Clear Sky Science · en
Identification and structural characterization of anthrax toxin receptor 2 as the Clostridium perfringens NetF receptor
Why this toxin matters for people and animals
Severe gut infections in dogs, horses, and livestock can appear suddenly, causing bloody diarrhea and sometimes death. A common culprit is the bacterium Clostridium perfringens, which harms cells by punching holes in them with powerful protein toxins. This study reveals, in molecular detail, how one such toxin, called NetF, finds and attacks specific animal cells. The work not only explains puzzling disease outbreaks in pets and farm animals, but also uncovers general rules for how pore-forming toxins latch onto our cells, a step that could be targeted by future therapies.
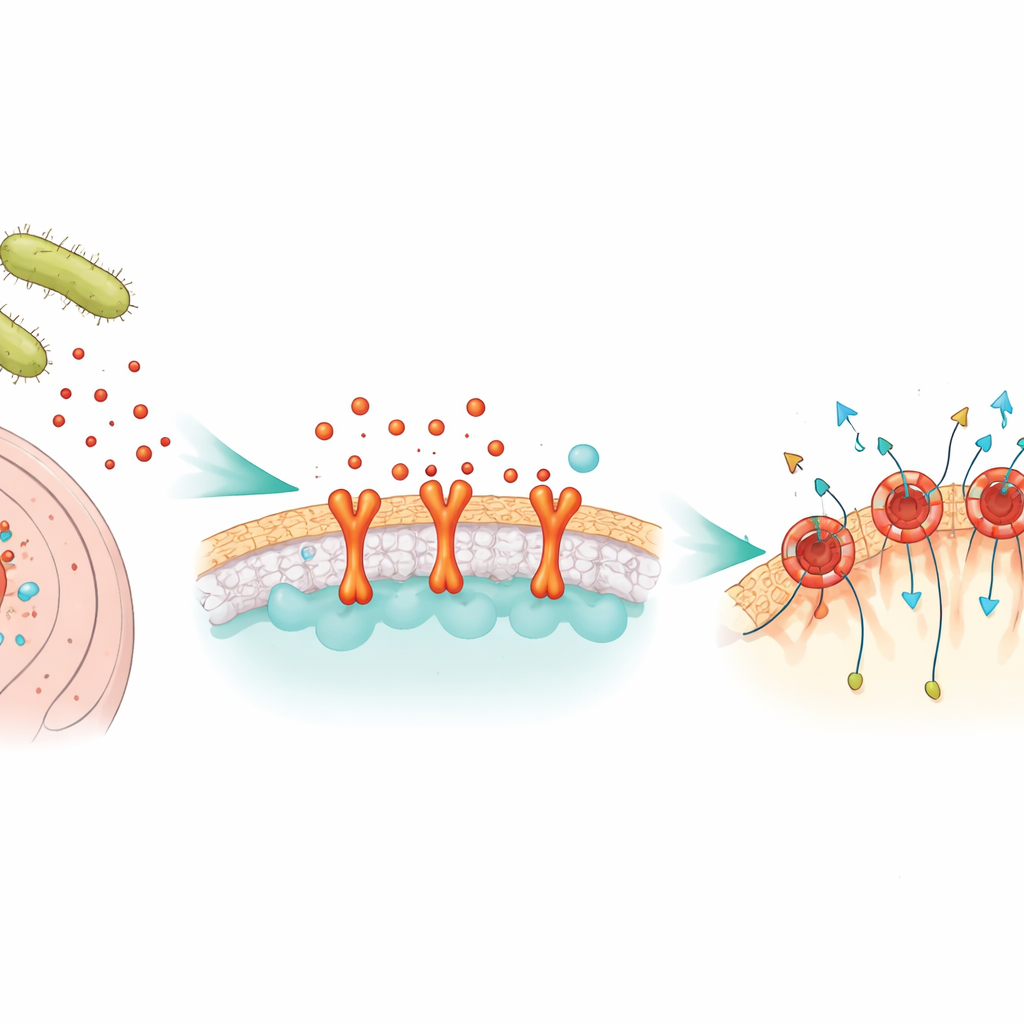
A bacterial weapon that drills holes
NetF belongs to a large family of “pore-forming” toxins that bacteria release as soluble single units. Once they touch a suitable cell, these units assemble into rings that insert into the cell membrane and form channels. Through these channels, vital molecules leak out and the cell collapses. Although similar toxins from Clostridium perfringens are known to cause deadly intestinal disease in pigs, poultry, and people, very little was known about how NetF chooses its victims or what its final pore looks like in atomic detail.
Finding the toxin’s docking handle
The researchers first asked why some cell types from humans, dogs, horses, and cattle are killed by NetF while others are unharmed. By comparing gene activity in sensitive and resistant cells, and by repeatedly exposing cells to NetF until rare survivors took over, they found that resistant cells consistently produced much less of a surface protein called ANTXR2. This molecule was already famous as the main receptor for anthrax toxin. When the team knocked out ANTXR2 using CRISPR gene editing, cells became resistant to NetF. When they added extra human, canine, equine, or bovine ANTXR2, previously resistant cells became highly sensitive, confirming that NetF relies on ANTXR2 as its docking handle across several animal species.
Watching the pore assemble at near-atomic scale
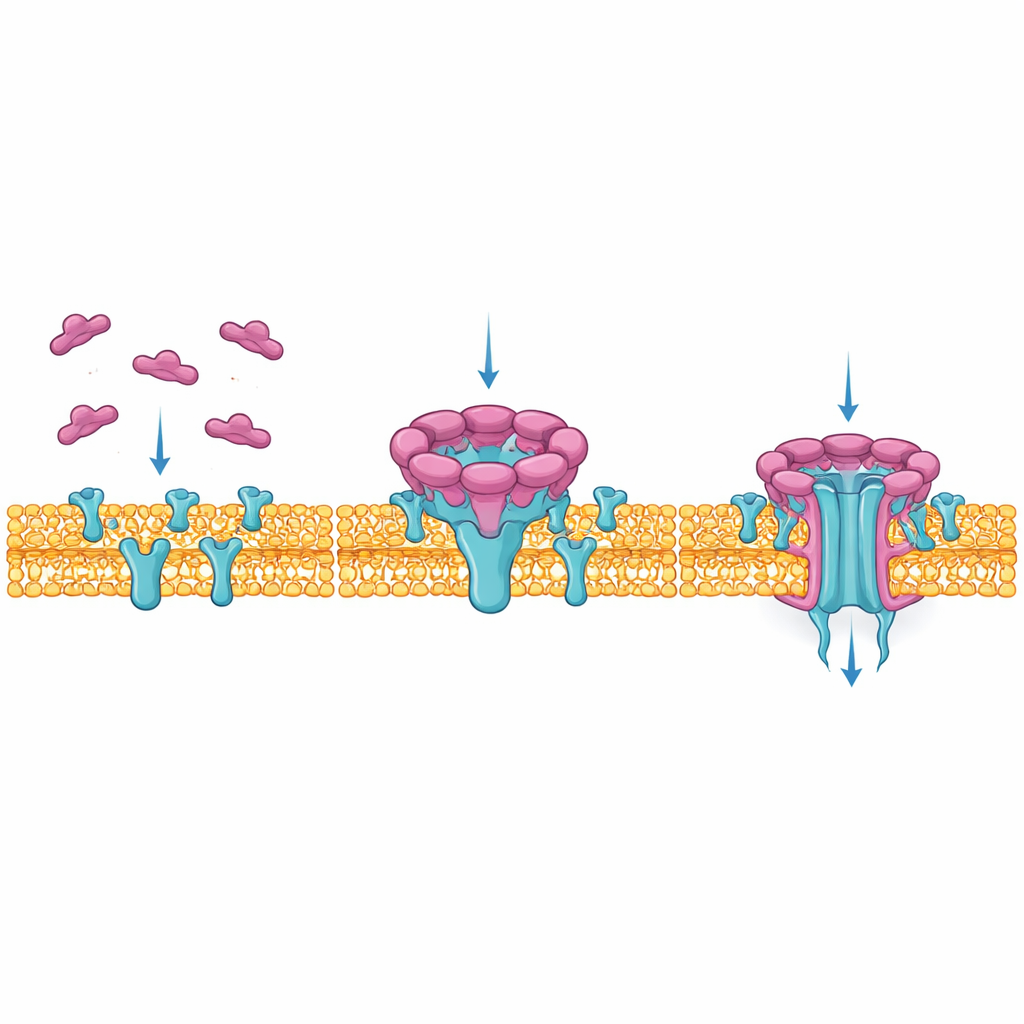
To see precisely how NetF builds its deadly channel, the authors used cryo‑electron microscopy, which images flash-frozen molecules at very high resolution. They re-created the toxin’s interaction with artificial membranes and found two main stages: a “pre‑pore,” where nine NetF units sit on the membrane surface, and a final pore, most often built from eight units that have pushed a long, tightly packed barrel of protein through the membrane. Comparing these stages showed how a flexible loop in each NetF molecule swings down and refolds into a 50‑ångström‑long barrel that matches the membrane’s thickness, creating a narrow, water‑filled tunnel through which ions and small molecules can pass.
How NetF grips both receptor and membrane
The team then solved the structure of NetF’s pore bound to the entire outer portion of ANTXR2. Unlike anthrax toxin, which grabs a metal‑binding site at the very tip of ANTXR2, NetF clamps onto the side of the receptor, contacting two different regions at once. This side‑on embrace positions the toxin close to the membrane and lets parts of NetF’s “rim” and “stem” dig into nearby lipids and cholesterol. The interface is unusually rich in tyrosine residues, which help stabilize both receptor binding and membrane anchoring. Subtle sequence differences in the receptor’s lower region, especially in mice, weaken this fit and help explain why mouse cells are naturally more resistant to NetF than cells from other mammals.
What these findings mean for disease
Taken together, the results show that NetF kills cells only when it can latch firmly onto ANTXR2 and at the same time engage surrounding membrane lipids to drive pore insertion. This dual recognition explains its strong activity in dogs and foals, where ANTXR2‑bearing cells in the gut are prime targets, and its weaker action in mice. More broadly, the study reveals a recurring strategy among pore‑forming toxins: binding close to the membrane through specific receptors that act as scaffolds for pore assembly. By mapping the exact contact points between NetF and ANTXR2, this work opens the door to designing molecules that could block the interaction, potentially protecting vulnerable animals from devastating intestinal disease.
Citation: Wang, C., Cattalani, F., Iacovache, I. et al. Identification and structural characterization of anthrax toxin receptor 2 as the Clostridium perfringens NetF receptor. Nat Commun 17, 2788 (2026). https://doi.org/10.1038/s41467-026-69526-6
Keywords: pore-forming toxin, Clostridium perfringens, NetF, ANTXR2 receptor, bacterial pathogenesis