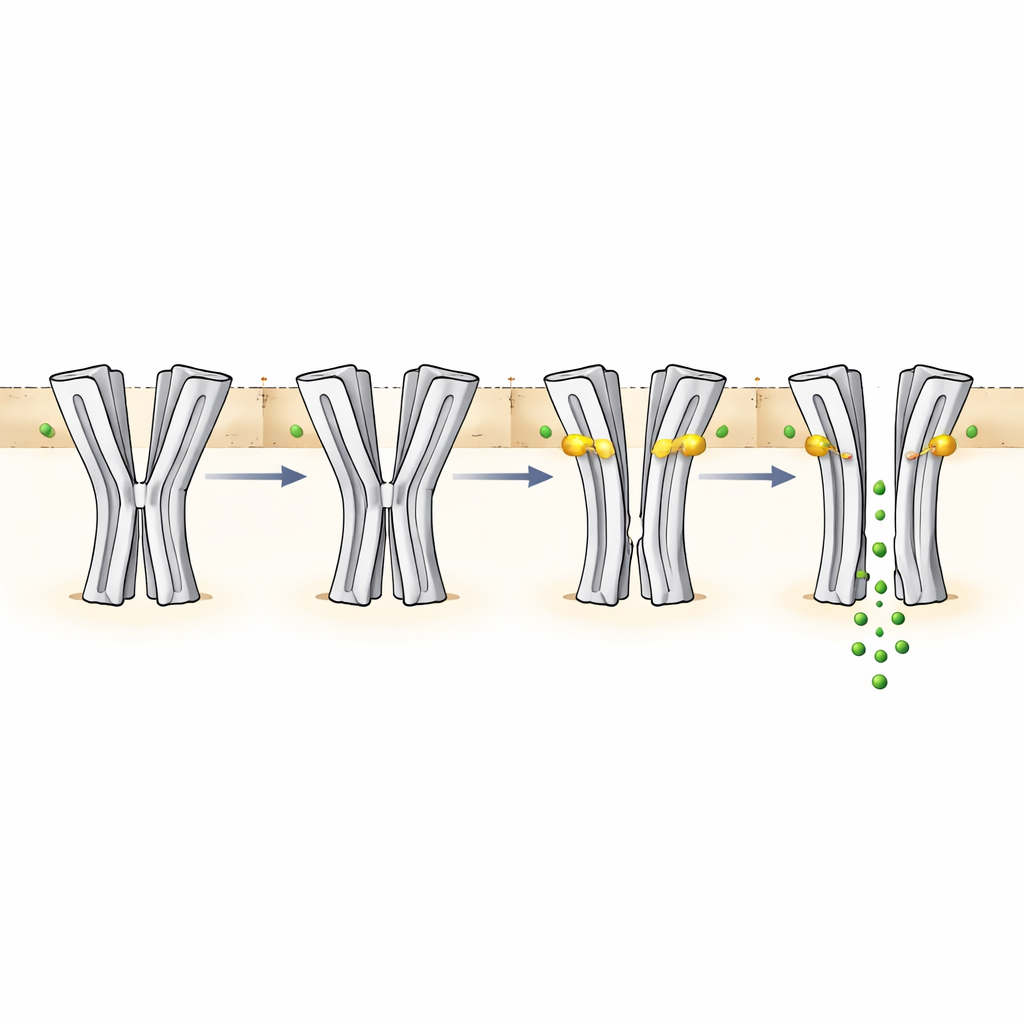
How pH and ivermectin reshape the pore
By measuring the size of the pore in each structure, the team could relate shape to function. At pH 6.5 without drug, the central constriction is too narrow for chloride ions to pass, matching the non-conducting state. At pH 9 without drug, the pore is still too tight at the middle, even though the environment would normally favor channel opening. This configuration is interpreted as a desensitized state that results from long exposure to alkaline conditions. When ivermectin wedges itself between segments of neighboring subunits in the membrane-spanning region, it pries the helices apart and widens the pore. At pH 6.5, this expansion creates a partially open path that still restricts ion flow near the selectivity filter at the bottom of the pore, consistent with the drug’s slow and modest activation of the channel. At pH 9 with ivermectin, the pore becomes wide enough along most of its length to support robust chloride conduction, in line with strong electrical currents recorded in functional experiments.
The hidden pH sensor and a broader control network
To pinpoint how SsCl detects pH, the authors combined computational predictions with structural comparisons and mutagenesis. They identified a cluster of amino acids—particularly several histidines and glutamic acids—in the outer, water-exposed part of the protein, near a region that in related receptors usually binds chemical messengers. One key pair, a histidine (H206) and a glutamic acid (E146), can form or break an ionic bond depending on pH. At lower pH, this bond helps lock certain structural elements in place; at higher pH, the bond weakens, allowing a subtle twist in this area to propagate down to the membrane-spanning helices that form the pore. Mutating H206 or E146 altered the pH range over which the channel activates, while double mutations could disrupt pH sensitivity yet still allow ivermectin to trigger currents. Additional residues near a flexible loop at the channel’s entrance also influenced how the protein responds to pH changes, supporting the idea of a distributed pH-sensing network rather than a single on–off switch.
From molecular insight to better scabies treatments
Overall, the work shows how a scabies mite ion channel senses its chemical surroundings and how ivermectin stabilizes particular shapes of the channel to promote or enhance opening. SsCl proves to be an unusual chloride channel whose desensitized shape resembles that of channels for positively charged ions and whose charge distribution along the pore is finely tuned to guide chloride ions. By resolving the channel’s structures in multiple functional states and mapping the residues that tune pH and drug sensitivity, the study provides a blueprint for designing new antiparasitic compounds. These next-generation drugs could latch onto similar sites or exploit the same pH-sensing machinery, offering fresh options to combat scabies in a world where current treatments are increasingly at risk of losing their power.
Citation: Kleiz-Ferreira, J., Brams, M., Harrison, P.J. et al. Structure of a pH-sensitive pentameric ligand-gated ion channel from the Sarcoptes scabies mite. Nat Commun 17, 3392 (2026). https://doi.org/10.1038/s41467-026-70575-0
Keywords: scabies, ion channels, ivermectin, cryo-electron microscopy, parasite control