Clear Sky Science · en
Mass spectrometry reveals the evolutionary conservation of phycobiliprotein complexes
Ancient microbes with a modern-day impact
Cyanobacteria—tiny, photosynthetic microbes—helped transform Earth’s early atmosphere by releasing oxygen and still underpin today’s global carbon and nitrogen cycles. They thrive from scorching hot springs to icy lakes, yet many share the same kind of light-harvesting machinery. This study asks a simple but profound question: how have these light-collecting parts stayed so good at their job for more than three billion years, even as cyanobacteria spread into wildly different environments?
How these microbes catch light
Cyanobacteria use large molecular “antenna” structures, called phycobilisomes, to capture sunlight and pass the energy to the core photosynthetic engines inside the cell. Phycobilisomes are built from colored proteins known as phycobiliproteins, chiefly phycocyanin and allophycocyanin, which assemble like stacked donuts. Each donut is made from repeating pairs of protein chains that bind pigment molecules. The overall size and shape of the antenna can vary a lot between species, but the individual building blocks look strikingly similar, hinting at a deeply conserved design.
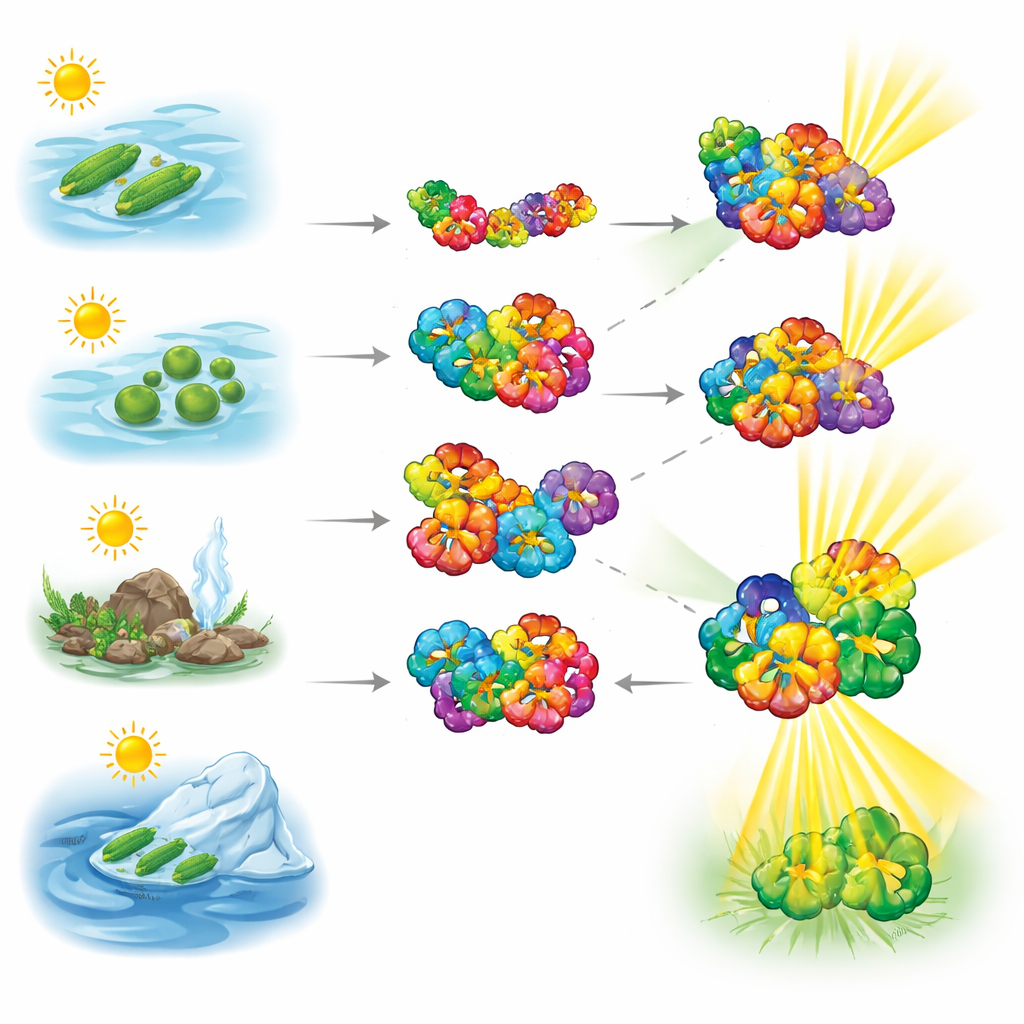
Probing flexible building blocks with precision weighing
To see how these light-harvesting proteins behave, the researchers used native mass spectrometry, a technique that gently weighs intact protein complexes without breaking them apart. They examined phycobiliproteins from cyanobacteria collected from very different habitats—hypersaline waters, freshwater, hot environments, and cold regions. The measurements showed that phycocyanin readily switches between smaller pairs and larger donut-like hexamers, revealing a highly dynamic nature. Allophycocyanin, by contrast, was much more likely to stay in its hexameric form, suggesting a sturdier core that anchors the antenna system.
Mixing parts from different species
The team then performed a molecular “mix-and-match” experiment. They combined purified phycobiliproteins from pairs of species that live in different environments or belong to distant branches of the cyanobacterial family tree. Mass spectrometry revealed that the proteins rapidly assembled into hybrid complexes: hexamers built from subunits originating in two different species. This happened even when the species were only distantly related, and even when the proteins were purified away from most of their usual helper components. However, one rule held firm: phycocyanin parts only mixed with other phycocyanin, and allophycocyanin with allophycocyanin—no mixed donuts containing both types were detected.
Atomic-level clues from structure prediction
To understand why some mixtures form easily and others never appear, the researchers turned to AlphaFold2, an advanced protein-structure prediction tool. They modeled both pure and hybrid hexamers and examined how closely the predicted protein surfaces fit together. Hybrid complexes made from the same phycobiliprotein type but from different species showed tight, confident interfaces, matching the experimental evidence that these assemblies are stable. In contrast, hypothetical hexamers containing a mix of phycocyanin and allophycocyanin showed poorer fits and fewer contacts, indicating that such combinations are structurally disfavored. Detailed comparison of amino-acid positions at the contact zones revealed a handful of conserved residues that act like shape-keys, ensuring that only matching types lock together well.
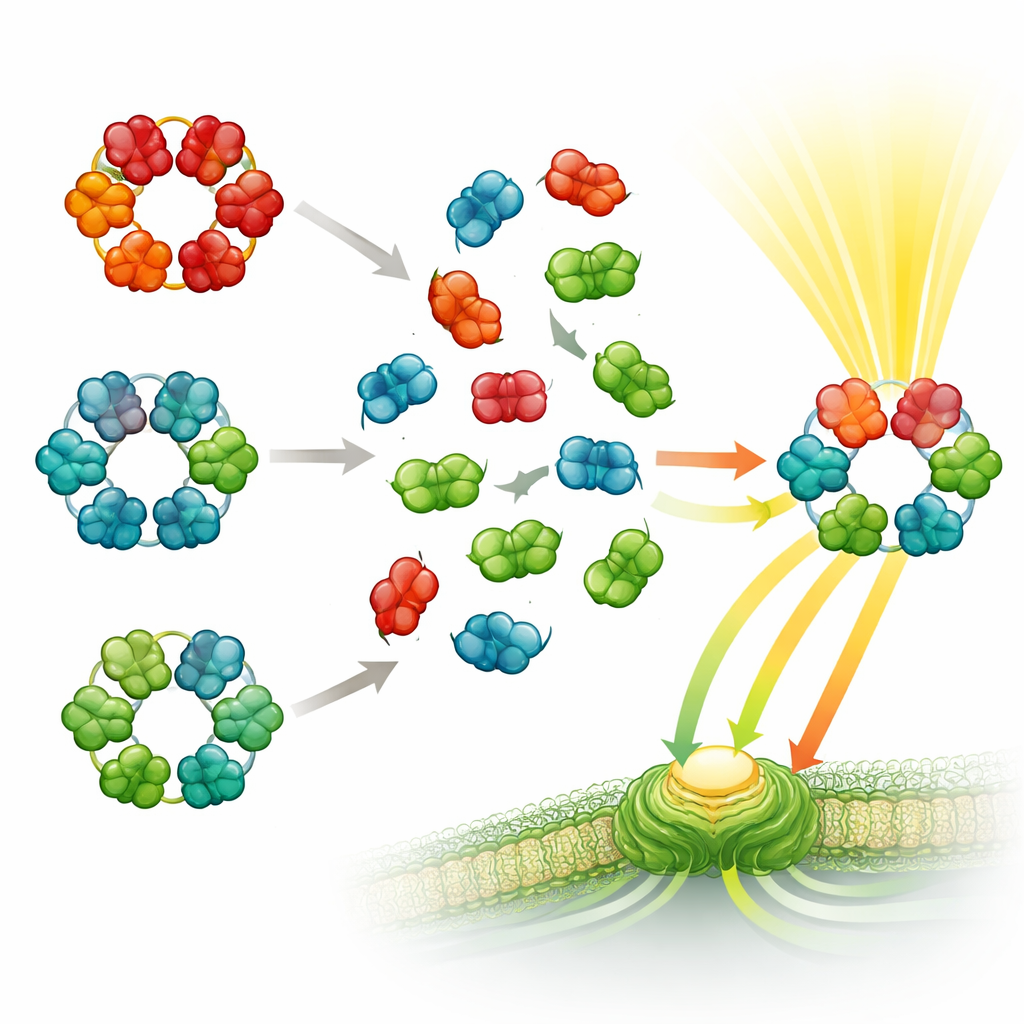
What this means for life and for technology
The findings suggest that the core design of phycobiliproteins has been strongly preserved over billions of years, allowing subunits from far-flung cyanobacteria to function almost interchangeably. At the same time, subtle changes at their contact surfaces prevent mismatched combinations of different phycobiliprotein types, keeping energy flow through the antenna highly efficient. This balance of flexibility and specificity likely helped cyanobacteria colonize many habitats while maintaining reliable photosynthesis. In practical terms, the work hints that it may be possible to engineer new light-harvesting systems—by swapping compatible subunits from different species—to tune which colors of light are captured, with potential applications in bioenergy, biotechnology, and sustainable materials.
Citation: Sound, J.K., Bianchini, G., Ashok, T.A. et al. Mass spectrometry reveals the evolutionary conservation of phycobiliprotein complexes. Nat Commun 17, 2834 (2026). https://doi.org/10.1038/s41467-026-69558-y
Keywords: cyanobacteria, photosynthesis, light harvesting, protein evolution, mass spectrometry