Clear Sky Science · en
Branched-chain α-keto acids impair glucose-stimulated insulin secretion in pancreatic β-cells under diabetes by reactivating the LDHA-lactate axis
Why this discovery matters for everyday health
Type 2 diabetes is often described as a problem of the body becoming insensitive to insulin, but this study shows that the cells that make insulin themselves can also be poisoned by certain byproducts of our diet. The researchers uncover how specific breakdown products of common amino acids can quietly sabotage insulin release, helping to explain why some people with diabetes struggle to keep blood sugar in check and pointing toward new ways to protect the pancreas.
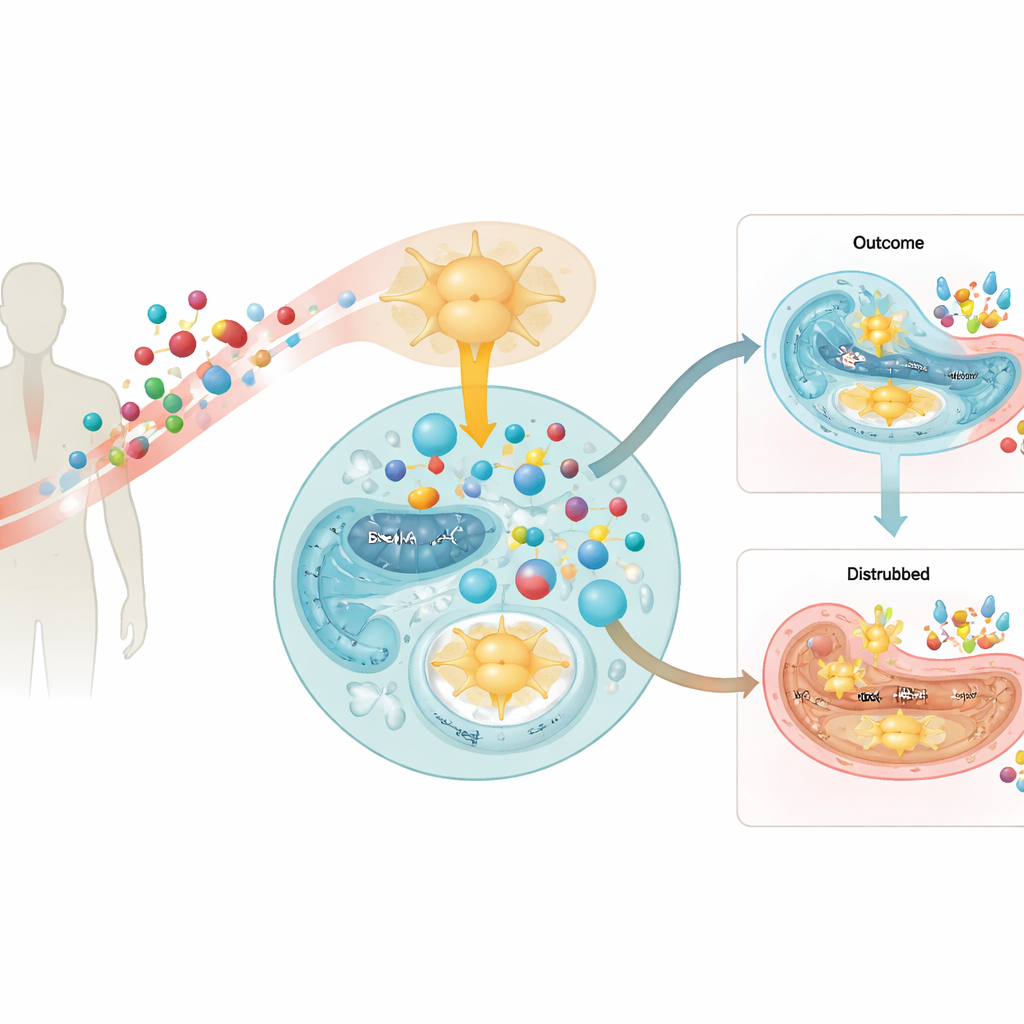
Diet building blocks with a hidden dark side
The work focuses on branched-chain amino acids, nutrients found in protein-rich foods and popular sports supplements. When the body breaks these amino acids down, it produces compounds called branched-chain alpha-keto acids. In healthy conditions, these keto acids are further burned in mitochondria, the cell’s power plants, to help fuel energy production. In people and mice with diabetes, however, the authors found that the machinery that disposes of these keto acids is slowed in the insulin-producing cells of the pancreas. As a result, the keto acids accumulate inside the islets where beta cells live.
Clues from human patients and animal models
By analyzing blood samples from people with diabetes and from community volunteers, the team showed that higher circulating levels of these keto acids were linked to poorer beta-cell function, even after accounting for insulin resistance. In diabetic mice and in human donor islets, key enzymes that normally clear the keto acids were present at lower levels or switched off by chemical tags, and the beta cells themselves contained more of the keto acids. When healthy mice were given drinking water enriched with these molecules, they gradually became less able to handle a sugar load, not because their muscles or liver ignored insulin, but because their beta cells stopped releasing enough of it when glucose rose.
How the energy pipeline in beta cells gets rewired
Under normal circumstances, glucose entering a beta cell is broken down to pyruvate, which is then shuttled into mitochondria and fed into the tricarboxylic acid cycle to make ATP, the energy signal that triggers rapid insulin release. Using isotope-labeled glucose and high-precision metabolic measurements, the researchers found that excess keto acids divert pyruvate away from this energy cycle. Instead of entering mitochondria, more of it is converted into lactate, a dead-end product in beta cells. This shift lowered ATP production and blunted the crucial first burst of insulin that normally follows a rise in blood sugar, while leaving other aspects of cell health, such as survival and general glucose uptake, largely intact.
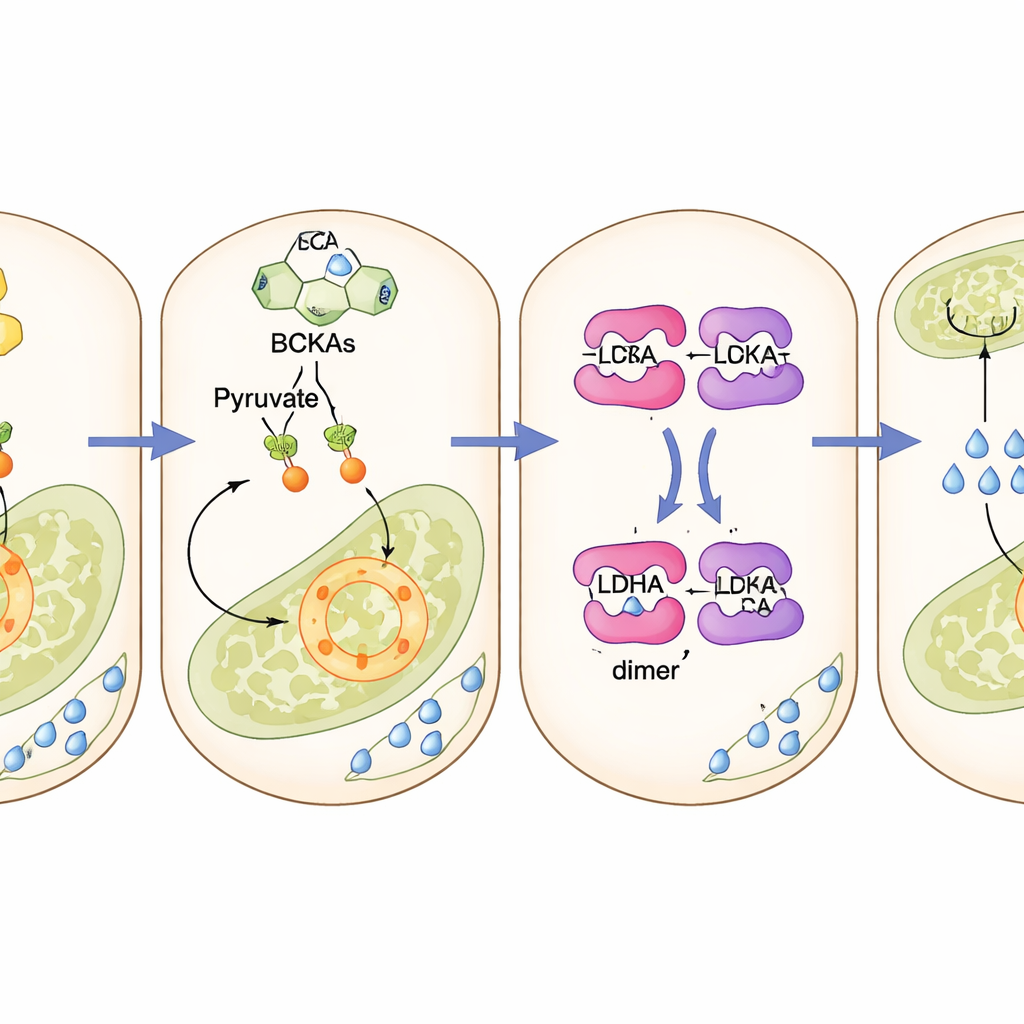
A dormant enzyme turned back on
At the heart of this diversion is an enzyme called lactate dehydrogenase A, which is normally kept quiet in beta cells so that glucose is channeled toward energy production rather than lactate. The study shows that the accumulated keto acids physically bind to this enzyme and act like tiny bridges that snap its subunits together into a more active form. Once activated, the enzyme pulls pyruvate toward lactate, starving the mitochondria of fuel. Blocking the enzyme genetically in beta cells, or using a small-molecule inhibitor, protected cells and mice from the harmful effects of keto acids and restored normal insulin release. Drugs that push the keto acids back into their usual breakdown route had a similar rescuing effect.
What this means for treating diabetes
To a non-specialist, the key message is that in diabetes the problem is not only how the body responds to insulin, but also how the insulin-producing cells handle certain dietary byproducts. When the breakdown of branched-chain amino acids is disturbed, their keto-acid offspring build up, flip on a normally silent enzyme, and reroute sugar away from energy production, weakening the pancreas’s ability to respond to glucose. This suggests that monitoring these keto acids in the blood could help flag failing beta cells, and that carefully adjusting branched-chain amino acid intake or designing medicines to restore their proper disposal may open new avenues to preserve or recover insulin secretion in type 2 diabetes.
Citation: Lin, H., Ho, M.Y.M., Wang, B. et al. Branched-chain α-keto acids impair glucose-stimulated insulin secretion in pancreatic β-cells under diabetes by reactivating the LDHA-lactate axis. Nat Commun 17, 3346 (2026). https://doi.org/10.1038/s41467-026-70004-2
Keywords: type 2 diabetes, insulin secretion, branched-chain amino acids, pancreatic beta cells, lactate metabolism