Clear Sky Science · en
Intellectual disability-causing mutations in KIF11 impair microtubule dynamics and dendritic arborization
How tiny cellular motors shape thinking
Our brains depend on billions of nerve cells that must wire up with exquisite precision. This study explores how a single molecular motor, called KIF11, helps sculpt the branching “tree” of nerve cell processes that receive signals, and how disease-causing changes in this motor may lead to intellectual disability. By watching living neurons under the microscope and probing KIF11’s mechanics in test tubes and in mice, the authors reveal how this protein fine‑tunes the internal scaffolding of neurons and, in turn, their ability to communicate.
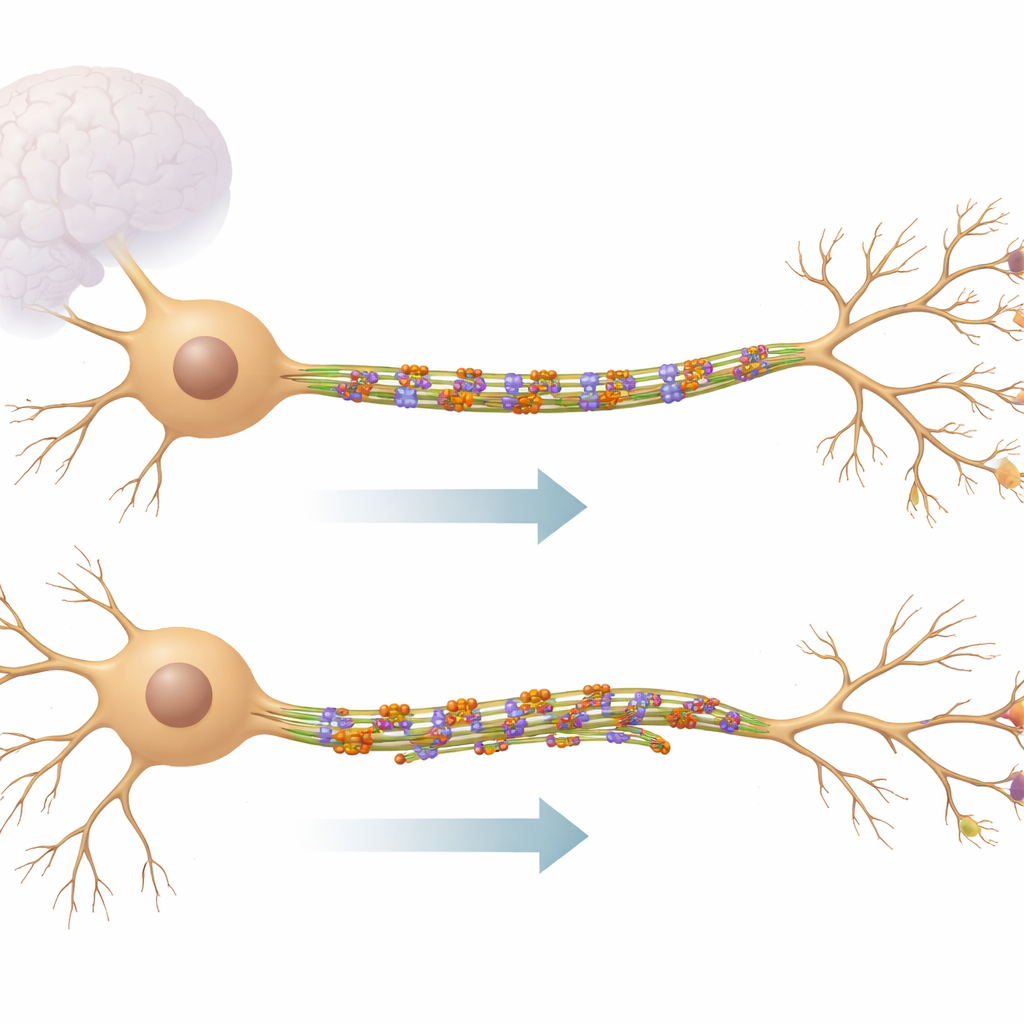
The neuron’s inner railway system
Neurons send and receive information through long, cable‑like extensions. Inside these cables run microtubules, stiff protein rods that act like railway tracks for moving supplies and help set the shape of the cell. In the main input branches, called dendrites, microtubules run in both directions, creating a mixed, dynamic network. KIF11 belongs to a family of tiny motors that usually work during cell division, but this work shows that it also remains active in fully developed brain cells. Instead of hauling cargo, KIF11 links neighboring microtubules together, acting more like a brake and stabilizer than a delivery truck.
Keeping growth in balance
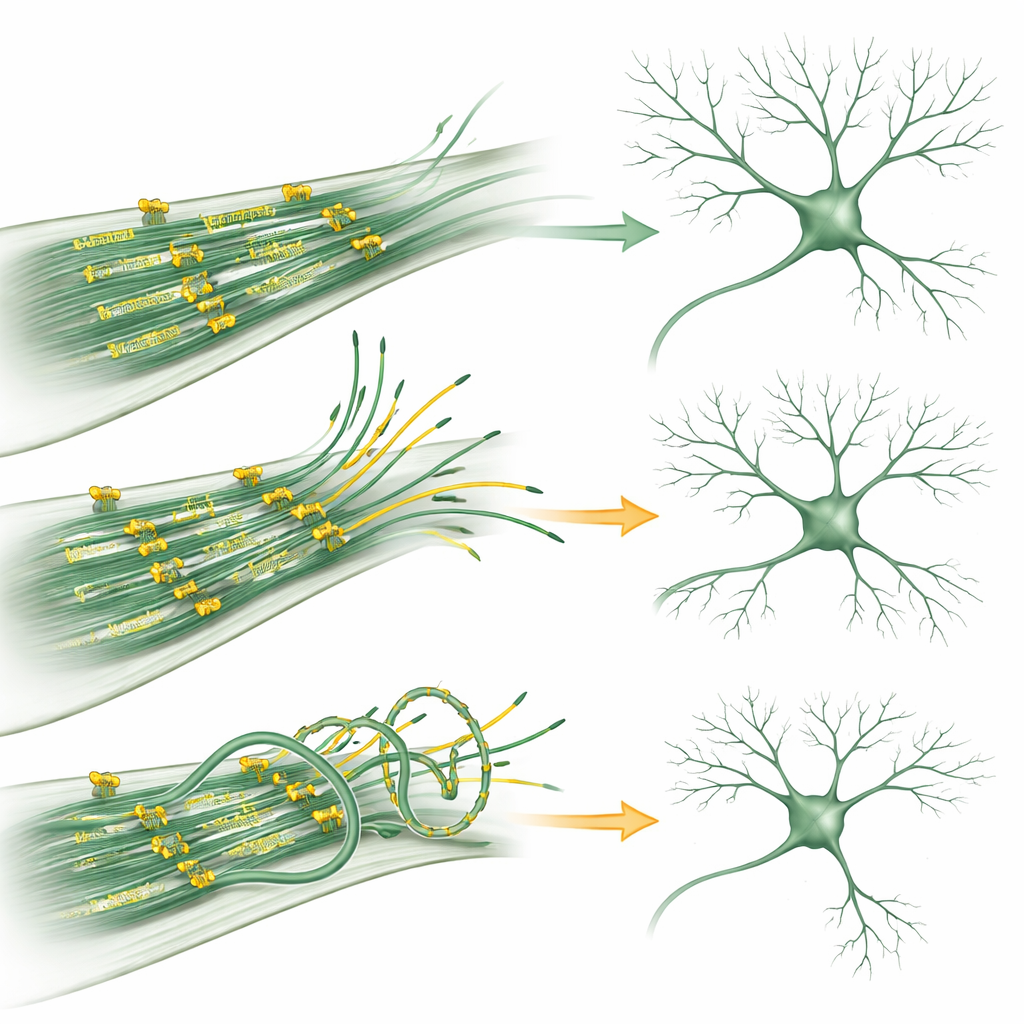
Using live imaging of mouse hippocampal neurons, the researchers reduced KIF11 levels or blocked its activity with drugs and tracked the tips of growing microtubules. They found that when KIF11 was inhibited, one particular class of microtubules—those with their “minus end” pointing outward—became more active, especially in secondary and tertiary dendritic branches. This boosted the appearance and growth of these tracks and encouraged new branch formation, making the dendritic tree more elaborate. Conversely, when KIF11 was overproduced, microtubule dynamics slowed, dendrites became less complex, and the internal tracks sometimes twisted and looped, suggesting that excessive force from KIF11 can bend and strain the scaffold.
When a motor goes wrong
Certain families affected by a rare condition called MCLID—marked by small head size, eye problems, swelling of limbs, and intellectual disability—carry mutations in the KIF11 gene. The team recreated two such mutations in the laboratory. In neurons, both altered forms of KIF11 dampened microtubule dynamics and reduced dendritic branching, similarly to KIF11 overproduction. Detailed biochemical tests revealed how: one mutant disrupted KIF11’s ability to assemble into its normal four‑part structure, weakening its capacity to link microtubules; the other still formed the proper complex but slid along microtubules much more slowly and had lower affinity for the fuel molecule ATP. Despite these differences, both mutants continued to exert abnormal forces on microtubules, upsetting the fine balance of stability and flexibility that neurons require.
From cell shape to brain signals
The authors then asked whether these structural changes matter for communication between neurons. By recording tiny spontaneous electrical events in cultured cells, they showed that increasing normal KIF11 or either mutant significantly reduced how often synaptic vesicles released their chemical messages, even though the size of each event stayed the same. In living mice, introducing the human‑like KIF11 variants into developing hippocampal neurons led to shorter and less branched dendritic trees, with the severity depending on the specific mutation and the stage of development. Together, these findings link faulty KIF11 activity to both the physical architecture of neurons and the flow of information across brain circuits.
Light‑controlled motors and a new view of disease
To test whether changing KIF11 activity locally is enough to remodel neurons, the researchers engineered a light‑sensitive version of the motor. Brief flashes of violet light switched KIF11 off in chosen dendrite segments, rapidly increasing microtubule growth and triggering the sprouting of new branches within minutes. Opposite light pulses switched KIF11 on and reduced growth while inducing kinks and loops in the microtubule bundles. These experiments support a simple picture: KIF11 acts as a rheostat, or dimmer switch, for the neuron’s internal rails—too little activity lets branches overgrow, while too much or the wrong kind of force stiffens or distorts them. In MCLID, mutant KIF11 proteins appear to apply unbalanced forces on microtubules, leading to unstable dendritic trees and weakened synaptic communication, which likely contributes to problems with learning and cognition.
Citation: Wingfield, J.L., Niese, L., Avchalumov, Y. et al. Intellectual disability-causing mutations in KIF11 impair microtubule dynamics and dendritic arborization. Nat Commun 17, 4125 (2026). https://doi.org/10.1038/s41467-026-70522-z
Keywords: neuronal microtubules, dendritic branching, kinesin motor proteins, intellectual disability, synaptic transmission