Clear Sky Science · en
A single mutation in nonstructural protein 1 is critical for the adaptive evolution of influenza B virus
Why this flu story matters
Influenza B is often thought of as the “milder cousin” of the better-known influenza A viruses, yet it quietly causes a large share of seasonal flu cases, especially in children. This study asks a deceptively simple question with big implications: how has influenza B changed over time to become better at living in people? The authors trace that change down to a single amino acid in one viral protein and show how that tiny tweak helps the virus slip past one of our cells’ earliest alarm systems.
A closer look at a quiet troublemaker
Influenza B viruses have circulated in humans for at least 80 years and now fall into two major families, called Yamagata and Victoria. Epidemiological studies show that in some seasons influenza B can cause up to half of all flu infections and a substantial fraction of hospital costs, particularly among school-age children. Yet compared with influenza A, we know far less about how influenza B adapts to the human body. Earlier work hinted that one viral protein, called NS1, evolves rapidly and might help the virus dodge our innate immune defenses, but the exact changes that matter were unknown.
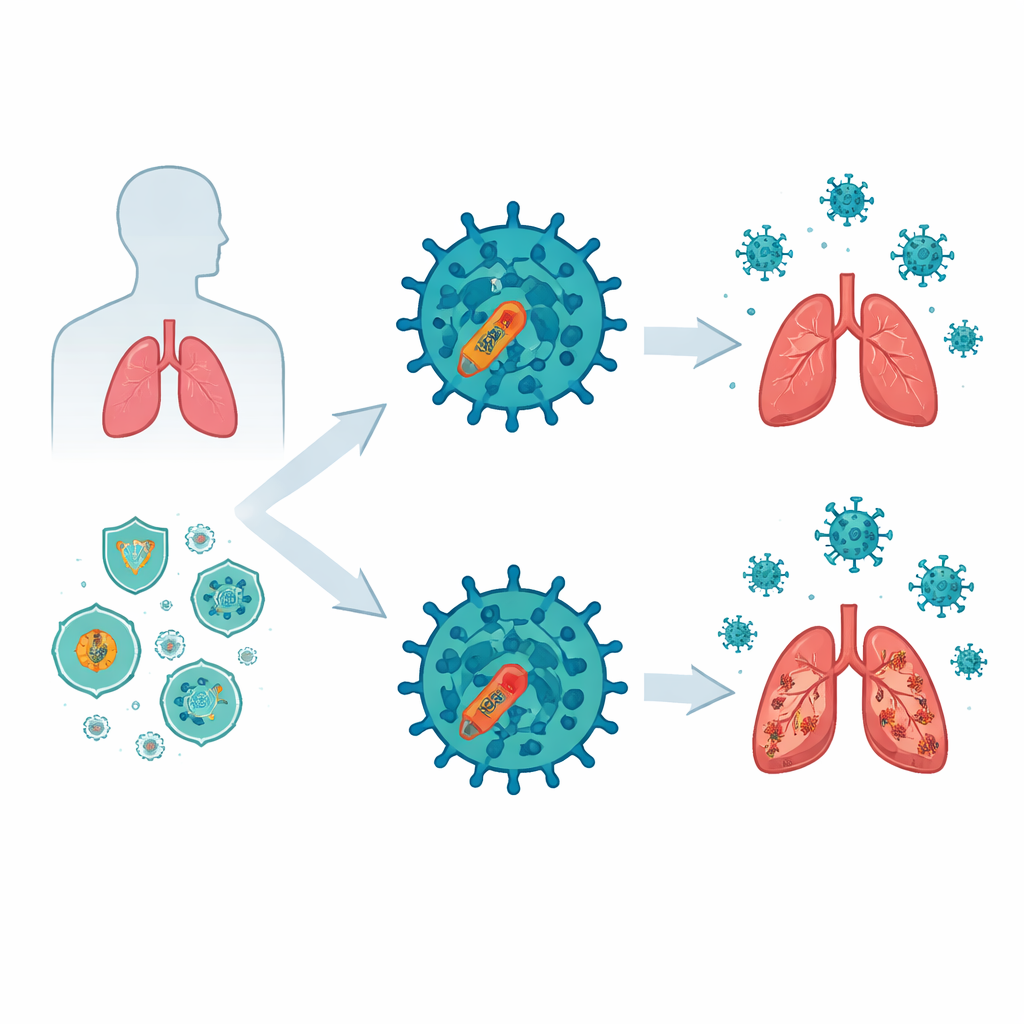
Newer viruses outplay our early defenses
The researchers compared older influenza B strains, including a classic 1940 virus and an isolate from 1988, with a recent strain from 2018. In human lung cells and in mice, the 2018 virus replicated to higher levels, caused more lung damage, and triggered less production of interferons—molecules that act as the body’s rapid-response antiviral signals—and interferon-stimulated genes. Infected mice lost more weight, had higher death rates, and showed more severe lung injury when exposed to the modern strain. These observations pointed to a clear pattern: recent influenza B viruses have become better at suppressing the host’s early alarm system, giving themselves more time and space to multiply.
The hidden clean-up trick inside cells
To understand how this happens, the team zoomed in on NS1, a multifunctional viral protein already known to interfere with immune signaling. They discovered that the NS1 from the 2018 strain has a special ability: it triggers a form of cellular “self-cleaning” directed at mitochondria, the energy-producing structures that also act as hubs for antiviral signaling. This pathway, called mitophagy, packages damaged mitochondria into double-membraned sacs that fuse with digestive compartments. When NS1 from the recent strain is present, it causes key mitochondrial proteins, including MAVS—a central relay in the interferon alarm pathway—to be swept into this disposal route and degraded. As MAVS levels drop, the cell’s ability to send out antiviral interferon signals collapses, while the virus replicates more efficiently.
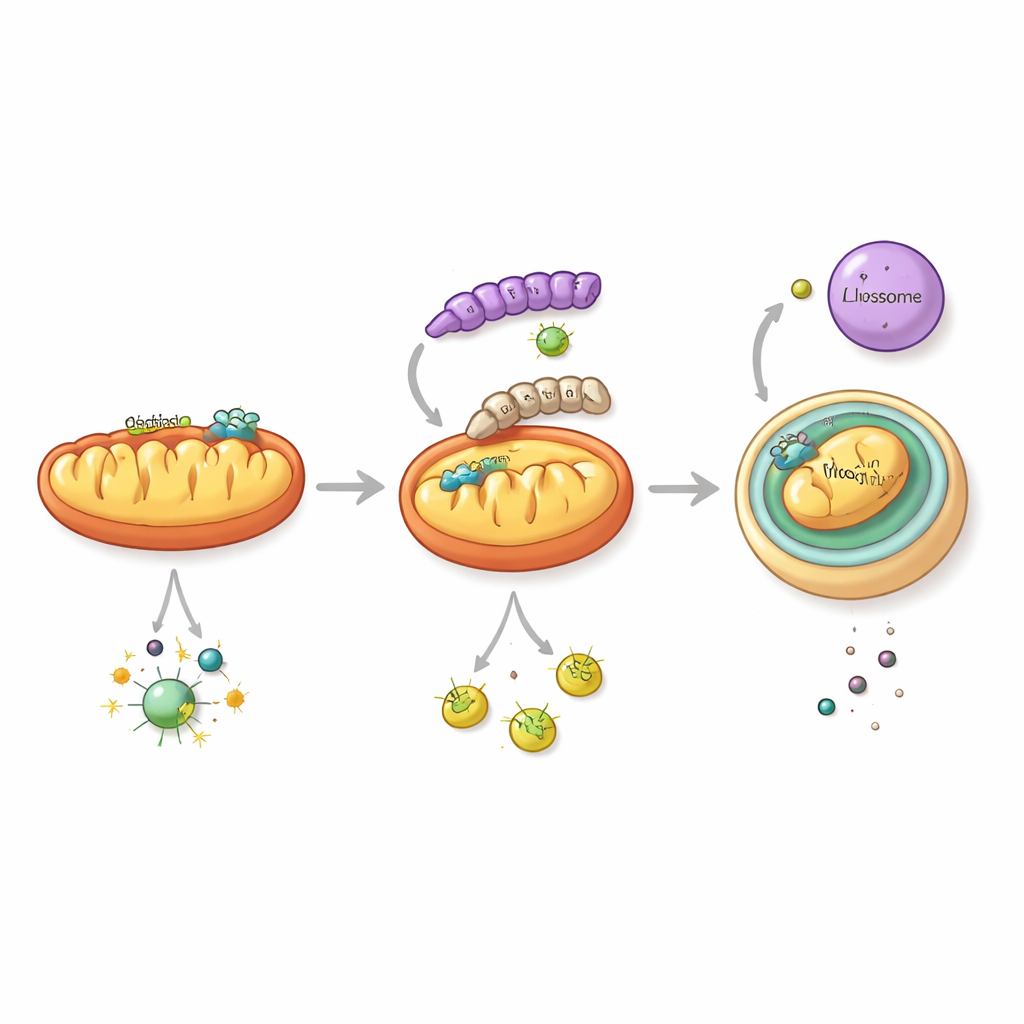
A single molecular swap with big impact
What makes the modern NS1 so good at hijacking mitophagy? By analyzing thousands of virus sequences, the scientists identified a recurring change at one specific spot in NS1: amino acid 247. In older strains this position is occupied by phenylalanine, but over the past two decades it has largely been replaced by leucine. Structural modeling and biochemical tests showed that this leucine allows NS1 to bind more tightly to LC3B, a key marker on the membranes that form autophagosomes, and to TUFM, a mitochondrial protein that helps organize mitophagy. Together, these interactions bring NS1 onto mitochondria and into the forming autophagosomes, promoting selective removal of MAVS-bearing mitochondria. When the researchers engineered a recent virus to “go back” to the old amino acid (leucine back to phenylalanine), the altered virus induced less mitophagy, spared more MAVS, triggered stronger interferon responses, and caused milder disease in mice.
What this means for future flu seasons
This work reveals that the long-term success of influenza B in humans is not only about changes in its surface proteins that help it escape antibodies, but also about subtle tuning of its ability to silence our earliest antiviral defenses. A single amino acid substitution in NS1 has given recent viruses a powerful way to dismantle the cell’s alarm machinery by redirecting mitochondria into the cellular trash route. For the general public, the message is that viruses can become more adept not just at infecting us, but at hiding from our immune systems in increasingly sophisticated ways. For researchers and public health officials, monitoring such adaptive changes in internal viral proteins like NS1 may be as important as tracking mutations in surface proteins when forecasting future flu threats and designing better vaccines and treatments.
Citation: Jiao, P., Jia, X., Bai, X. et al. A single mutation in nonstructural protein 1 is critical for the adaptive evolution of influenza B virus. Nat Commun 17, 3353 (2026). https://doi.org/10.1038/s41467-026-70211-x
Keywords: influenza B virus, innate immunity, viral evolution, autophagy and mitophagy, NS1 protein mutation